Volume 11, Number 6—June 2005
Dispatch
Rifampicin Resistance in Tuberculosis Outbreak, London, England
Abstract
Mycobacterium tuberculosis isolates cultured from 6 patients associated with an isoniazid-resistant M. tuberculosis outbreak acquired rifampicin resistance. The rpoB gene sequence showed that resistance was associated with rare mutations in each isolate. Three isolates had a mutation outside the rifampicin resistance–determining region.
In January 2000, 4 cases of smear-positive pulmonary tuberculosis (TB) caused by Mycobacterium tuberculosis in young men from the local community were identified during a 1-week period at a hospital in north London, United Kingdom (1). Three isolates were shown to be isoniazid-monoresistant TB. Further investigation showed 155 confirmed or probable cases, 132 in London and 23 outside London, which suggests a large outbreak of a unique strain in the London area (2). Confirmed case-patients were defined as patients with isolates of M. tuberculosis resistant to isoniazid that had the same band pattern on restriction length polymorphism typing (RFLP); these patients were residents of London at the time of their diagnosis, which had been made since January 1995 (2). Probable cases were defined as for confirmed cases except that isolates underwent rapid epidemiologic typing [RAPET] but are awaiting RFLP typing (2). RAPET is a rapid screening molecular typing method developed at the Mycobacterium Reference Unit (MRU) (3). In October 2002, the TB isolate from 1 patient associated with the outbreak had developed resistance to rifampicin. Initially, the isolate was tested with a commercial line probe rifampicin resistance–determining hybridization assay, Inno-LiPA (Innogenetics Belgium, Gent, Belgium). However, the line probe assay failed to identify rifampicin resistance in this isolate, and rifampicin resistance was only detected on phenotypic antimicrobial sensitivity testing at MRU. Subsequently, 5 additional rifampicin-resistant isolates from different patients associated with this outbreak were identified at MRU. The aim of this study was to determine the basis for the rifampicin resistance in these 6 isolates by sequencing the entire rpoB gene.
Rifampicin resistance can occur as a result of mutations on the rpoB gene that encodes the β-subunit of RNA polymerase (4). More than 95% of these mutations occur on an 81-bp fragment of the gene between bases 1276 and 1356 (432–458 in the rpoB gene of M. tuberculosis and codon 507–534 in the Escherichia coli rpoB gene) (5,6). This region is known as the rifampicin resistance–determining region (RRDR), or "hotspot," and is used as a target for direct sequencing and commercial line probe assays.
The isoniazid-monoresistant and multidrug-resistant tuberculosis (MDR-TB) isolates were obtained from the patients' source hospital or MRU. All isolates were identified as belonging to the same strain of M. tuberculosis (RAPET or IS6110 typing), and all drug-susceptibility testing was carried out at MRU according to standard procedures. The wildtype control isolate used for these studies was M. tuberculosis, H37Rv (ATCC, 9360 National Collection of Type Culture, London, UK). Three isolates, 018, 483, and 915, were from patients in whom MDR-TB developed as a result of poor compliance with therapy, whereas isolates 604, T7, and 371 were from patients who contracted primary MDR-TB.
DNA was prepared from the 6 isolates of M. tuberculosis by emulsifying 2–3 colonies in 400 μL Tris-EDTA buffer and heating the suspension in a water bath for 40 min at 80°C. Polymerase chain reaction (PCR) was performed on the extracted DNA with 6 sets of primers designed to amplify 6 overlapping fragments of the rpoB gene from the 6 M. tuberculosis isolates (Table). Ten microliters of DNA was added to the PCR mix containing 81.4 μL PCR-quality water, 10 μL potassium chloride buffer (Bioline Ltd., London, UK), 0.4 μL of each primer (100 mmol) (Sigma-Genosys Ltd., Haverhill, UK), 3 μL deoxynucleoside triphosphates (5 mmol), and 1 μL Taq (Bioline). The amplification was performed on a Techgene thermal cycler (Techne, Princeton, NJ, USA). PCR products were separated by gel electrophoresis on a 1.5% agarose gel, and DNA bands were stained with ethidium bromide. Primers and excess nucleotides were removed from the amplified DNA with a PCR clean-up kit (Qiagen, Inc., Valencia, CA, USA). The amount of DNA in the cleaned-up product was quantified by comparing the intensity of the band to bands of known intensity in a HyperLadder marker (Bioline).
