Hepatitis B
CDC Yellow Book 2024
Travel-Associated Infections & DiseasesINFECTIOUS AGENT: Hepatitis B virus
ENDEMICITY
Worldwide
High prevalence in Africa and the Western Pacific
TRAVELER CATEGORIES AT GREATEST RISK FOR EXPOSURE & INFECTION
PREVENTION METHODS
Ensure sterile medical and dental techniques
Use safe injection practices
Hepatitis B is a vaccine-preventable disease
DIAGNOSTIC SUPPORT
Infectious Agent
Hepatitis B virus (HBV) is a small, circular, partially double-stranded DNA virus in the family Hepadnaviridae.
Transmission
HBV is transmitted by contact with contaminated blood, blood products, and other body fluids (e.g., semen). Travelers could be exposed to HBV through poor infection control during dental or medical procedures, receipt of blood products, injection drug use, tattooing or acupuncture, or unprotected sex.
Epidemiology
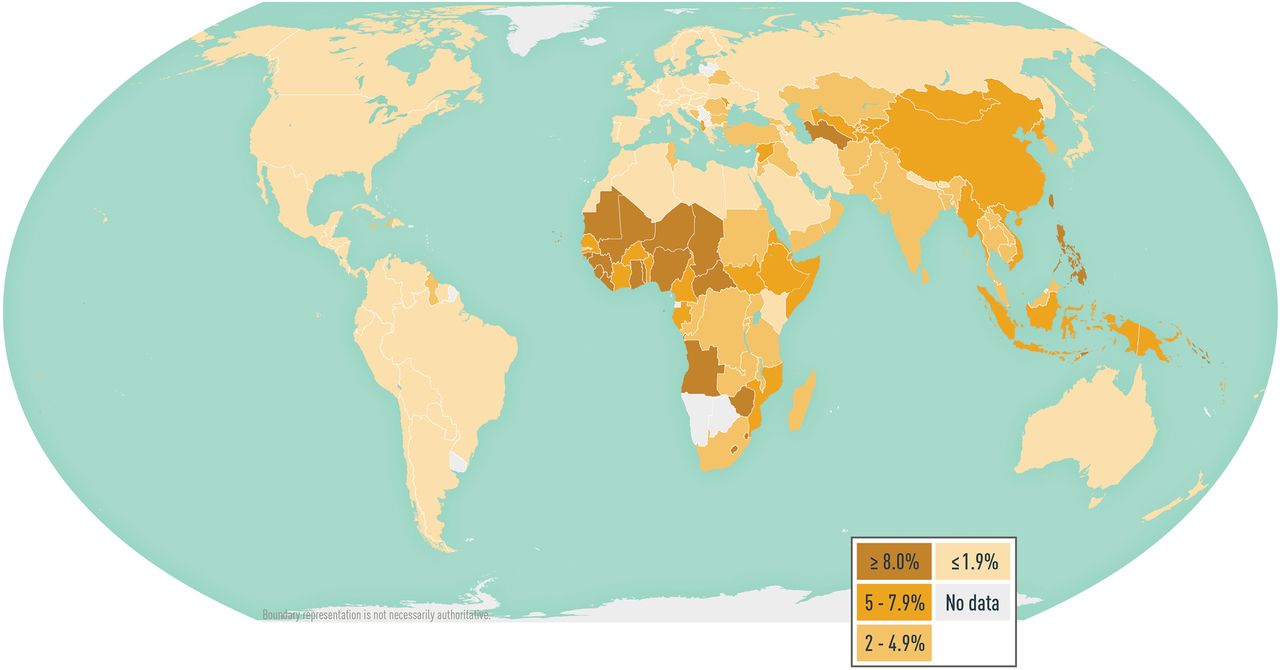
HBV is a leading cause of chronic hepatitis, liver cirrhosis, and hepatocellular carcinoma worldwide. In 2015, an estimated 257 million people globally were living with chronic HBV infection; that year, HBV caused an estimated 887,000 deaths. HBV infections are likely underestimated, however, because accurate data are lacking from many countries (Map 5-07).
Data demonstrating the specific risk to travelers are lacking; published reports of travelers acquiring hepatitis B are rare, however, and the risk for travelers who do not have high-risk behaviors or exposures is low. The risk for HBV infection might be higher in countries where the prevalence of chronic HBV infection is ≥2% (e.g., in the western Pacific and African regions); expatriates, missionaries, and long-term development workers in those regions might be at increased risk for HBV infection. All travelers should be aware of how HBV is transmitted and take measures to minimize their exposures.
Map 5-07 Worldwide prevalence of hepatitis B virus infection
Disease data source: 2021 estimates of hepatitis B virus disease burden. CDA Foundation Polaris Observatory. Available from: https://cdafound.org/polaris-countries-distribution/.
