Volume 15, Number 2—February 2009
Dispatch
Novel Human Parechovirus from Brazil
Abstract
Human parechoviruses (HPeVs) were detected by reverse transcription–PCR in 16.1% of 335 stool samples from children <6 years of age with enteritis in Salvador, Brazil. Whole genome sequencing of 1 sample showed a novel HPeV that has been designated as HPeV8.
The human parechovirus (HPeV) species are small, nonenveloped RNA viruses that belong to the highly diversified family Picornaviridae (1). HPeV types 1 and 2 had been known as echoviruses 22 and 23 within the genus Enterovirus but were recognized in the early 1990s as an independent genus (2). Recognition of clinical relevance is increasing after 4 novel types were more recently described (3–7).
Seroprevalence studies from different countries indicate that almost the entire human adult population is infected. Predominantly in infants, HPeVs can cause a variety of clinical symptoms, including diarrhea, and respiratory infection (8,9). Recent data point toward substantial involvement in severe conditions, such as meningitis and infant sepsis, for which HPeV may constitute the second most frequent causative virus after enterovirus in young children (10). Different HPeV types may cause different clinical diseases (10,11). Unconnected diseases might be caused by yet unrecognized HPeVs.
To identify possibly unrecognized HPeVs, we systematically searched for HPeVs in patients in Brazil with enteritis. We used stool samples for the study because the related enteroviruses are preferentially transmitted through feces. Because reverse transcription–PCR (RT-PCR) for the genus Enterovirus cannot detect HPeVs (9,10), a broad-range real-time RT-PCR assay was developed that can detect all known parechovirus types. The assay also can detect new HPeV types (9).
The study cohort comprised 335 stool samples from Brazilian infants and children <6 years of age with acute diarrhea, defined as >3 watery stools in the previous 24 hours and lasting no longer than 13 days. From February 2006 through August 2007, children were seen as outpatients or were hospitalized at the University Hospital in Salvador de Bahia, Brazil, because of severe dehydration. All analyses were performed by using an ABI 7500 real-time RT-PCR (Applied Biosystems, Foster City, CA, USA) and an ABI 3100 automated sequencing platform (Applied Biosystems) at the local Infectious Disease Research Laboratory. Informed consent was obtained from the mothers of all enrolled patients. The study was approved by the institutional ethics committee.
We performed RNA extraction and real-time RT-PCR as described (9). A total of 16.1% of samples tested positive for HPeV. Many samples yielded low HPeV RNA concentrations indicated by threshold cycles later than 32 in real-time RT-PCR.
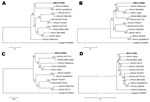
The viral protein (VP) 1 capsid protein gene has been established for molecular typing of HPeV (3). Consensus primers for amplification and sequencing of the VP1 gene were developed. Primer sequences were VP1 forward 5′-CCATARTGYTTRTARAARCCYCT-3′ and VP1 reverse 5′-CARAAYTCDTGGGGYTCMCARATGG-3′. VP1 RT-PCR was successful in only 11 of 54 HPeV-positive samples, consistent with low RNA concentrations in most samples. Ten of the 11 sequenced samples were of known and well-characterized types (type 1, 7 samples; type 5, 2 samples; and type 6, 1 sample). These samples were not further analyzed. One sample showed a VP1 sequence that clustered with none of the known HPeV types in phylogenetic analysis (Figure 1). To determine whether this virus represented a new type, its complete genome except the first 27 nucleotides of the 5′ terminus was amplified by overlapping PCR fragments, and the full nucleotide sequence was determined as described previously (9) (GenBank accession no. EU716175).
The best matching sequence was HPeV type 4 with 76.3% aa identity (Table). Lowest aa identity was 69.3% between the new sequence and type 5. Genetic identity to Ljungan virus, a rodent parechovirus, was only 44.5%, comparable to all previously described HPeVs. Phylogenetic segregation from all known HPeVs was obvious not only in VP1 but also along the structural proteins VP0 and VP3 (aa identities with established HPeV types ranges 71.2%–80.2% in VP0 and 74.1%–80.0% in VP3) (Figure 1).
Nonstructural family Picornaviridae genes are highly recombined (1,3,12,13), resulting in a mosaic structure that limits their utility in phylogenetic analysis. This characteristic also was the case for the whole nonstructural region (comprising regions P2 and P3) of the novel virus. It was not clearly segregated from that of all other HPeV prototype strains, showing closest overall relationship with HPeV type 1 strain Harris and the HPeV6 prototype strain NII561-2000 (Figure 1). To identify possible recombination in the P2/3 region of the novel virus, we conducted Sim Plot analysis (http://sray.med.som.jhmi.edu/SCRoftware/simplot) (Figure 2). We found no clear similarity with any of the established HPeV prototype strains. In contrast to other HPeV prototype strains, BootScan analysis showed no evidence for recombination with other prototype strains in the nonstructural gene region. However, this analysis could also not exclude recombination with any other HPeV because of the small number of HPeV full genome sequences currently available.
The new HPeV lacked a typical RGD (Arg-Gly-Asp) aa motif in the VP1 C terminus. This motif has proven important for HPeV type 1 infectivity, presumably because of interaction with cellular receptors (14). Such a motif is present in all known HPeV strains except type 3, and some researchers have suggested that the latter may use a different receptor for cell entry (3,13).
Most HPeV types have been identified only recently. The associated spectrum of diseases is not fully understood and probably has been underestimated. Recent data indicate that HPeVs may cause severe clinical conditions, such as infant sepsis and meningitis, in addition to acute diarrhea (10). Prevalence in young children with diarrhea was >16% in previous studies; more important, <8% of meningitis cases showed evidence of HPeV (9–11). The molecular ecology of HPeV seems especially relevant in view of their diversified and strain-dependent pathogenesis (10,11).
This report on HPeVs from Brazil confirms their global distribution. The level of diversification between the novel parechovirus and established HPeV types is clearly higher than the 20% aa distance in the VP1 protein, which resembles the distance between serotypes of enteroviruses (1,3) and exceeds the definition threshold of HPeV types (3). During revision of this report, the virus received the designation HPeV8 by the ICTV Picornavirus Study Group (www.picornastudygroup.com/types/parechovirus/hpev.htm). Like HPeV3, HPeV8 lacks the RGD motif; some researchers have suggested that HPeV3 may use a different receptor than other HPeV types for cell entry (3,13). Of all HPeV types, type 3 has been most strongly associated with severe neurologic and systemic clinical conditions (10,11,15). The lack of an RGD motif might implicate a different cell or tissue tropism for HPeV8 as well. The search for unknown HPeVs should be extended to other clinical conditions thus far not associated with HPeV.
Dr Drexler is a physician and clinical virologist affiliated with the University of Bahia, the Bernhard Nocht Institute, and the University of Bonn. His research interests are the implementation of methods for affordable viral load monitoring and the characterization of novel human and zoonotic viruses.
Acknowledgments
We are grateful to C. Pedroso, C. Brites, E. Netto, D. Brasil Pedral-Sampaio, A. Borges Carneiro, and D. Custódio Leal for outstanding support.
The study was funded by a research grant from the Foundation for Research Support of the State of Bahia (FAPESB), project code APR 125/2006. C.D. was supported by European Union contract no. SSPE-CT-2005-022639.
References
- Stanway G, Brown F, Christian P, Hovi T, Hyypiä T, King AM, Family Picornaviridae. In: Fauquet CM, Mayo MA, Maniloff J, Desselberger U, Ball LA, editors. Virus taxonomy: eighth report of the International Committee on Taxonomy of Viruses. London: Elsevier/Academic Press; 2005. p. 757–78.
- Hyypia T, Horsnell C, Maaronen M, Khan M, Kalkkinen N, Auvinen P, A distinct picornavirus group identified by sequence analysis. