Volume 6, Number 2—April 2000
Research
Serologic Response to Culture Filtrate Antigens of Mycobacterium ulcerans during Buruli Ulcer Disease
Abstract
Buruli ulcer (BU) is an emerging necrotic skin disease caused by Mycobacterium ulcerans. To assess the potential for a serodiagnostic test, we measured the humoral immune response of BU patients to M. ulcerans antigens and compared this response with delayed-type hypersensitivity responses to both Burulin and PPD. The delayed-type hypersensitivity response generally supported the diagnosis of BU, with overall reactivity to Burulin in 28 (71.8%) of 39 patients tested, compared with 3 (14%) of 21 healthy controls. However, this positive skin test response was observed primarily in patients with healed or active disease, and rarely in patients with early disease (p=0.009). When tested for a serologic response to M. ulcerans culture filtrate, 43 (70.5%) of 61 BU patients had antibodies to these antigens, compared with 10 (37.0%) of 27 controls and 4 (30.8%) of 13 tuberculosis patients. There was no correlation between disease stage and the onset of this serum antibody response. Our findings suggest that serologic testing may be useful in the diagnosis and surveillance of BU.
Buruli ulcer (BU) disease, caused by Mycobacterium ulcerans, is characterized by severe necrotizing ulcers. The disease, which is found worldwide but primarily in tropical climates (1,2), occurs predominantly in children ages 5 to 14 (3) and is associated with severe illness and permanent disabilities in >26% of patients (1,3). Over the past decade, the incidence of BU disease has dramatically increased in several West African countries, notably Ghana, Côte d'Ivoire, Benin, and Togo (1,2). In some West African communities, BU has replaced tuberculosis (TB) and leprosy as the most prevalent mycobacterial disease, affecting up to 22% of the population (4). However, the true global impact of the disease is unknown because reliable tools for its rapid diagnosis and surveillance are lacking. In addition, neither the mode of transmission nor the environmental reservoir for M. ulcerans is known, although observational studies suggest that M. ulcerans infection is transmitted through the skin after contact with contaminated water, vegetation (5), or insects (6).
Current treatment for cutaneous disease is primarily surgical. Ulcerative lesions require wide debridement and skin grafting in specialized health-care facilities, long hospital stays (7), and increased cost to the health-care system and the community (7). Antibiotics are ineffective once ulcers are present, possibly because M. ulcerans is absent from lesions or subcutaneous tissues have already been destroyed, preventing penetration of antimycobacterial drugs (3). Antimicrobial therapy has not been studied in preulcerative lesions because apparently patients rarely visit primary health-care providers with painless nodular lesions and diagnostic tests are not available to clearly identify M. ulcerans infection before it progresses to clinical disease. Early identification and surgical excision of these preulcerative nodular lesions can be curative and does not require inpatient care (8). We investigated the serologic response to the M. ulcerans culture filtrate (MUCF) of BU patients at different stages of disease to determine whether serodiagnosis is feasible as a tool for early diagnosis and intervention of BU disease.
BU patients were identified through a case-control study conducted in 1991 in the Daloa region of Côte d'Ivoire (3). Patients with early ulcerative BU were identified by small, painless, or minimally painful nodules or edema, or ulcerative lesions for <2 weeks. Patients with ulcerative BU were identified by lesions that were chronic (>2 weeks duration), were painless or minimally painful, or had undermined edges. Patients with healed BU were identified by stellate scars with retraction in areas of prior ulceration. Descriptive and clinical data were obtained from all BU patients and controls (3). Blood specimens were collected from 82 BU patients and 164 healthy controls in the Daloa region.
Because TB is endemic in many of the areas where BU occurs, 13 TB patients from the United States were used as controls for cross-reactivity to MUCF. Use of sera from these patients ensured the lowest probability for exposure to M. ulcerans, so that only cross-reactivity to antigens common to both M. tuberculosis and M. ulcerans would be measured. TB patients were identified through an ongoing study in Atlanta by clinical presentation, a positive PPD response, and isolation of M. tuberculosis from sputum. All specimens tested negative for HIV and were strongly reactive to many of the culture filtrate antigens from M. tuberculosis (data not shown). Informed consent was obtained for all participants, and protocols were approved by the Institutional Review Boards of the Centers for Disease Control and Prevention and the Ministry of Health of Côte d'Ivoire for the BU study and the Institutional Review Board at Emory University for the TB study.