Clinical Presentation
HBV infection primarily affects the liver. Typically, the incubation period for hepatitis B is 90 days (range 60–150 days). Newly acquired acute HBV infections only cause symptoms some of the time, and signs and symptoms vary by age. Most children <5 years of age and immunosuppressed adults are asymptomatic when newly infected, whereas 30%–50% of newly infected people aged ≥5 years have signs and symptoms. When present, typical signs and symptoms of acute infection include abdominal pain, anorexia, fatigue, fever, jaundice, joint pain, malaise, nausea and vomiting, light (clay-colored) stool, and dark urine. The overall case-fatality ratio of acute hepatitis B is ≈1%.
Some acute HBV infections resolve on their own, but some develop into chronic infection. The risk for acute hepatitis B to progress to chronic HBV infection depends on the age at the time of initial infection as follows: >90% of neonates and infants, 25%–50% of children aged 1–5 years, and <5% of older children and adults. Most people with chronic HBV infection are asymptomatic and have no evidence of liver disease. Fifteen percent to 40% of people with chronic HBV infection will, however, develop liver cirrhosis, hepatocellular carcinoma, or liver failure, and 25% of chronically infected people die prematurely from these complications. People infected with HBV are susceptible to infection with hepatitis D virus; coinfection increases the risk for fulminant hepatitis and rapidly progressive liver disease.
Diagnosis
Hepatitis B is a nationally notifiable disease. The clinical diagnosis of acute HBV infection is based on signs or symptoms consistent with viral hepatitis and elevated hepatic transaminases and cannot be distinguished from other causes of acute hepatitis. Serologic markers specific for hepatitis B are necessary to diagnose HBV infection and for appropriate clinical management (Table 5-11). These markers can differentiate between acute, resolving, and chronic infection.
See information on how to obtain hepatitis B diagnostic support from the Centers for Disease Control and Prevention (CDC) Infectious Diseases Laboratories, including contact information, which samples to send, and how to send samples. Select Hepatitis B Genotyping for research use only, and Hepatitis B Serology and Quantitative PCR if testing regulated by Clinical Laboratory Improvement Amendments is needed.
Table 5-11 Interpretation of serologic test results for hepatitis B virus infection1
| CLINICAL STATE |
HBsAg |
TOTAL ANTI-HBs |
TOTAL ANTI-HBc |
ACTION |
|---|---|---|---|---|
|
Chronic infection |
Positive |
Negative |
Positive |
Link to hepatitis B-directed care2 |
|
Acute infection |
Positive |
Negative |
Positive (HBc IgM) |
Link to hepatitis B-directed care2 |
|
Resolved infection |
Negative |
Positive |
Positive |
Counseling, reassurance |
|
Immune (immunization) |
Negative |
Positive3 |
Negative |
Reassurance |
|
Susceptible (never infected and no evidence of immunization) |
Negative |
Negative |
Negative |
Vaccinate |
|
Isolated core antibody4 |
Negative |
Negative |
Positive |
Depends on situation |
Abbreviations: HBsAg, hepatitis B surface antigen; anti-HBc, antibody to hepatitis B core antigen; anti-HBs, antibody to hepatitis B surface antigen.
1Adapted from Abara WE, Qaseem A, Schillie S, McMahon BJ, Harris AM. Hepatitis B vaccination, screening, and linkage to care: best practice advice from the American College of Physicians and the Centers for Disease Control and Prevention. Ann Intern Med. 2017;167(11):794–804.
2Hepatitis B-directed care includes a physical examination and laboratory evaluation for liver transaminase, hepatitis B virus DNA, and hepatitis B e antigen.
3An anti-HBs titer of ≥10 mIU/mL correlates with protection only after a documented, complete hepatitis B vaccine series.
4If false-positive results are suspected, repeat testing. If results are from past infection or passive transfer to infants born to HBsAg-positive mother, no specific action is needed. If results could indicate occult hepatitis B virus infection, inform patient of risks from future chemotherapy, immunosuppression, or hepatitis C virus infection antiviral therapy, and consider checking hepatitis B virus DNA.
Treatment
No medications are available to treat acute HBV infection; treatment is supportive. Several antiviral medications are available for people with chronic HBV infection. People with chronic HBV infection should be under the care of a health professional and receive a thorough physical examination and laboratory testing to determine the need for antiviral therapy and ongoing monitoring for hepatocellular carcinoma and liver damage. See American Association for the Study of Liver Diseases (AASLD) practice guidelines for the treatment of chronic HBV infection.
Prevention
Vaccines
Indications for Use
Administer Hepatitis B vaccine to all unvaccinated people traveling to areas with intermediate to high prevalence of chronic HBV infection, namely, countries with HBV surface antigen positivity prevalence ≥2% (Map 5-07). See complete vaccination information and recommendations for the United States.
