Volume 12, Number 1—January 2006
Dispatch
Pathogen Transmission and Clinic Scheduling
Cite This Article
Citation for Media
Abstract
We developed a model of pathogen dissemination in the outpatient clinic that incorporates key kinetic aspects of the transmission process, as well as uncertainty regarding whether or not each incident patient is contagious. Assigning appointments late in the day to patients suspected of being infectious should decrease pathogen dissemination.
Pathogen dissemination within hospitals has been extensively analyzed (1). However, it has been the subject of far fewer investigations within the outpatient clinic. We developed a model of pathogen dissemination in the outpatient clinic and explored the anticipated effects of a system-based intervention (temporal segregation of suspected infectious and noninfectious patients) and an individual-based intervention (increased compliance with hand hygiene) on the risk that an uncontaminated patient will become contaminated during a clinic visit. An annotated copy of the model, as well as more detailed simulations and supporting material, may be obtained from the corresponding author.
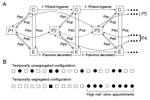
We treat pathogen dissemination as a stochastic (chance-based) sequence of discrete encounters between incident patients (P) who are either infectious or noninfectious, a caregiver (C), and the environment (fomites, such as surfaces or waiting room magazines, [E]), each of which can be contaminated or uncontaminated. Four classes of encounter exist: 1) caregiver-patient, 2) caregiver-environment, 3) patient-environment, and 4) patient-patient. For each class of encounter, a user specifies a probability that a contaminated or infectious participant can transmit the pathogen to an uncontaminated participant (caregiver, environment [fomites], and surrounding patients). A contaminated caregiver can, in turn, contaminate subsequent patients (Figure 1). The contamination probabilities are not predicated on a specific mode of transmission but represent the gross probability that a contaminated or infectious participant will contaminate an uncontaminated participant during an encounter. With the exception of patient-to-patient transmission (see below) the contamination probabilities can be asymmetrical. Asymmetrical contamination probabilities allow consideration of droplet transmission; e.g., an infectious patient could cough on a caregiver, who then transfers the pathogen from his or her hands to the next patient.
An infectious patient can also contaminate the 4 patients surrounding him or her in the clinic queue (2 preceding and 2 following patients). The model does not incorporate patient-patient transmission by contaminated patients, given the relatively low frequency of intimate patient-to-patient physical contact in the clinic.
If contaminated, the caregiver may be decontaminated, e.g., by hand hygiene or pathogen attrition, with a specified probability after each patient visit. Similarly, a specified probability exists that the environment, if contaminated, will be decontaminated between visits. These 2 decontamination processes (for the caregiver and the environment) are independent. Both the caregiver and the environment can be recontaminated after decontamination. The contamination probabilities for transmission from a contaminated environment to a patient or the caregiver are fixed (i.e., not a function of the number of preceding infectious persons).
The model generates a random number for each class of encounter between potentially contaminated or infectious and uncontaminated participants during each clinic appointment slot. If this random number is less than the contamination probability specific to the class of interaction being considered, the uncontaminated member becomes contaminated during the encounter.
The user specifies the population prevalence of the pathogen. A screening instrument for classifying patients as high or low risk for being infectious is assumed to exist. We assume that the screening instrument can be applied before the patient's clinic visit (at the time of appointment scheduling). The screening instrument could comprise a symptom inventory, knowledge of recent travel, household exposure, membership in a known high-risk group, or a combination of these or other elements. Such screening instruments exist for methicillin-resistant Staphylococcus aureus, vancomycin-resistant enterococci, and severe acute respiratory syndrome (SARS) (2–5). The postulated instrument has a defined sensitivity and specificity; values that can be combined with the population prevalence of infectious patients to determine how many patients deemed at high risk or low risk are actually contagious.
Incident patients are assigned to appointment times following 1 of 2 protocols. In the baseline protocol, patients are randomly assigned appointments without regard to their risk status. In the segregated protocol, patients at high risk are assigned to appointments at the end of the clinic day (Figure 1). Clinics that segregate high-risk patients to later appointments might also adopt more stringent infection control strategies during the portion of the day populated by high-risk patients. Accordingly, the user can specify values for each contamination probability that differ between low-risk (early in the day) and high-risk (late in the day) clinic slots.
We modeled 1 day in a clinic in which 20 patients are seen by 1 caregiver. The model predicts the likelihood (risk) that a previously uncontaminated patient will become contaminated during his or her clinic visit. The model also predicts the risk that a patient who is classified as high-risk based on the screening instrument, but who is not infectious, will be contaminated in the segregated configuration.
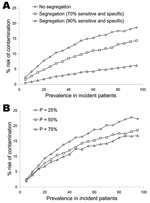
Figure 2 illustrates the effect of temporally segregating patients deemed to be at high risk of being infectious to clinic appointments late in the clinic schedule (panel A) and the consequences of changes in caregiver hand-hygiene compliance (panel B). Relevant pathogen-contamination probabilities were arbitrarily fixed at 20%, a level that is reasonable, and possibly conservative, for a wide range of pathogens (Table 1) (6–14). Temporal segregation substantially decreases the risk for pathogen dissemination, an effect that is at least comparable in magnitude to that arising from changes in the likelihood of effective hand hygiene within the clinically relevant range (15).
