Volume 15, Number 12—December 2009
Dispatch
Molecular Model of Prion Transmission to Humans
Abstract
To assess interspecies barriers to transmission of transmissible spongiform encephalopathies, we investigated the ability of disease-associated prion proteins (PrPd) to initiate conversion of the human normal cellular form of prion protein of the 3 major PRNP polymorphic variants in vitro. Protein misfolding cyclic amplification showed that conformation of PrPd partly determines host susceptibility.
The agents responsible for the transmissible spongiform encephalopathies (TSEs) are called prions. Although their precise biochemical composition is a matter of debate, they are known to occur in a series of strains, each with a characteristic disease phenotype and host range (1). A central event in neuropathogenesis of TSEs is conversion of the normal cellular form of the prion protein (PrPC) to the pathognomonic disease-associated isoform (PrPd) (2). In the absence of a known nucleic acid genome, it has been proposed that the strain-like properties of different TSE agents are encoded by distinct self-propagating conformational variants (conformers) of PrPd (3). The best developed method available for typing these PrPd isoforms uses limited proteolysis and classification of the protease-resistant prion protein (PrPres) in terms of the sizes of the nonglycosylated fragment(s) produced and the ratio of the 3 possible glycoforms (3). If distinct conformers and glycotypes of PrPd are responsible for diversity of prion strains, then they would be expected to be able to impose these molecular characteristics onto PrPC of the same amino acid sequence (when transmitted or replicating within a species) and onto PrPC of a different primary sequence (when transmitted between species). In support of this theory, the agent responsible for the TSE of cattle, called bovine spongiform encephalopathy (BSE), the accepted cause of variant Creutzfeldt-Jakob disease (vCJD) in humans (4), has been shown to be transmissible to at least 7 species (1), resulting in propagation of PrPd that retains the characteristic molecular signature of the original BSE prion strain (5–7).
Current thinking favors a seeded polymerization model for the conversion of PrPC into PrPd, which has led to the development of several cell-free in vitro conversion model systems (8). One such system is protein misfolding cyclic amplification (PMCA) (9), in which small amounts of PrPd introduced (seeded) into substrate containing excess PrPC and other essential conversion cofactors can be amplified to readily detectable levels by sequential cycles of sonication and incubation. We have previously reported that the molecular characteristics, electrophoretic mobility, and glycoform ratio of the PrPres associated with the vCJD PrPd conformer were faithfully reproduced by PMCA (10). However, the efficiency of amplification achieved depended on the substrate’s prion protein gene codon 129 (PRNP-129) genotype. The most efficient amplification was achieved in a methionine homozygous (PRNP-129MM) substrate; the least efficient, in a valine homozygous (PRNP-129VV) substrate. To estimate the molecular component of transmission barriers for particular TSE agents between species, we used PMCA reactions to amplify PrPd associated with vCJD (10), bovine BSE (11), ovine scrapie (12), and experimental ovine BSE (13) and substrates prepared from humanized transgenic mouse brain tissue expressing each of the 3 main PRNP polymorphic variants found in Caucasian human populations (PRNP-129MM, MV, and VV) (14).
We prepared seed and substrate homogenates as 10% (wt/vol) homogenates in PMCA conversion buffer (10). Seed homogenates were diluted into substrate homogenates so that all PMCA reactions contained equivalent amounts of PrPd based on the PrPres levels in each seed homogenate. PrPres levels were determined by Western blot titration that used monoclonal antibody (MAb) 6H4 after limited proteinase K digestion. The reaction mixes were split into 2 aliquots; 1 aliquot was stored immediately at –80°C (−PMCA), and the other was subjected to 48 cycles of PMCA (+PMCA) (10). To assess the degree of PrPd amplification achieved from each seed in each substrate, the samples −/+ PMCA were subjected to limited proteinase K digestion, and PrPres was detected by Western blotting with MAb 6H4 (which recognizes human, bovine, and ovine PrP) and MAb 3F4 (which selectively recognizes only human PrP and would therefore specifically identify PrPres formed from human PrPC).
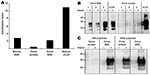
Using MAb 6H4 to probe Western blots, we noted amplification of vCJD, bovine BSE, and ovine BSE PrPres in the PRNP-129MM substrate (Figure 1, panel A, top) but not in the PRNP-129VV substrate (Figure 1, panel A, bottom). Semiquantitative assessment of these Western blots by densitometry showed that the degree of amplification of vCJD PrPres was considerably greater than that of bovine or ovine BSE in the PRNP-129MM substrate (Figure 2, panel A). A more sensitive and discriminatory Western blot conducted by using MAb 3F4 confirmed efficient amplification of vCJD, bovine BSE, and ovine BSE PrPres in the PRNP-129MM substrate (Figure 1, panel B, top), weaker amplification in the PRNP-129MV substrate (Figure 1, panel B, middle), and little, if any, amplification in the PRNP-129VV substrate (Figure 1, panel B, bottom). In all substrates, the amplified PrPres retained the electrophoretic mobility and glycoform ratio associated with BSE-related PrPres. No amplification of ovine scrapie PrPres was evident after PMCA in any of the PRNP humanized transgenic mouse brain substrates (Figure 1, panels A, B). The difference between ovine scrapie and ovine BSE in ability to seed amplification in PRNP-129MM substrate was a robust phenomenon evident in brain samples from 3 different ARQ/ARQ sheep with each disease (Figure 2, panel B). However, failure of the ovine scrapie seed to amplify was not caused by a general lack of competence to do so or by inappropriate amplification conditions because robust amplification of ovine scrapie PrPres was evident after PMCA in a substrate prepared from normal ARQ/ARQ sheep brain (Figure 2, panel C).