Administration
Several hepatitis B vaccines are available (Table 5-12). Hepatitis B vaccines are administered either as a 2-dose series at 0 and 1 month (Heplisav-B [Dynavax Technologies Corporation]), or as a 3-dose series at 0, 1, and 6 months (Engerix-B [GlaxoSmithKline], Recombivax HB [Merck], PreHevbrio [VBI], and the combined hepatitis A and hepatitis B vaccine, Twinrix [GlaxoSmithKline]). Heplisav-B is licensed for a 2-dose schedule for adults aged ≥18 years; the second dose should be given ≥1 month after the first dose.
Engerix B and Recombivax HB have also been licensed for use according to alternative vaccination schedules. Engerix-B can be administered using a 4-dose schedule, with the first 3 doses given within 2 months and a booster at 12 months (doses at 0, 1, 2, and 12 months). Recombivax HB can be given using a 2-dose schedule for children aged 11–15 years. Twinrix can be used on an accelerated 4-dose schedule (0, 7, and 21–30 days, with a booster at 12 months) to promote long-term immunity.
Always consult the prescribing information when administering alternative schedules and formulations. Whenever feasible, use the same manufacturer’s vaccines to complete the patient’s vaccine series; do not, however, defer vaccination when the manufacturer of previously administered doses is unknown or when the vaccine from the same manufacturer is unavailable. The 2-dose Heplisav-B vaccine series only applies when both doses in the series consist of Heplisav-B. Series consisting of a combination of 1 dose of Heplisav-B and a vaccine from a different manufacturer should adhere to the 3-dose schedule.
Protection from the standard vaccination series is robust, and >95% of healthy people achieve immunity after completion of the vaccination series. Serologic testing and booster vaccination are not recommended before travel for immunocompetent adults who have been previously vaccinated. Consider postvaccination serologic testing, however, for people whose subsequent clinical management depends on knowledge of their immune status, including health care personnel and public safety workers at risk for blood or body fluid exposure; those who require (or might require) outpatient hemodialysis; HIV-infected people; sex partners of HBsAg-positive people; and other immunocompromised people (e.g., hematopoietic stem-cell transplant recipients or people receiving chemotherapy).
Table 5-12 Hepatitis B vaccines
|
VACCINE |
TRADE NAME (MANUFACTURER) |
AGE IN YEARS |
DOSE & ROUTE |
SCHEDULE |
BOOSTER |
|---|---|---|---|---|---|
|
Hepatitis B vaccine, recombinant with novel adjuvant (1018) |
Heplisav-B (Dynavax Technologies) |
≥18 |
0.5 mL (20 µg HBsAg and 3,000 µg of 1018) IM |
DOSE 1: ≥18 years old DOSE 2: 1 month after DOSE 1 |
None |
|
Hepatitis B vaccine, recombinant1 |
Engerix-B (GlaxoSmithKline) |
0–19 |
0.5 mL (10 µg HBsAg) IM |
STANDARD DOSE 1: 0–19 years old DOSE 2: 1 month after DOSE 1 DOSE 3: 6 months after DOSE 1 |
None |
|
Hepatitis B vaccine, recombinant1 |
Engerix-B (GlaxoSmithKline) |
0–10 |
0.5 mL (10 µg HBsAg) IM |
ACCELERATED DOSE 1: 0–10 years old DOSE 2: 1 month after DOSE 1 DOSE 3: 2 months after DOSE 1 |
12 months after DOSE 1 |
|
Hepatitis B vaccine, recombinant1 |
Engerix-B (GlaxoSmithKline) |
11–19 |
1 mL (20 µg HBsAg) IM |
ACCELERATED DOSE 1: 11–19 years old DOSE 2: 1 month after DOSE 1 DOSE 3: 2 months after DOSE 1 |
12 months after DOSE 1 |
|
Hepatitis B vaccine, recombinant1 |
Engerix-B (GlaxoSmithKline) |
≥20 |
1 mL (20 µg HBsAg) IM |
STANDARD DOSE 1: ≥20 years old DOSE 2: 1 month after DOSE 1 DOSE 3: 6 months after DOSE 1 |
None |
|
Hepatitis B vaccine, recombinant1 |
Engerix-B (GlaxoSmithKline) |
≥20 |
1 mL (20 µg HBsAg) IM |
ACCELERATED DOSE 1: ≥20 years old DOSE 2: 1 month after DOSE 1 DOSE 3: 2 months after DOSE 1 |
None |
|
Hepatitis B vaccine, recombinant1 |
Recombivax HB (Merck) |
0–19 |
0.5 mL (5 µg HBsAg) IM |
STANDARD DOSE 1: 0–19 years old DOSE 2: 1 month after DOSE 1 DOSE 3: 6 months after DOSE 1 |
None |
|
Hepatitis B vaccine, recombinant1 |
Recombivax HB (Merck) |
11–15 |
1 mL (10 µg HBsAg) IM |
ADOLESCENT, ACCELERATED DOSE 1: 11–15 years old DOSE 2: 4–6 months after DOSE 1 |
None |
|
Hepatitis B vaccine, recombinant1 |
Recombivax HB (Merck) |
≥20 |
1 mL (10 µg HBsAg) IM |
STANDARD DOSE 1: ≥20 years old DOSE 2: 1 month after DOSE 1 DOSE 3: 6 months after DOSE 1 |