Table 2 addresses the ethical concern of the increase in contamination risk faced by noninfectious incident patients who are classified as high risk, using the same system inputs shown in Figure 2. The absolute risk for contamination faced by noninfectious but high-risk patients increases at all levels of prevalence and screening tool sensitivity and specificity; however, this increase does not exceed 6% (in the setting of a completely ineffective screening tool). More extensive analysis demonstrates that selectively deploying more aggressive infection-control practices to slots designated as high risk can lower the risk faced by noninfectious but nominally high-risk patients to below its value in the unsegregated configuration. These data are available from the corresponding author.
The results do not address direct patient-to-patient pathogen transmission; such transmission does not change the qualitative predictions of the model. The model does not incorporate either the potential for the environmental pathogen load to increase over time or differences in host susceptibility. The potential for cross-transmission of additional pathogens that have a higher prevalence in the high-risk group is also not addressed. For example, a population of patients deemed at high risk of having influenza or SARS might also have a higher prevalence of other transmissible pathogens, such as Mycobacterium tuberculosis.
Improved hand hygiene, barrier precautions, patient use of hand sanitizers or facemasks, and avoidance of environmental fomites can each diminish dissemination risk. However, these measures depend on individual behavior, rendering them susceptible to implementation failure. Temporal segregation adds a system-based layer of protection to such behavior-based interventions, providing an additional barrier to pathogen dissemination that is not critically dependent on individual compliance. When additional data addressing pathogen transmission within the clinic become available (currently such data are sparse), mathematical models could help guide the allocation of resources required to support system-based infection-control measures. Moreover, numerical experimentation could help inform the design of infection-control strategies for pathogens that have transmission dynamics that are not yet well characterized.
Dr Hotchkiss is assistant professor of critical care medicine at the University of Pittsburgh School of Medicine. He has a long-standing interest in mathematical modeling; his more recent interests include dynamic systems analyses of infectious processes and pathogen dissemination.
Acknowledgments
We thank the anonymous reviewers for their insightful suggestions and criticisms.
This work was supported by National Institutes of Health grant R21AI55818-02 to J.R.H.
References
- Furuno JP, Harris AD, Wright MO, McGregor JC, Venezia RA, Zhu J, Am J Infect Control. 2004;32:436–40. DOIPubMedGoogle Scholar
- Tacconelli E, Karchmer AW, Yokoe D, D'Agata EM. Clin Infect Dis. 2004;39:964–70. DOIPubMedGoogle Scholar
- Su CP, Chiang WC, Ma MH, Chen SY, Hsu CY, Ko PC, Ann Emerg Med. 2004;43:34–42. DOIPubMedGoogle Scholar
- Leung GM, Rainer TH, Lau FL, Wong IO, Tong A, Wong TW, Ann Intern Med. 2004;141:333–42.PubMedGoogle Scholar
- Tenorio AR, Badri SM, Sahgal NB, Hota B, Matushek M, Hayden MK, Clin Infect Dis. 2001;32:826–9. DOIPubMedGoogle Scholar
- Smith TL, Iwen PC, Olson SB, Rupp ME. Infect Control Hosp Epidemiol. 1998;19:515–8. DOIPubMedGoogle Scholar
- Zachary KC, Bayne PS, Morrison VJ, Ford DS, Silver LC, Hooper DC. Infect Control Hosp Epidemiol. 2001;22:560–4. DOIPubMedGoogle Scholar
- Noskin GA, Stosor V, Cooper I, Peterson LR. Infect Control Hosp Epidemiol. 1995;16:577–81. DOIPubMedGoogle Scholar
- Boyce JM, Potter-Bynoe G, Chenevert C, King T. Infect Control Hosp Epidemiol. 1997;18:622–7. DOIPubMedGoogle Scholar
- Bean B, Moore BM, Sterner B, Peterson LR, Gerding DN, Balfour HH Jr. J Infect Dis. 1982;146:47–51. DOIPubMedGoogle Scholar
- Chen YC, Huang LM, Chan CC, Su CP, Chang SC, Chang YY, Emerg Infect Dis. 2004;10:782–8.PubMedGoogle Scholar
- Samore MH, Venkataraman L, DeGirolami PC, Arbeit RD, Karchmer AW. Am J Med. 1996;100:32–40. DOIPubMedGoogle Scholar
- Lankford MG, Zembower TR, Trick WE, Hacek DM, Noskin GA, Peterson LR. Emerg Infect Dis. 2003;9:217–23.PubMedGoogle Scholar
Figures
Tables
Cite This ArticleCrossRef reports the first author should be "Weinstein" not "Hota" in reference 14 "Hota, 2004".
Table of Contents – Volume 12, Number 1—January 2006
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
John R. Hotchkiss, Department of Critical Care Medicine, University of Pittsburgh, 646 Scaife Hall, 3550 Terrace St, Pittsburgh, PA 15261, USA; fax: 412-647-8060
Top