Volume 11, Number 3—March 2005
Letter
Rectal Lymphogranuloma Venereum, France
Cite This Article
Citation for Media
To the Editor: Lymphogranuloma venereum (LGV), a sexually transmitted disease (STD) caused by Chlamydia trachomatis serovars L1, L2, or L3, is prevalent in tropical areas but occurs sporadically in the western world, where most cases are imported (1). LVG commonly causes inflammation and swelling of the inguinal lymph nodes, but it can also involve the rectum and cause acute proctitis, particularly among men who have sex with men. However, LGV serovars of C. trachomatis remain a rare cause of acute proctitis, which is most frequently caused by Neisseria gonorrhoeae or by non-LGV C. trachomatis (2). In 1981, in a group of 96 men who have sex with men with symptoms suggestive of proctitis in the United States, Quinn et al. found that 3 of 14 C. trachomatis infections were caused by LGV serovar L2 (3). In France, 2 cases of rectal LGV were reported in an STD clinic in Paris from 1981 to 1986 (4). In 2003, an outbreak of 15 rectal LGV cases was reported among men who have sex with men in Rotterdam; 13 were HIV-infected, and all reported unprotected sex in neighboring countries, including Belgium, France, and the United Kingdom (5). At the same time, a rise in C. trachomatis proctitis (diagnosed by using polymerase chain reaction [PCR]; [Cobas Amplicor Roche Diagnostic System, Meylan, France]) was detected in 3 laboratories in Paris and in the C. trachomatis national reference center located in Bordeaux. To identify the serovars of these C. trachomatis spp., all stored rectal specimens were analyzed by using a nested omp1 PCR-restriction fragment length polymorphism assay. The amplified DNA product was digested by restriction enzymes. Analysis of digested DNA was performed by electrophoresis. Patterns were compared visually with reference patterns (6).
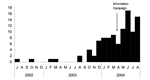
From January 1, 2003, to March 31, 2004, a total of 44 of 124 male rectal swabs were positive for C. trachomatis. Of those, 38 were identified as belonging to the L2 serotype, which confirms the diagnosis of rectal LGV. Epidemiologic information was retrospectively obtained by clinicians through review of medical records, telephone interview, or both. A complete history was available for 14 of the 38 cases. All 14 men reported unprotected anal sex with anonymous male sex partners in France, and none reported a stay in an LGV-endemic area. Their mean age was 40 years (31–50); 8 were HIV-infected, and 9 had another concomitant STD. The mean duration of symptoms before LGV diagnosis was 50 days (range 11–120 days). All 14 patients had symptoms of acute proctitis, including rectal pain, discharge, and tenesmus, and 3 (all HIV-infected) had fever. Deep, extended rectal ulcerations were reported in 8 patients, 3 of whom were HIV-infected and had lesions suggestive of rectal carcinoma. In 1 patient in whom a late diagnosis was made 4 months after the onset of symptoms, a rectal tumorlike stricture was observed. All 14 patients were treated with tetracycline for a mean duration of 16 days (range 10–60 days).
An information campaign among microbiologists and clinicians and a sentinel LGV surveillance system were launched in April 2004. Subsequently, LGV was diagnosed in 65 additional male patients, some retrospectively. In total, rectal LGV was diagnosed in 103 patients from July 2002 to August 2004 (Figure).
Prompt diagnosis and treatment is indeed paramount to prevention and control. Diagnosis may be further hampered because rectal LGV may mimic other conditions such as rectal carcinoma or Crohn disease. Treatment duration should be no shorter than 21 days, and follow-up examinations should be conducted until all signs and symptoms have resolved (7,8). If left untreated, rectal LGV could lead to serious complications such as rectal stricture (1). If recently exposed to infection, sexual contacts should receive prophylactic treatment to prevent reinfection and to eliminate a potential reservoir. The emergence of rectal LGV, characterized by deep mucosal ulcerations and frequently occurring in HIV-infected men who have sex with men, is a serious concern for the gay community in Europe.
References
- Perrine PL, Stamm WE. Lymphogranuloma venereum. In: Holmes KK, Sparling PF, Mardh PA, Lemon SM, Stamm WE, Piot P, et al., editors. Sexually transmitted diseases. New York: McGraw-Hill; 1999. p. 423–32.
- Klausner JD, Kohn R, Kent C. Etiology of clinical proctitis among men who have sex with men. Clin Infect Dis. 2004;38:300–2. DOIPubMedGoogle Scholar
- Quinn TC, Goodell SE, Mkrtichian E, Schuffler MD, Wang SP, Stamm WE, Chlamydia trachomatis proctitis. N Engl J Med. 1981;305:195–200. DOIPubMedGoogle Scholar
- Scieux C, Barnes A, Bianchi I, Casin I, Morel P, Perol Y. Lymphogranuloma venereum: 27 cases in Paris. J Infect Dis. 1989;160:662–8. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Lymphogranuloma venereum among men having sex with men—Netherlands, 2003–2004. MMWR Morb Mortal Wkly Rep. 2004;53:985–8.PubMedGoogle Scholar
- Rodriguez P, Vekris A, de Barbeyrac B, Dutilh B, Bonnet J, Bebear C. Typing of Chlamydia trachomatis by restriction endonuclease analysis of the amplified major outer membrane protein gene. J Clin Microbiol. 1991;29:1132–6.PubMedGoogle Scholar
- Centers for Diseases Control and Prevention. Sexually transmitted diseases treatment guidelines 2002. MMWR Recomm Rep. 2002;51(RR-6):1–78.PubMedGoogle Scholar
- Clinic Effectiveness Group. National guidelines for the management of lymphogranuloma venereum. Sex Transm Infect. 1999;75:40–2.
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 11, Number 3—March 2005
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Magid Herida, Institut de Veille Sanitaire, 12 Rue du Val d’Osne 94415, Saint-Maurice, France; fax: 33141796766
Top