None |
|
Hepatitis B vaccine, recombinant1 |
PreHevbrio (VBI) |
≥18 |
1 mL (10 µg HBsAg) IM |
DOSE 1: ≥18 years old DOSE 2: 1 month after DOSE 1 DOSE 3: 6 months after DOSE 1 |
None |
|
Combined hepatitis A and B vaccine |
Twinrix (GlaxoSmithKline) |
≥18 |
1 mL (720 ELU HAV + 20 µg HBsAg) IM |
STANDARD DOSE 1: ≥18 years old DOSE 2: 1 month after DOSE 1 DOSE 3: 6 months after DOSE 1 |
None |
|
|
|
≥18 |
1 mL (720 ELU HAV + 20 µg HBsAg) IM |
ACCELERATED DOSE 1: ≥18 years old DOSE 2: 7 days after DOSE 1 DOSE 3: 21–30 days after DOSE 1 |
12 months after DOSE 1 |
Abbreviations: HBsAg, hepatitis B surface antigen; IM, intramuscular; ELU, ELISA units inactivated HAV; HAV, hepatitis A virus.
1Consult the prescribing information for differences in dosing for patients receiving hemodialysis, and other immunocompromised patients.
Special Situations
The accelerated Twinrix vaccination schedule (0, 7, and 21–30 days, plus booster at 12 months) can be used for people traveling on short notice who face imminent HBV exposure or for emergency responders to disaster areas. Alternatively, Heplisav-B can be used as a 2-dose series at 0 and 4 weeks to protect against hepatitis B alone. Ideally, vaccination with Heplisav-B should begin ≥1 month before travel so the full vaccine series can be completed before departure. When using vaccines other than Heplisav-B, begin vaccination ≥6 months before scheduled travel. Because some protection is provided by 1 or 2 doses, initiate the vaccine series, if indicated, even if the series cannot be completed before departure. Vaccines will not confer optimal protection, however, until after the series is completed; advise travelers to complete the vaccine series upon return.
Safety & Adverse Reactions
Safe hepatitis B vaccines are available for people of all ages, and serious adverse reactions are rare. The most common adverse reactions are soreness at the injection site (3%–29%) and low-grade fever (temperature >99.9°F [37.7°C]; 1%–6%). Hepatitis B vaccines should not be administered to people with a history of hypersensitivity to any vaccine component, including yeast. The vaccine contains a noninfectious recombinant protein (hepatitis B surface antigen) and an adjuvant (either 1018 [small synthetic immunostimulatory cytidine- phosphate-guanosine oligodeoxynucleotide motif for Heplisav-B] or aluminum [for Engerix-B, Recombivax HB, PreHevbrio, Twinrix]).
HBV infection affecting a pregnant person can result in serious disease for the mother and chronic infection for the newborn. Limited data indicate no apparent increased risk for adverse events to the mother (or the developing fetus) after maternal vaccination with Engerix-B, Recombivax HB, or Twinrix; no data are available on the use of Heplisav-B or PreHevbrio in pregnant or breastfeeding people. Until safety data are available for Heplisav-B and PreHevbrio, therefore, pregnant or breastfeeding people needing hepatitis B vaccination should receive Engerix-B, Recombivax HB, or Twinrix.
Personal Protective Measures
As part of the pretravel education process, educate all travelers about exposure risks for hepatitis B and other bloodborne pathogens, including activities or procedures that involve piercing the skin or mucosa; receiving blood products; contaminated equipment used during cosmetic (e.g., tattooing or piercing), dental, or medical procedures; injection drug use; and unprotected sexual activity. Caution travelers against providers who use inadequately sterilized or disinfected equipment, who reuse contaminated equipment, or who do not use safe injection practices (e.g., reusing disposable needles and syringes).
HBV and other bloodborne pathogens can be transmitted if medical equipment is not sterile or if personnel do not follow proper infection-control procedures. Travelers should consider the health risks when receiving dental or medical care overseas; US embassy country-specific websites might have information on medical concerns. Advise travelers to strongly consider health risks before obtaining a body piercing or a tattoo when traveling to destinations where adequate sterilization or disinfection procedures might not be available or practiced.
CDC website: Hepatitis B
The following authors contributed to the previous version of this chapter: Aaron M. Harris