Forward and reverse cycle sequencing reactions were performed with the Big Dye Terminator Cycle Sequencing Ready Reaction DNA sequencing kit (Applied Biosystems, Inc., Foster City, CA, USA). Briefly, 40 ng of cleaned-up DNA was added to 10.8 μL PCR-quality water, 3 μL buffer, 3.2 μL of forward or reverse 1 mmol primer, and 1 μL of cycle sequencing ready reaction mix. The labeled DNA was precipitated by adding 14.5 μL PCR-quality water, 62.5 μL 95% ethanol, and 3 μL sodium acetate solution (2.3 mol/L) and centrifuging (13,000 × g, 15 min, 4°C). The supernatant was removed with a fine-tipped pipette, and the pellet was cleaned with 200 μL 70% ethanol and then recentrifuged (13,000 × g, 15 min, 4°C). Again the supernatant was removed, and the pellet was dried at 37°C for 30 min. Four microliters of formamide and 1 μL of dextran loading buffer were added to each pellet, and 1.5 μL of sample was added to each well of the sequencing gel. The 6 fragments of the rpoB gene from each of the 6 isolates were then sequenced with an ABI 377 Applied Biosystems sequencer. The sequences obtained from each isolate were joined together to form a continuous whole gene sequence, aligned with ClustalW (http://www.ebi.ac.uk/clustalw/) and compared to the wildtype to identify base-pair mismatches.
In addition, all 6 isolates were tested with a line probe resistance–determining hybridization assay, Inno-LiPA (Innogenetics Belgium), as described in the manufacturer's instructions. Briefly, an 81-bp region of the rpoB gene was amplified with biotinylated primers, which yielded a biotinylated target sequence, and hybridized with specific oligonucleotide probes immobilized on a parallel strip. After hybridization, streptavidin labeled with alkaline phosphatase was used to detect any hybrids. Inno-LiPA consists of 10 oligonucleotide probes (19–23 bases in length), encompassing the 81-bp region (RRDR) of the rpoB gene. One is specific for M. tuberculosis complex, whereas the other 5 partially overlapping wildtype probes (S1–S5) cover the region from positions 507 to 534 of the rpoB gene. These S-probes hybridize to the wildtype (rifampicin-sensitive) DNA sequence. Failure of any of these S-probes to hybridize indicates that a mutation has occurred. Four other probes (R2, R4a, R4b, and R5) are specific for amplicons carrying the most common rpoB mutation that confers rifampicin resistance.
The results of the Inno-LiPA assay showed 5 of the 6 isolates, which were phenotypically rifampicin-resistant, were negative. The target DNA hybridized to all 5 wildtype S-probes and none of the R-probes, which demonstrated that the assay failed to detect rifampicin resistance in these 5 isolates. The Inno-LiPA assay cannot detect mutations outside the RRDR. Failure to detect rare mutations within the RRDR may be caused by nonspecific hybridization of the wildtype S-probes because of slight fluctuations in temperature during the hybridization process. Isolate 483 showed a weak DNA hybridization reaction with the S4 probe, indicating that a mutation was present on codon 451 (codon 526 in Escherichia coli). The exact nature of the mutation could not be determined with this assay.
Analysis of the sequence data identified specific mutations in rpoB in all 6 strains studied (Figure 1). Three had mutations within the RRDR, and of these, 2 were C-to-G mutations at codon 456, inducing a serine to tryptophan amino acid conversion (S456W). The third was an A-to-G mutation at codon 451, resulting in a change from histidine to arginine (H451R) (Figure 1). No other mutations were identified in the DNA sequences outside the RRDR in these 3 isolates. The 3 other isolates had the mutation G to T at codon 176, outside the RRDR, which caused a change from valine to phenylalanine (V176F). Neither RRDR mutations nor any other mutations were found in the rpoB gene sequences of these 3 isolates.
Of the mutations found within the RRDR, H526R occurs in <4%, and S531W occurs in ≈1.4% of all rifampicin-resistant isolates, respectively (7). Mutations outside the RRDR account for <4% of rifampicin resistance, and few have been described (7–9). In a previous study (7), V176F was found in 5 of 18 isolates with no mutations in the RRDR from Asia (9,10) and Africa (11,12). To our knowledge, this is the first time this type of mutation has been detected in strains isolated in the United Kingdom.
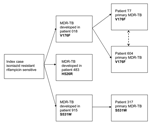
Three different mutations in the rpoB gene, V176F, H526R and S531W, were detected in the isolates associated with the isoniazid-resistant outbreak. Given that all the mutations found in this study are rare and that MDR-TB developed during treatment in 3 patients (018, 483, and 915) (Figure 2), the rifampicin-resistance mutations observed in these 3 patients likely occurred on 3 independent occasions, as a result of poor compliance with therapy. Two of these patients, 018 and 915, were contacts of the 3 patients who subsequently had primary MDR-TB (patients T7 and 604 [V176F] and 317 [S531W]) (Figure 2).
