Volume 19, Number 1—January 2013
Dispatch
Klebsiella pneumoniae Antimicrobial Drug Resistance, United States, 1998–2010
Abstract
We studied antimicrobial-resistant Klebsiella pneumoniae for 1998–2010 by using data from The Surveillance Network. Susceptibility results (n = 3,132,354) demonstrated significant increases in resistance to all antimicrobial drugs studied, except tetracycline. Cross-resistance among carbapenem-resistant K. pneumoniae was lower for tetracycline and amikacin.
Klebsiella spp. are among the most common pathogens isolated in intensive care units (ICUs), and K. pneumoniae is the most frequently encountered carbapenemase-producing Enterobacteriaceae (1). Increasing antimicrobial drug resistance, including carbapenem-resistant K. pneumoniae (CRKP), accounts for substantial increases in illness and death (1). Few antimicrobial therapy options exist for infections caused by CRKP (2).
The emergence of K. pneumoniae resistance to carbapenems is well documented (3). However, few studies have analyzed the trends and prevalence of in vitro K. pneumoniae antimicrobial drug resistance since carbapenem resistance emerged in the United States during the late 1990s (4). Furthermore, few investigations have examined antimicrobial drug resistance with regard to specimen source or cross-resistance patterns among CRKP.
We examined the prevalence of K. pneumoniae antimicrobial drug resistance in US inpatients using a large national surveillance system. Our objectives were to analyze K. pneumoniae antimicrobial drug resistance among US inpatients, resistance patterns by specimen source, and cross-resistance among imipenem-resistant K. pneumoniae isolates.
We examined inpatients’ antimicrobial susceptibility test results from The Surveillance Network (TSN) Database-USA (Eurofins Medinet, Chantilly, VA, USA) for 1998–2010. TSN is a nationally representative repository of antimicrobial susceptibility results from ≈200 community, government, and university health care institutions in the United States and has been used in investigations of trends and prevalences of antimicrobial drug resistance (5). Susceptibility testing of isolates is conducted onsite by using Food and Drug Administration (FDA)–approved testing methods and interpreted by using Clinical Laboratory Standards Institute breakpoint criteria for all agents except tigecycline, for which FDA breakpoints were used. Details of quality control in TSN Database-USA have been described (6). No institutional review board approval was needed for this research because no personal identifying information was collected.
K. pneumoniae antimicrobial susceptibility results were stratified by specimen source (blood, sputum, urine, and wounds). Imipenem-resistant K. pneumoniae isolates from 2010 were examined for cross-resistance to other antimicrobial agents and prevalence in ICU versus non-ICU settings. We used χ2 testing to determine whether changes in K. pneumoniae antimicrobial drug resistance were statistically significant from 2000 to 2010 and whether 2010 antimicrobial drug resistance differed by specimen source. The α level was set at 0.05. Analyses were performed by using R version 2.11.0 (www.r-project.org).
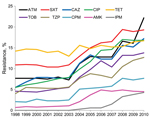
We analyzed a total of 3,132,354 K. pneumoniae antimicrobial susceptibility results for 1998–2010 (Table 1). Statistically significant increases in antimicrobial drug resistance to all agents (p<0.0001) except tetracycline (p = 0.0745) (Figure 1) were observed. Resistance to imipenem first appeared in TSN Database-USA in 2004 and rose gradually to 4.3% by the end of our study period. In 2010, K. pneumoniae resistance to tigecycline was 2.6% (data not shown). The largest increases in antimicrobial drug resistance from 1998 to 2010 were observed for aztreonam (7.7% to 22.2%), ceftazidime (5.5% to 17.2%), and ciprofloxacin (5.5% to 16.8%). Changes in resistance were smaller for tetracycline (14.2% to 16.7%) and amikacin (0.7% to 4.5%).
In 2010, isolates from the lower respiratory tract showed higher levels of resistance than did isolates from urine for all antimicrobial agents (p<0.0001) except tetracycline (p = 0.54) (Table 2). CRKP was more prevalent in ICU settings than in non-ICU settings (6.3% vs. 3.8%, respectively) (Technical Appendix).
Imipenem-resistant isolates of K. pneumoniae showed the lowest resistance to tetracycline (19.9%) and amikacin (36.8%). High prevalence of cross-resistance was observed for ciprofloxacin (96.4%) (Figure 2).