Our results are best appreciated in terms of the molecular interaction between seed PrPd and substrate PrPC, specifically the species-specific amino acid sequence and PRNP polymorphic status of PrPC and PrPd and the PrPd conformers involved (Table). Regardless of the seed PrP amino acid sequence, the PrPd conformers associated with bovine BSE, ovine BSE, and human vCJD were amplified in the humanized mouse substrate and displayed similar PRNP-129 genotype preferences (PRNP-129MM >PRNP-129MV >PRNP-129VV). In contrast, the PrPd conformer associated with the ovine scrapie strain, although sharing the same PrP amino acid sequence as the PrPd in ovine BSE, could not be amplified in any of the PRNP humanized mouse substrates but could be amplified in a sheep brain substrate. These observations are consistent with conformation of a TSE agent’s PrPd (rather than solely its amino acid sequence) having a role in determining the susceptibility of a host’s PrPC to conversion. They similarly suggest that these molecular factors could in turn have a powerful influence on disease susceptibility and incubation time.
To date, all clinical cases of vCJD have occurred in persons with the PRNP-129MM genotype, as might be predicted from the efficiency of amplification of BSE-related PrPd shown here. Extrapolating from these results, one would predict that the next genotypic group most likely to show susceptibility to the BSE agent would be heterozygous (MV) at codon 129 of the PRNP gene, as previously suggested from the corresponding in vivo transmission studies (14).
In the wake of BSE epidemics in the United Kingdom and elsewhere, enhanced surveillance has identified apparently new TSEs (15), raising concerns regarding animal and human health. PMCA with suitable substrate sources could provide a rapid way to estimate the molecular component of transmission barriers for particular TSE agents between species, including humans. These estimates could thus indicate whether, like classical scrapie, the agents represent little risk for human health or whether, like classical BSE, they represent cause for concern.
Dr Jones is a postdoctoral research fellow at the National CJD Surveillance Unit, University of Edinburgh. His primary research interests are the application of in vitro PrPd amplification techniques, such as PMCA, to prion disease research in general and incorporation of these techniques into a confirmatory screening assay to detect vCJD-associated PrPd in human plasma as a surrogate marker of vCJD infectivity in blood.
Acknowledgment
This work was funded by the European Network of Excellence NeuroPrion (FOOD-CT-2004-506579), the Scottish National Blood Transfusion Services, and the Chief Scientist Office of the Scottish Government (CZB/4/357). The National CJD Surveillance Unit is funded by the Department of Health and the Scottish Government.
References
- Collinge J, Clark AR. A general model of prion strains and their pathogenicity. Science. 2007;318:930–6. DOIPubMedGoogle Scholar
- Will RG, Ironside JW, Zeidler M, Cousens SN, Estibeiro K, Alperovitch A, Lancet. 1996;347:921–5. DOIPubMedGoogle Scholar
- Collinge J, Sidle KC, Meads J, Ironside J, Hill AF. Molecular analysis of prion strain variation and the aetiology of ‘new variant’ CJD. Nature. 1996;383:685–90. DOIPubMedGoogle Scholar
- Head MW, Bunn TJ, Bishop MT, McLoughlin V, Lowrie S, McKimmie J, Prion protein heterogeneity in sporadic but not variant Creutzfeldt-Jakob disease: UK cases 1991–2002. Ann Neurol. 2004;55:851–9. DOIPubMedGoogle Scholar
- Eloit M, Adjou K, Coulpier M, Fontaine JJ, Hamel R, Lilin T, BSE agent signatures in a goat. Vet Rec. 2005;156:523–4.PubMedGoogle Scholar
- Supattapone S. Prion protein conversion in vitro. J Mol Med. 2004;82:348–56. DOIPubMedGoogle Scholar
- Saborio GP, Permanne B, Soto C. Sensitive detection of pathological prion protein by cyclic amplification of protein misfolding. Nature. 2001;411:810–3. DOIPubMedGoogle Scholar
- Jones M, Peden AH, Prowse CV, Groner A, Manson JC, Turner ML, In vitro amplification and detection of variant Creutzfeldt-Jakob disease PrPSc. J Pathol. 2007;213:21–6. DOIPubMedGoogle Scholar
- Yull HM, Ritchie DL, Langeveld JP, van Zijderveld FG, Bruce ME, Ironside JW, Detection of type 1 prion protein in variant Creutzfeldt-Jakob disease. Am J Pathol. 2006;168:151–7. DOIPubMedGoogle Scholar
- Jeffrey M, Martin S, Thomson JR, Dingwall WS, Begara-McGorum I, Gonzalez L. Onset and distribution of tissue prp accumulation in scrapie-affected Suffolk sheep as demonstrated by sequential necropsies and tonsillar biopsies. J Comp Pathol. 2001;125:48–57. DOIPubMedGoogle Scholar
- Gonzalez L, Chianini F, Martin S, Siso S, Gibbard L, Reid HW, Comparative titration of experiment ovine BSE infectivity in sheep and mice. J Gen Virol. 2007;88:714–7. DOIPubMedGoogle Scholar
- Bishop MT, Hart P, Aitchison L, Baybutt HN, Plinston C, Thomson V, Predicting susceptibility and incubation time of human-to-human transmission of vCJD. Lancet Neurol. 2006;5:393–8. DOIPubMedGoogle Scholar
- Manson JC, Cancellotti E, Hart P, Bishop MT, Barron RM. The transmissible spongiform encephalopathies: emerging and declining epidemics. Biochem Soc Trans. 2006;34:1155–8. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 15, Number 12—December 2009
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Michael Jones, National CJD Surveillance Unit, School of Molecular and Clinical Medicine, University of Edinburgh, Western General Hospital, Edinburgh, Scotland EH4 2XU, UK
Top