Volume 19, Number 12—December 2013
Letter
Concurrent Parasitic Infections in a Renal Transplant Patient
To the Editor: Protozoan pathogens, including Entamoeba histolytica, Giardia, Cryptosporidium, Cyclospora, Cystoisospora, and microsporidia such as Enterocytozoon bieneusi, are well-known agents of diarrhea and a major public health problem in developing countries. Infection with Cyclospora cayetanensis and E. bieneusi can occur in immunocompromised and immunocompetent persons. Severe diarrhea and weight loss along with anorexia, nausea, and low-grade fever occur in immunocompromised persons, particularly those with HIV/AIDS and transplant recipients who are taking immunosuppressive drugs (1,2). However, transient diarrhea occurs in immunocompetent persons, notably in travelers returning from countries with poor hygienic standards (1–3).
We report on a kidney transplant recipient who had uncontrollable diarrhea and weight loss in whom C. cayetanensis and E. bieneusi were detected in biopsy specimens; the diarrhea resolved after treatment with drugs that act specifically on these 2 parasites. The patient was a 55-year-old man from the Dominican Republic living in New York, NY, USA; he had a history of long-term diabetes, coronary disease, and alcoholism. He had undergone a cadaveric renal transplant 14 months earlier and had an uneventful posttransplant course. After returning from visiting family in the Dominican Republic, he sought treatment for acute, profuse watery diarrhea in early November, 2009. He had >10 watery bowel movements daily that were associated with a 20-lb weight loss. His symptoms persisted for 2 months, and he required 2 hospitalizations for the diarrhea.
Results of 4 repeat fecal specimen tests (routine diagnostic microscopy and culture) were negative for parasites. Colonoscopy findings were normal; because of evidence of leukocytes in the feces and elevated fecal fat level, however, he received empirically prescribed metronidazole. Because his diarrhea and weight loss persisted, an upper endoscopy was performed, which revealed the presence of microsporidia. He then received albendazole for 3 weeks without substantial benefit.
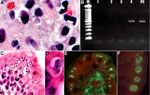
The biopsy specimens were sent to the Centers for Disease Control and Prevention (Atlanta, GA, USA) for further analysis. Biopsy slides were stained with hematoxylin and eosin and with Gram chromotrope (4) and examined by microscopy. The Gram chromotrope–stained slide revealed oval spores, pinkish-red in color, measuring ≈1 µm (5). These spores were supra nuclear in position and were consistent with E. bieneusi (Figure, panel A). The tissue sections were scraped from the slides, DNA was extracted, and conventional PCR was performed by using E. bieneusi–specific primers as described (5); the sizes of the amplified product in the tissue DNA specimen and in the E. bieneusi control specimen were identical (Figure, panel B), confirming the presence of E. bieneusi. On further microscopic examination of the Gram chromotrope and the hematoxylin and eosin–stained slides, oval bodies (8–10 µm) were seen. A few of these oval bodies exhibited 4 spindle-shaped structures which were identified provisionally as merozoites of a coccidian parasite (Figure, panel C). Others had morula-like internal structure (Figure, panel D). We hypothesized that the coccidian parasite could either be C. cayetanensis or Cystoisospora hominis. Because the parasites, in various stages, were just beneath the surface of the epithelium, rather than deep within the epithelium, we believed this organism to be a Cyclospora sp. rather than a Cystoisospora sp. We searched the serum bank of the Division of Parasitic Diseases, Centers for Disease Control and Prevention, and identified a serum sample from a person with a case of C. cayetanensis cyclosporiasis. An indirect immunofluorescence test was performed by using this serum on a deparaffinized section of the tissue biopsy specimen. Different stages of the coccidian organism were labeled brightly and produced apple-green fluorescence against a red counterstain (Eriochrome Black T), indicating that the parasite could possibly be a Cyclospora sp. (Figure, panels E, F). We considered that the Cyclospora-positive serum sample obtained from this particular patient may not be species-specific, since he might have also been infected with Cystoisospora. Therefore, we performed a real-time PCR assay that can distinguish C. cayetanensis from other coccidian parasites to identify the parasite definitively (3). DNA recovered from tissue in paraffin sections was successfully amplified and detected with this assay (data not shown), confirming the presence of C. cayetanensis.
The patient’s illness was treated with albendazole for E. bieneusi infection and with trimethoprim and sulfamethoxazole for C. cayetanensis infection. The patient’s diarrhea subsided after 1 week, and several subsequent fecal samples were negative for microsporidia spores and Cyclospora oocysts. His immunosuppressive medications were reduced, and he remained diarrhea-free for the following 3-year period of April 2010 to April 2013.
References
- Ortega YR, Sanchez R. Update on Cyclospora cayetanensis, a food-borne and waterborne parasite. Clin Microbiol Rev. 2010;23:218–34 . DOIPubMedGoogle Scholar
- Didier ES. Microsporidiosis: an emerging and opportunistic infection in humans and animals. Acta Trop. 2005;94:61–76 . DOIPubMedGoogle Scholar
- Verweij JJ, Laeijendecker D, Brienen EA, van Leishout L, Polderman AM. Detection of Cyclospora cayetanensis in travelers returning from the tropics and subtropics using microscopy and real-time PCR. Int J Med Microbiol. 2003;293:199–202 . DOIPubMedGoogle Scholar
- Moura H, Schwartz DA, Bornay-Llinares F, Sodre FC, Wallace S, Visvesvara GS. A new and improved “quick-hot Gram-chromotrope” technique that differentially stains microsporidian spores in clinical samples, including paraffin-embedded tissue sections. Arch Pathol Lab Med. 1997;121:888–93 .PubMedGoogle Scholar
- da Silva AJ, Schwartz DA, Visvesvara GS, de Moura H, Slemenda SB, Pieniazek NJ. Sensitive PCR diagnosis of infections by Enterocytozoon bieneusi (microsporidia) using primers based on the region coding for small subunit rRNA. J Clin Microbiol. 1996;34:986–7 .PubMedGoogle Scholar
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 19, Number 12—December 2013
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Michael Arrowood, Centers for Disease Control and Prevention, 4770 Buford Highway NE, Mailstop F36, Atlanta, GA 30341-3724, USAMichael Arrowood, Centers for Disease Control and Prevention, 4770 Buford Highway NE, Mailstop F36, Atlanta, GA 30341-3724, USA
Top