Volume 14, Number 7—July 2008
Research
Transmission of Bartonella henselae by Ixodes ricinus
Figure 3
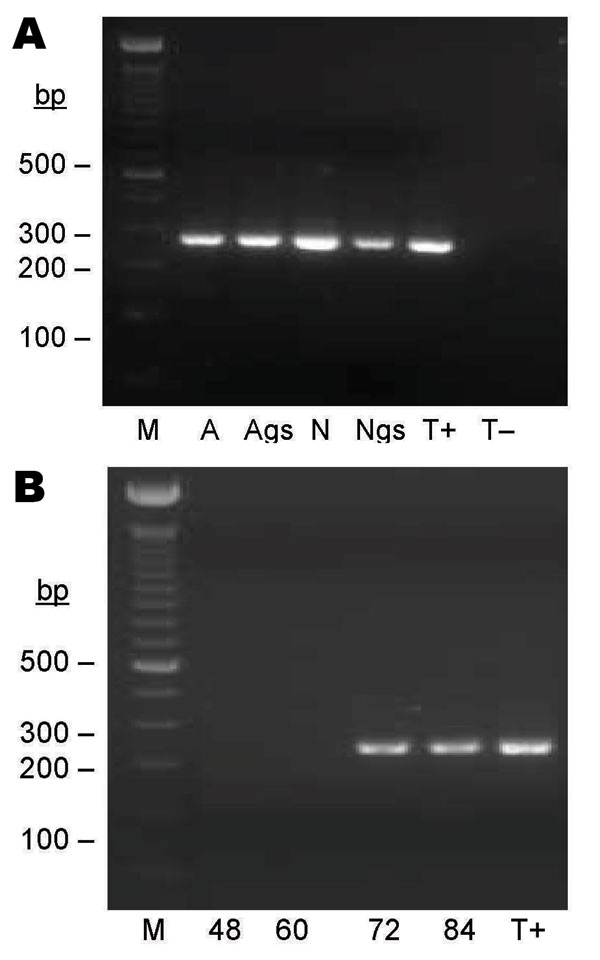
Figure 3. Seminested PCR detection of Bartonella spp. DNA after partial refeeding of infected ticks. A) Bartonella spp. DNA detection in Ixodes ricinus ticks fed on B. henselae–infected blood at previous development stages and refed for 84 h on uninfected blood. Lane M, 100-bp DNA molecular mass; lane A, carcass of female adult; lane Ags, salivary glands of female adult, lane N, carcass of nymph; lane Ngs, salivary glands of nymph; lane T+, B. bacilliformis DNA; lane T–, nymph fed on uninfected ovine blood. B) Bartonella spp. DNA detection in blood isolated from feeders. Lane M, 100-bp DNA molecular mass marker; lane 48, ovine blood after 48 h of tick attachment on skin; lane 60, ovine blood after 60 h of tick attachment on skin; lane 72, ovine blood after 72 h of tick attachment on skin; lane 84, ovine blood after 84 h of tick attachment on skin; lane T+, B. bacilliformis DNA.