Skin Testing
During sera collection, Burulin (9) and PPD (purified protein derivative of M. tuberculosis, Connaught Laboratories, U.K.) were administered intradermally into the flexor surface of the forearms of 39 BU patients and 21 controls, and the diameter of induration was recorded at 48 and 72 hours. The criterion for using Burulin and PPD dual testing to confirm clinical cases of BU has been reported (3,9), and the cumulative delayed-type hypersensitivity (DTH) findings for this study have been described (Table 1) (3). Sera were also tested for reactivity to MUCF antigen by Western blot (Table 2).
Preparation of MUCF Antigen for Serologic Analysis
M. ulcerans strain S-WT (from the Centers for Disease Control and Prevention culture collection; Atlanta) was recovered from a 1-ml lyophilisate in 10% glycerol by incubation in 5 ml Middlebrook 7H9 with OADC supplement (Remel, Lenexa, KS) at 32.5°C for 7 days. This 5-ml inoculum was passaged into 25 ml Middlebrook 7H9 broth supplemented with tryptose and glucose (7H9TG; 0.1% wt:vol and 2% wt:vol, respectively) and incubated at 32.5°C for 10 days, followed by passage into 100 ml of 7H9TG and growth at 32.5°C for 10 days. Cultivation in this protein- and serum-free media allowed isolation and analysis of specific secreted mycobacterial proteins. The 100-ml culture was passaged into 1L of 7H9TG, incubated at 32.5°C in 850-cm2 roller bottles, and rotated at 90 rotations per hour. Bacilli were harvested during mid-log phase growth by centrifugation. The 1L of MUCF, containing mycobacterial proteins of interest, was concentrated at 4°C under N2 by using a stirred-cell apparatus (Amicon Inc., Beverly, MA) with a 10-kDa molecular weight membrane. MUCF was exchanged into 0.1 M NH4HCO3 by dialysis, lyophilized, and purified by separation of residual small molecular weight contaminants from antigens of interest by size exclusion chromatography. MUCF was loaded onto a fast-performance liquid chromatography system (FPLC) (Pharmacia, Piscataway, NJ) equipped with a G-25 superfine Sephadex desalting column (Pharmacia, Piscataway, NJ), UV monitor, and conductivity monitor for separation and detection of proteins and salts, respectively. MUCF was eluted with an isocratic gradient of 0.1 M NH4HCO3. MUCF protein was quantitated by bicinchoninic acid assay (Pierce Chemical Co., Rockford, IL) and lyophilized until needed. Approximately 5 mg of MUCF protein was obtained per liter of culture.
Aliquots of 8 g of MUCF protein were resolved by discontinuous sodium dodecylsulfate polyacrylamide gel electrophoresis (SDS-PAGE) (10) by using 10% to 20% gradient 10-well gels (Novex, San Diego, CA). MUCF proteins were visualized by staining with silver (11), and this profile was reexamined throughout these experiments to ensure that the MUCF proteins did not degrade upon storage; no antigen variability was seen during our studies. 7H9TG media alone processed in the same way contained no proteins by silver stain analysis after SDS-PAGE and bicinchoninic acid assay.
Amino Acid Sequence Analyses
The N-terminal amino acid sequences of MUCF proteins were determined and compared with known mycobacterial sequences. One hundred micrograms of MUCF protein was resolved by two-dimensional gel electrophoresis (2D-GE) by the method of O'Farrell (12) with the modifications of Sonnenberg and Belisle for resolution of mycobacterial proteins (13). 2D-GE resolved proteins were transferred to 0.1 mm polyvinyl difluoride (PVDF) membrane (14) and visualized by staining with 0.5% Coomassie blue R250 in 40% methanol/10% acetic acid for 2 minutes. The N-terminal amino acid sequence was obtained by loading membranes containing the excised spot of interest onto a Procise-cLC automated Edman sequencer (Applied Biosystems, Foster City, CA) and reading the generated chromatographs against amino acid standards.
Statistical Analyses
Statistical analyses were done by the Mantel-Haenszel chi-square method. Statistical significance for the chi-square p values and Fisher's exact p values was evaluated at α = 0.05. Fisher's exact p value was used when the expected cell count was <5. Odds ratios (OR) were determined by calculating the ratio of the odds of exposure among cases to that among controls. The test for specificity was determined by dividing the true negatives (n = 24; disease-negative and 70 kDa-negative) by the total number of controls (n = 27). All analyses were done on BU patients and healthy controls from the Daloa region of Côte d'Ivoire, where the disease is endemic, unless otherwise specified. All data were analyzed by using Exact 2.0b software (D. Martin and H. Austin, Atlanta, GA).
