Volume 5, Number 3—June 1999
Synopsis
Adhesins as Targets for Vaccine Development
Figure
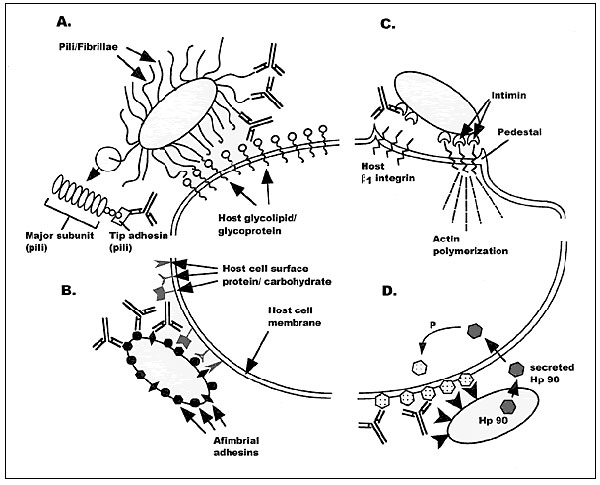
Figure. Four mechanisms of bacterial adherence where anti-adhesin vaccines could potentially block colonization and infection. A shows pili or fibrillae protruding from the bacterial surface. These proteinaceous appendages bind to host cell surface molecules, usually carbohydrates, by adhesin proteins located at the distal tip of the pilus/fibrillar organelle. Antibodies targeting the adhesin protein block the bacterial/host interaction. B demonstrates a similar process of bacterial/epithelial cell interactions mediated by afimbrial adhesin proteins. In this case, antibodies directed against the bacterial surface proteins should also block attachment and colonization by impeding the ability of the bacteria to associate with mucosal tissues. C illustrates that some bacteria establish intimate associations with eukaryotic cells by intimin proteins, resulting in cytoskeletal rearrangements, host cell signaling, possible internalization of the bacteria, and in many cases systemic disease. Blocking the intimate association/adherence may also be another strategy to prevent bacterial infections. D shows a novel mechanism whereby bacteria secrete their own receptor protein, which is internalized by the target host cell, phosphorylated, and embedded in the eukaryotic cell as a new receptor for tight binding by the bacterium. Theoretically, blocking the secreted receptor (Hp90) before it is internalized by the host cell could provide another mechanism to block bacterial adherence and infection.