In our study, the proportion of K. pneumoniae isolates resistant to carbapenems was lower than those previously reported (7,8). In 2010, we observed a resistance rate of 4.3% for imipenem. The Centers for Disease Control and Prevention (CDC) reported that, among health care–associated infections, 8% of Klebsiella spp. isolates were carbapenem resistant in 2007 compared with <1% in 2000 (9). Most studies of K. pneumoniae antimicrobial drug resistance have focused on patient populations with higher exposures to antimicrobial agents, such as those in critical care and academic hospital settings. In contrast, the lower prevalence of CRKP in our study might have resulted from a wider variety of institution types and inclusion of isolates from hospital patients outside of the critical care setting. Furthermore, within our study, a high percentage of isolates were from urine and showed lower levels of resistance than did isolates from respiratory samples. Interpretive breakpoint criteria for the antimicrobial agents included did not change during the study period.
The low cross-resistance to tetracycline among CRKP and stable resistance rate of K. pneumoniae to this agent during the study period are noteworthy. In our analysis of cross-resistance among imipenem-resistant K. pneumoniae, tetracycline had the greatest antimicrobial activity against CRKP. Although resistance of K. pneumoniae increased for all antimicrobial agents studied, resistance to tetracycline increased only slightly from 1998 to 2010. Later-generation tetracyclines may prove useful in the treatment of CRKP-related infections because of their improved tissue penetration, antimicrobial activity, and decreased propensity to develop antimicrobial drug resistance compared with their older counterparts (10). Tigecycline, a glycylcycline antimicrobial agent that is structurally similar to tetracycline, has been used to treat CRKP-related infections and is often active against carbapenemase–producing K. pneumoniae (11,12). Data for tigecycline that used FDA interpretive breakpoints showed K. pneumoniae antimicrobial drug resistance was 2.6% in 2010. Tigecycline data were included only for 2010 because the drug was not FDA approved until 2005 and an insufficient number of results were available before 2010.
The widespread transmission of carbapenemase-producing K. pneumoniae has become the most common cause of carbapenem resistance among Enterobacteriaceae in the United States (13) and probably accounts for most of the imipenem resistance shown in this study. The spread of carbapenemase-producing organisms threatens to extend carbapenem resistance to the community (14). The increasing antimicrobial drug resistance to K. pneumoniae in our study, a concurrent lack of novel antimicrobial agent development (15), and limited therapeutic options available for treating CRKP-related infections add further urgency to improve prevention efforts and treatment strategies.
Our study data have strengths and limitations. The strengths are the wide variety of antimicrobial agents included, the number of laboratories reporting data, the nationally representative geographic distribution of these institutions, and the large number of isolates. Geography is a critical consideration with surveillance of this organism because distribution of K. pneumoniae antimicrobial drug resistance varies within the United States (13). The limitations of these data include a lack of central laboratory testing and the variety of test methods used. Because of a lack of Clinical Laboratory Standards Institute or FDA interpretive breakpoints for K. pneumoniae and colistin or fosfomycin, these data were not collected by TSN Database-USA and were not included in this study. Resistance to carbapenems might have been underreported at the beginning of our study period because of a lower frequency of susceptibility testing of these agents and the inability of antimicrobial susceptibility test methods to detect low-level carbapenem resistance.
Our study shows that K. pneumoniae antimicrobial drug resistance increased for every antimicrobial class studied except tetracyclines. Cross-resistance among imipenem-resistant K. pneumoniae was high for ciprofloxacin but lower for tetracycline and amikacin. This emerging problem presents a major threat to public health and warrants due diligence in future surveillance efforts.
Mr Sanchez is studying medicine and epidemiology as a physician assistant student at The George Washington University. His research interests are infectious disease epidemiology and emerging antimicrobial drug resistance.
Acknowledgments
We thank Eurofins-Medinet for providing the data and recognize Alexis M. Baird and Jacqueline N. Wilkins for their contributions in editing and formatting this work.
No authors received financial support for doing this research, nor do they have any conflicts of interest to disclose. No organizations have a financial interest in the completion of this work.