Seventeen (43.6%) of the 39 skin-tested BU patients had induration of > 10 mm within 72 hours of Burulin administration, with PPD induration at least 3 mm smaller than that with Burulin (Table 1). Only 3 (14.3%) of the 21 skin-tested healthy controls from the area had a similar DTH pattern (Table 1). In addition, 11 (28.2%) of the 39 BU patients had an equally strong response to Burulin and PPD, which suggests that Burulin skin testing may not be reliable in regions where BU and TB are endemic and BCG vaccination is widely used (Table 1). Dual-positive responses included, 28 (71.8%) of the 39 BU patients were Burulin positive. However, when this total Burulin-positive population was delineated by disease stage, positive responses were observed in most of the patients with healed (93.8%) and ulcerative disease (64.7%), but in few (33.4%) patients with early ulcerative disease (Table 2). When healed and active Burulin responses were compared with the DTH response seen in early ulcerative BU patients, a significant difference between the disease stages was found (Fisher's exact p = 0.009).
These skin-tested BU patient and control populations from the Daloa region were then tested for reactivity of their sera to MUCF by Western blot, and Burulin induration was compared with serum antibody response (Table 2). Twenty-six (66.7%) of the 39 skin-tested BU patients had an antibody response to the MUCF, versus 7 (33.3%) of the 21 skin-tested controls (chi-square p = 0.014). BU patient populations had approximately the same positive antibody responses to the MUCF, regardless of disease stage. Specifically, 68.8% of the healed patients were antibody positive, with active ulcerative disease at 64.7% and early disease at 66.7% (Table 2). When the antibody responses of healed and active patients were compared with those seen in early BU patients, the serologic response did not differ significantly by disease stage (Fisher's exact p = 0.999). Therefore, serologic testing may be useful in the early diagnosis and surveillance of BU.
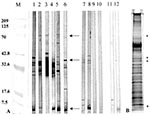
The antibody response to the MUCF was then determined for all 61 clinically diagnosed BU patients and 27 healthy controls, and antibody reactivity to individual antigens was recorded. Three M. ulcerans antigens of 70, 38/36, and 5 kDa were commonly recognized by BU patient sera antibodies (Figure). The 36-kDa antigen was found to be a degradation product of the 38-kDa antigen (unpub. results). Forty-three (70.5%) of 61 BU patient sera had antibodies to at least 1 of 3 antigens, compared with 10 (37.0%) of 27 sera from healthy controls from the area and 4 (30.8%) of 13 sera from TB patients (Table 3). The antibody response to the 70-kDa M. ulcerans antigen was associated with BU disease (OR = 12.33; chi-square p = 0.00002), and the response to this protein was consistent throughout early to late disease stage (OR = 12.4; chi-square p = 0.0000001; Table 3). Results were similar for the 38/36-kDa antigen (OR = 5.94; chi-square p = 0.0016), regardless of disease stage (OR = 3.00; chi-square p = 0.0026). The N-terminal amino acid sequence of the 38 kDa protein was DPAPAPAPRD, and had mycobacterial sequences that precede glycosylation sites (15). The serologic response to the 5-kDa antigen, although common in the seroreactive specimens tested, did not differ significantly between BU patients and healthy controls from disease-endemic areas or TB patients (chi-square p = 0.764 and Fisher's exact p = 0.999, respectively); nor were antibodies to this antigen observed in BU patients at the early ulcerative stage (Table 3). A cross-reactive serologic response to MUCF was observed in some TB patients who had not been exposed to M. ulcerans and in some healthy controls from disease-endemic areas (Figure; representative sample and Table 3). These positive responses may be due to exposure to other mycobacterial pathogens with similar antigens as M. ulcerans or BU disease undetectable by clinical diagnosis, respectively.
Diagnosis of BU currently relies on the clinical presentation of the ulcerative disease stage, by which time the infection has already caused damage, therapeutic options are limited, and outcome is associated with severe disease (3,7,8). Preulcerative nodules and edemous lesions, when identified, frequently contain numerous extracellular mycobacteria (1,2). Thus, diagnosis of BU disease at this early stage may lead to more effective treatment. Indeed, diagnosis of preulcerative BU disease is associated with a better therapeutic outcome and reduced illness (8), and accurate surveillance at the preulcerative stage would enable assessment of the prevalence and global impact of BU disease. Surveillance may also identify risk factors as well as prevention strategies for BU.