Our study highlights problems associated with using a line probe hybridization assay to detect rifampicin resistance in M. tuberculosis. The Inno-LiPA assay cannot detect mutations outside the RRDR, and failure to detect rare mutations within the RRDR may be caused by nonspecific hybridization of the wildtype S-probes because of slight fluctuations in temperature during the hybridization process. In this study, all the rare mutations, inside and outside the RRDR, were detected by sequencing the entire rpoB gene. The inability to identify MDR-TB isolates results in treatment failure and increased risk for transmission of resistant disease in the community. We recommend that, when the index of suspicion for MDR-TB is high and the line probe assays fail to detect mutations conferring rifampicin resistance, the entire rpoB gene should be sequenced to prevent unnecessary delay in diagnosing MDR-TB.
Dr. Jenkins works as a registered clinical scientist in the Department of Microbiology at the Royal Free Hospital in London. Her research involves molecular typing and identification of pathogenic bacteria in general, with a specific interest in Mycobacterium species.
Acknowledgment
We are grateful to Francis Drobniewski and Malcolm Yates for providing us with the MDR-TB isolates and to Sarah Batt and Clare Ling for their help with sequencing.
References
- Ruddy MC, Davies AP, Yates MD, Yates S, Balasegaram S, Drabu Y, Outbreak of isoniazid resistant tuberculosis in north London. Thorax. 2004;59:279–85. DOIPubMedGoogle Scholar
- Davies AP, Ruddy MC, Neely F, Ruggles R; Outbreak Control Committee. Outbreak of isoniazid resistant tuberculosis in north London 1999–2004. Dulwich, London: Health Protection Agency Regional Epidemiology Unit; 2004.
- Yates MD, Drobniewski FA, Wilson SM. Evaluation of a rapid PCR-based epidemiological typing method for routine studies of Mycobacterium tuberculosis. J Clin Microbiol. 2002;40:712–4. DOIPubMedGoogle Scholar
- Jin DJ, Gross CA. Mapping and sequencing of mutations in the Escherichia coli rpoB gene that lead to rifampicin resistance. J Mol Biol. 1988;202:45–58. DOIPubMedGoogle Scholar
- Miller LP, Crawford JT, Shinnick TM. The rpoB gene of Mycobacterium tuberculosis. Antimicrob Agents Chemother. 1994;38:805–11.PubMedGoogle Scholar
- Ramaswamy S, Musser JM. Molecular genetic basis of antimicrobial agent resistance in Mycobacterium tuberculosis: 1998 update. Tuber Lung Dis. 1998;79:3–29. DOIPubMedGoogle Scholar
- Heep M, Brandstatter B, Rieger U, Lehn N, Richter E, Rusch-Gerdes S, Frequency of rpoB mutations inside and outside the cluster I region in rifampin-resistant clinical Mycobacterium tuberculosis isolates. J Clin Microbiol. 2001;39:107–10. DOIPubMedGoogle Scholar
- Kiepiela P, Bishop K, Kormuth E, Roux L, York DF. Comparison of PCR-heteroduplex characterization by automated DNA sequencing and line probe assay for the detection of rifampicin resistance in Mycobacterium tuberculosis isolates from KwaZulu-Natal, South Africa. Microb Drug Resist. 1998;4:263–9. DOIPubMedGoogle Scholar
- Hirano K, Abe C, Takahashi M. Mutations in the rpoB gene of rifampin-resistant Mycobacterium tuberculosis strains isolated mostly in Asian countries and their rapid detection by line probe assay. J Clin Microbiol. 1999;37:2663–6.PubMedGoogle Scholar
- Yang B, Koga H, Ohno H, Ogawa K, Fukuda M, Hirakata Y, Relationship between antimycobacterial activities of rifampicin, rifabutin and KRM-1648 and rpoB mutations of Mycobacterium tuberculosis. J Antimicrob Chemother. 1998;42:621–8. DOIPubMedGoogle Scholar
- Caugant DA, Sandven P, Eng J, Jeque JT, Tonjum T. Detection of rifampin resistance among isolates of Mycobacterium tuberculosis from Mozambique. Microb Drug Resist. 1995;1:321–6. DOIPubMedGoogle Scholar
- Cockerill FR III, Williams DE, Eisenach KD, Kline BC, Miller LK, Stockman L, Prospective evaluation of the utility of molecular techniques for diagnosing nosocomial transmission of multidrug-resistant tuberculosis. Mayo Clin Proc. 1996;71:221–9. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 11, Number 6—June 2005
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Claire Jenkins, Department of Medical Microbiology, Royal Free Hospital, London, NW3 2QG, United Kingdom; fax: 44-20-7794-4433
Top