References
- Won SY, Munoz-Price LS, Lolans K, Hota B, Weinstein RA, Hayden MK, Emergence and rapid regional spread of Klebsiella pneumoniae carbapenemase-producing Enterobacteriaceae. Clin Infect Dis. 2011;53:532–40. DOIPubMedGoogle Scholar
- Schwaber MJ, Carmeli Y. Carbapenem-resistant Enterobacteriaceae: a potential threat. JAMA. 2008;300:2911–3. DOIPubMedGoogle Scholar
- Naas T, Nordmann P, Vedel G, Poyart C. Plasmid-mediated carbapenem-hydrolyzing beta-lactamase KPC in a Klebsiella pneumoniae isolate from France. Antimicrob Agents Chemother. 2005;49:4423–4. DOIPubMedGoogle Scholar
- Rossi F, Baquero F, Hsueh P-R, Paterson DL, Bochicchio GV, Snyder TA, In vitro susceptibilities of aerobic and facultatively anaerobic gram-negative bacilli isolated from patients with intra-abdominal infections worldwide: 2004 results from SMART (Study for Monitoring Antimicrobial Resistance Trends). J Antimicrob Chemother. 2006;58:205–10. DOIPubMedGoogle Scholar
- Sanchez GV, Master RN, Karlowsky JA, Bordon JM. In vitro antimicrobial resistance of urinary E. coli among U.S. outpatients from 2000 to 2010. Antimicrob Agents Chemother. 2012;56:2181–3. DOIPubMedGoogle Scholar
- Sahm DF, Marsilio MK, Piazza G. Antimicrobial resistance in key bloodstream bacterial isolates: electronic surveillance with the Surveillance Network Database–USA. Clin Infect Dis. 1999;29:259–63. DOIPubMedGoogle Scholar
- Bratu S, Mooty M, Nichani S, Landman D, Gullans C, Pettinato B, Emergence of KPC-possessing Klebsiella pneumoniae in Brooklyn, New York: epidemiology and recommendations for detection. Antimicrob Agents Chemother. 2005;49:3018–20. DOIPubMedGoogle Scholar
- Rhomberg PR, Jones RN. Summary trends for the Meropenem Yearly Susceptibility Test Information Collection Program: a 10-year experience in the United States (1999–2008). Diagn Microbiol Infect Dis. 2009;65:414–26. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Guidance for control of infections with carbapenem-resistant or carbapenemase-producing Enterobacteriaceae in acute care facilities. MMWR Morb Mortal Wkly Rep. 2009;58:256–60 .PubMedGoogle Scholar
- Agwuh KN, MacGowan A. Pharmacokinetics and pharmacodynamics of the tetracyclines including glycylcyclines. J Antimicrob Chemother. 2006;58:256–65. DOIPubMedGoogle Scholar
- Poulakou G, Kontopidou FV, Paramythiotou E, Kompoti M, Katsiari M, Mainas E, Tigecycline in the treatment of infections from multi-drug resistant gram-negative pathogens. J Infect. 2009;58:273–84. DOIPubMedGoogle Scholar
- Humphries RM, Kelesidis T, Dien Bard J, Ward KW, Bhattacharya D, Lewinski MA. Successful treatment of pan-resistant Klebsiella pneumoniae pneumonia and bacteraemia with a combination of high-dose tigecycline and colistin. J Med Microbiol. 2010;59:1383–6. DOIPubMedGoogle Scholar
- Kitchel B, Sundin DR, Patel JB. Regional dissemination of KPC-producing Klebsiella pneumoniae. Antimicrob Agents Chemother. 2009;53:4511–3. DOIPubMedGoogle Scholar
- Gupta N, Limbago BM, Patel JB, Kallen AJ. Carbapenem-resistant Enterobacteriaceae: epidemiology and prevention. Clin Infect Dis. 2011;53:60–7. DOIPubMedGoogle Scholar
- Spellberg B, Guidos R, Gilbert D, Bradley J, Boucher HW, Scheld WM, The epidemic of antibiotic-resistant infections: a call to action for the medical community from the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:155–64. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 19, Number 1—January 2013
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Jose Bordon, Providence Hospital, Department of Medicine, Section of Infectious Diseases, 1160 Varnum St, NE, Washington, DC 20017, USA
Top