Consistent with previous reports (3,9), DTH to Burulin, a crude preparation of M. ulcerans lysate (9), was observed in many (71.8%) of the clinically diagnosed BU patients. This overall response was similar to the well-defined cell-mediated DTH responses reported for other mycobacterial infections (16-18), including M. tuberculosis (19,20). In contrast, in BU patients, the Burulin response was associated with ulcerative and healed disease, but not with early ulcerative disease. Thus, unlike other DTH-based monitoring programs that indicate mycobacterial infection before and during disease in immunocompetent persons, Burulin testing may be more useful for confirming ulcerative disease or monitoring past disease with M. ulcerans in conjunction with PPD results.
The marked shared reactivity to PPD and Burulin antigens by BU patients, as measured by similar induration upon intradermal injection, was expected to some degree, considering the commonality of antigenic lipids on the mycobacterial surface, genetic relatedness of mycobacterial pathogens, opportunity of exposure to both mycobacterial pathogens and environmental mycobacteria, and rate of BCG vaccination in the region. When Burulin-positive results were analyzed in conjunction with either the presence of a BCG vaccination scar or recollection of BCG vaccination, 12 of 14 BCG-vaccinated BU patients were Burulin positive, and 6 of these 12 patients were also PPD positive. Additionally, all the Burulin- and PPD-positive controls either recalled BCG vaccination or had a BCG scar (data not shown).
We found that a specific response to the MUCF was observed in BU patient sera, but unlike the Burulin DTH response, the antibody response did not correlate with disease stage (Table 2). The antibody reactivity in the sera from some of the controls from disease-endemic areas may reflect preclinical infection with M. ulcerans or exposure to other mycobacteria and cross-reactivity to antigens common to mycobacterial species. Interestingly, 10 (55.6%) of the 18 antibody-negative BU patients, versus 15 (33%) of the 43 antibody-positive BU patients, recalled or had a BCG vaccination scar, which suggests that BCG vaccination did not contribute to the antibody response of BU patients to MUCF (data not shown).
Thirty percent of the TB patient sera were reactive to MUCF, with reactivity principally towards lower molecular weight proteins and not the 70- or 38/36-kDa proteins (Figure; Table 3). N-terminal amino acid sequence of one of these lower molecular weight proteins, a 31-kDa protein, was found to be FSRPGLPVEY and demonstrated homology with the mycolyl transferases found in M. tuberculosis and other mycobacteria (21). Thus, reactivity of the TB patient sera based on antibodies generated against common mycobacterial antigens is the likely explanation for the few TB patient sera reactive to MUCF.
Our preliminary results indicate the usefulness of the MUCF in detecting BU; specifically, individual antigens in the MUCF may be used for the development of a sensitive and specific test for BU (Table 3). Both the 70- and 38/36-kDa proteins were indicators of BU disease, and antibody responses to both of these proteins were similarly present in BU-reactive patient serum samples, regardless of disease stage. In addition, reactivity to the 70-kDa protein was specific for M. ulcerans infection (88.8%).
The 70- and 38/36-kDa proteins may be useful for the development of a serologic test for BU in areas where TB is endemic. Based on these results, we are isolating and characterizing these two M. ulcerans antigens for additional testing. The N-terminal sequence for the 38/36-kDa protein has already been determined as part of the purification process for this protein, and identification and purification of the 70-kDa protein are under way. If a serologic test proves to be diagnostic, early excision of nodules and antimycobacterial chemotherapy trials of patients could reduce the public health and socioeconomic impact of this emerging disease.
Dr. Dobos is a senior postdoctoral fellow in the laboratory of Dr. C. Harold King at the Emory University School of Medicine. Her current research interests focus on bacterial proteomic analysis. She will continue studying the proteins of Mycobacterium ulcerans for development of future vaccine and diagnostic candidates.
Acknowledgments
We thank John Stanford for providing the Burulin reagent, Jennifer Ekmark for discussion and critical reading of the manuscript, Merywin Wigley for laboratory assistance, Carolyn Carter for N-terminal sequence analysis, the Emory Medical Care Foundation for collection and use of the TB patient and control sera (HIC # 455-96), and the National Center for Infectious Diseases for the initial case study of BU disease in Côte d'Ivoire.
This study was supported in part by Centers for Disease Control and Prevention Cooperative agreement No. U50/CCU416560-01-1, Novel Diagnostic Tests for Infections of Public Health Significance, and by an Oak Ridge Institute for Science and Education Fellowship awarded to Karen M. Dobos.
References
- Horsburgh CR Jr, Meyers WM. Buruli ulcer. In: Pathology of emerging infections. Horsburgh CR Jr, Nelson AM, editors. Washington: American Society for Mibrobiology Press; 1997. p. 119-26.
