Volume 11, Number 11—November 2005
Letter
Borrelia spielmanii Erythema Migrans, Hungary
To the Editor: Lyme disease is the most frequent tickborne human infection in the northern hemisphere. At least 5 species of the Borrelia burgdorferi sensu lato complex, B. burgdorferi sensu stricto, B. afzelii, B. garinii, B. bissettii, and B. lusitaniae, have a pathogenic role in human Lyme disease in central Europe (1–3). A sixth pathogenic strain, A14S, has been isolated from 1 Dutch (4) and 2 German patients with erythema migrans (5). This strain was also detected in 4 questing Ixodes ricinus ticks in Germany (6,7) and 1 in the Czech Republic (8). A14S has recently been described as a new species, B. spielmanii (9); its main reservoir host is probably the garden dormouse (Eliomys quercinus), but B. spielmanii could not be detected in mice or voles. Richter et al. (9) could not find ticks harboring B. spielmanii in 3 of 5 examined areas in Germany. They were present almost exclusively in a single area where the prevalence of infection with this genotype was 15 (6%) of 251. We describe the isolation of this novel Lyme disease spirochete from a human patient with erythema migrans in Hungary.
Since 1999, we have regularly isolated Borrelia burgdorferi sensu lato from skin biopsy specimens of erythema migrans and acrodermatitis chronica atrophicans taken from patients at the Center for Tick-borne Diseases, Budapest, Hungary. To identify the Borrelia species occurring in Hungarian Lyme disease patients, we have started to molecularly analyze cultured isolates that originate from erythema migrans of different patients. DNA was isolated from 8 bacterial pellets by using QIAamp DNA mini kit (Qiagen, Hilden, Germany). Primers BSL-F and BSL-R were used; these amplify an ≈250-bp region of the outer surface protein (osp) A gene from all Lyme disease spirochetes (10). We added 2 μL extracted DNA to a 20-μL reaction mixture composed of 1.0 U HotStartTaq DNA polymerase, 200 μmol/L of each dNTP, 25 pmol of each primer, and 1.5 mmol/L MgCl2 (HotStartTaq Master Mix, Qiagen). An initial denaturation step at 94°C for 15 min was followed by 40 cycles of denaturation at 94°C for 30 s, annealing at 58°C for 30 s, and extension at 72°C for 30 s. Final extension was done at 72°C for 5 min. Amplified DNA was subjected to electrophoresis in a 1.5% agarose gel that was prestained with ethidium bromide and viewed under UV light. After purification, the dideoxy chain termination (Applied Biosystems Division, Foster City, CA, USA) was used for sequencing. Obtained sequences were checked with Chromas v.1.45 and compared to sequence data available from GenBank by using BLAST (http://www.ncbi.nlm.nih.gov/BLAST/). New sequences were submitted to GenBank.
Six sequences (DQ007298, DQ007299, DQ007300, DQ007301, DQ007302, DQ007303) showed 100% homology to B. afzelii Khab 625 strain (AY502599). One (DQ007297) of the remaining 2 samples showed 99.6% similarity with B. burgdorferi B31 (AE000790), and the other (AY995900) showed 99.21% similarity with B. spielmanii (AF102057).
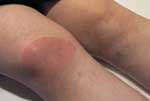
The patient whose culture showed B. spielmanii was a 42-year-old woman with a homogenous erythema migrans, diagnosed on September 24, 1999. The erythema was 10 cm in diameter on the front surface of the knee at the first visit (Figure). The immunoglobulin M (IgM) and IgG Borrelia immunoblot that applied B. afzelii (ACA1) antigen was negative in serum drawn on the seventh day after the appearance of erythema migrans. The patient did not remember a tick bite and had not traveled abroad during the previous 6 months. She complained of an "extremely unusual," intense, serous nasal discharge that started 3 weeks before the appearance of erythema migrans and of a moderate headache; both disappeared spontaneously 2 weeks before treatment.
Our results show at least 3 distinct species of B. burgdorferi sensu lato in Hungary. In addition to B. burgdorferi sensu stricto and B. afzelii, known throughout Europe, we detected the recently described species B. spielmanii among randomly selected samples. Together with 2 previous publications (4,5), our observation also suggest that B. spielmanii has a pathogenic role in human Lyme disease. Although B. spielmanii is distributed more focally than other species of the B. burgdorferi sensu lato complex (9), it occurs from the Netherlands through Germany and Czech Republic to Hungary (4,5,7,8).
Acknowledgment
We acknowledge the advice and the positive control samples provided by Martin J. Kenny.
References
- Postic D, Ras NM, Lane RS, Hendson M, Baranton G. Expanded diversity among Californian Borrelia isolates and description of Borrelia bissettii sp. nov. (formerly Borrelia group DN127). J Clin Microbiol. 1998;36:3497–504.PubMedGoogle Scholar
- Maraspin V, Cimperman J, Lotric-Furlan S, Ruzic-Sabljic E, Jurca T, Picken RN, Solitary borrelial lymphocytoma in adult patients. Wien Klin Wochenschr. 2002;114:515–23.PubMedGoogle Scholar
- Collares-Pereira M, Couceiro S, Franca I, Kurtenbach K, Schafer SM, Vitorino L, First isolation of Borrelia lusitaniae from a human patient. J Clin Microbiol. 2004;42:1313–8. DOIPubMedGoogle Scholar
- Wang G, van Dam AP, Dankert J. Phenotypic and genetic characterization of a novel Borrelia burgdorferi sensu lato isolate from a patient with Lyme borreliosis. J Clin Microbiol. 1999;37:3025–8.PubMedGoogle Scholar
- Fingerle V, Michel H, Schulte-Spechtel U, Göttner G, Hizo-Teufel C, Hofmann H, A14S—a new Borrelia burgdorferi s.l. genospecies as relevant cause of human disease [abstract]. Int J Med Microbiol. 2004;294(Suppl 1):207.
- Rauter C, Oehme R, Dietrich I, Engele M, Hartung T. Distribution of clinically relevant Borrelia genospecies in ticks assessed by a novel, single-run real-time PCR. J Clin Microbiol. 2002;40:36–43. DOIPubMedGoogle Scholar
- Michel HB, Wilske B, Hettche G, Göttner G, Heimerl C, Reischl U, An ospA-polymerase chain reaction/restriction fragment length polymorphism–based method for sensitive detection and reliable differentiation of all European Borrelia burgdorferi sensu lato species and OspA types. Med Microbiol Immunol (Berl). 2003;193:219–26. DOIPubMedGoogle Scholar
- Derdáková M, Beati L, Pet'ko B, Stanko M, Fish D. Genetic variability within Borrelia burgdorferi sensu lato genospecies established by PCR-single-strand conformation polymorphism analysis of the rrfA-rrlB intergenic spacer in Ixodes ricinus ticks from the Czech Republic. Appl Environ Microbiol. 2003;69:509–16. DOIPubMedGoogle Scholar
- Richter D, Schlee DB, Allgöwer R, Matuschka FR. Relationships of a novel Lyme disease spirochete, Borrelia spielmani sp. nov., with its hosts in central Europe. Appl Environ Microbiol. 2004;70:6414–9. DOIPubMedGoogle Scholar
- Demaerschalck I, Benmessaoud A, Dekesel M, Hoyois B, Lobet Y, Hoet P, Simultaneous presence of different Borrelia burgdorferi genospecies in biological fluids of Lyme-disease patients. J Clin Microbiol. 1995;33:602–8.PubMedGoogle Scholar
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 11, Number 11—November 2005
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
András Lakos, Center for Tick-borne Diseases, Visegrádi u. 14. H-1132 Budapest, Hungary; fax: 361-329-3898
Top