- Dobos KM, Horsburgh CR Jr, Ashford DA, Quinn FD, King CH. Emergence of a unique group of necrotizing mycobacterial diseases. Emerg Infect Dis. 1999;5:367–78. DOIPubMedGoogle Scholar
- Marston BJ, Diallo MO, Horsburgh CR Jr, Diomande I, Saki MZ, Kanga J-M, Emergence of Buruli ulcer disease in the Daloa region of Côte d'Ivoire. Am J Trop Med Hyg. 1995;52:219–24.PubMedGoogle Scholar
- Amofah GK, Sagoe-Moses C, Adjei-Acquah C, Frimpong EH. Epidemiology of Buruli ulcer in Amansie West district, Ghana. Trans R Soc Trop Med Hyg. 1993;87:644–5. DOIPubMedGoogle Scholar
- Hayman J. Postulated epidemiology of Mycobacterium ulcerans infection. Int J Epidemiol. 1991;20:1093–8. DOIPubMedGoogle Scholar
- Portaels F, Elsen P, Guimaraes-Peres A, Fonteyne PA, Meyers WM. Insects in the transmission of Mycobacterium ulcerans infection. Lancet. 1999;353:986. DOIPubMedGoogle Scholar
- Asiedu K, Etuaful S. Socioeconomic implications of Buruli ulcer in Ghana: a three-year review. Am J Trop Med Hyg. 1998;59:1015–22.PubMedGoogle Scholar
- Amofah G, Asamoah S, Afram-Gyening G. Effectiveness of excision of pre-ulcerative Buruli lesions in field situations in a rual district in Ghana. Trop Doct. 1998;28:81–3.PubMedGoogle Scholar
- Stanford TL, Revill WDL, Gunthorpe WJ, Grange JM. The production and preliminary investigation of Burulin, a new skin test for Mycobacterium ulcerans infection. J Hyg (Lond). 1975;74:7–16. DOIPubMedGoogle Scholar
- Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227:680–5. DOIPubMedGoogle Scholar
- Morrissey JH. Silver stain for proteins in polyacrylamide gels: a modified procedure with enhanced, uniform sensitivity. Anal Biochem. 1981;117:307–10. DOIPubMedGoogle Scholar
- O'Farrell PH. High resolution two dimensional analysis of proteins. J Biol Chem. 1975;250:4007–21.PubMedGoogle Scholar
- Sonnenberg MG, Belisle JT. Definition of Mycobacterium tuberculosis culture filtrate proteins by two dimensional polyacrylamide gel electrophoresis, N-terminal amino acid sequencing, and electrospray mass spectrometry. Infect Immun. 1997;65:4515–24.PubMedGoogle Scholar
- Towbin H, Staehelin T, Gordon J. Electrophoretic transfer of protein from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979;76:4350–4. DOIPubMedGoogle Scholar
- Dobos KM, Khoo KH, Swiderek KM, Brennan PJ, Belisle JT. Definition of the full extent of glycosylation of the 45-kilodalton glycoprotein of Mycobacterium tuberculosis. J Bacteriol. 1996;178:2498–506.PubMedGoogle Scholar
- De Vries RR. An immunogenetic view of delayed type hypersensitivity. Tubercle. 1991;72:161–7. DOIPubMedGoogle Scholar
- Sengupta U. Studies on lepromin and soluble antigens of M. leprae: their classification standardization and use. Indian J Lepr. 1991;63:457–65.PubMedGoogle Scholar
- Shield MJ, Stanford JL, Paul RC, Carswell JW. Multiple skin testing of tuberculosis patients with a range of new tuberculins, and a comparison with leprosy and Mycobacterium ulcerans infection. J Hyg (Lond). 1977;78:331–48. DOIPubMedGoogle Scholar
- Dannenberg AM Jr. Delayed-type hypersensitivity and cell-mediated immunity in the pathogenesis of tuberculosis. Immunol Today. 1991;7:228–33. DOIGoogle Scholar
- Orme IM, Andersen P, Boom WH. T cell response to Mycobacterium tuberculosis. J Infect Dis. 1993;167:1481–97.PubMedGoogle Scholar
- Belisle JT, Vissa VD, Sievert T, Takayama K, Brennan PJ, Besra GS. Role of the major antigen of Mycobacterium tuberculosis in cell wall biogenesis. Science. 1997;276:1420–2. DOIPubMedGoogle Scholar
Figure
Tables
Cite This ArticleTable of Contents – Volume 6, Number 2—April 2000
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
C. Harold King, Emory University School of Medicine, 69 Butler St. S.E., Atlanta, GA 30303, USA; fax: 404-880-9305
Top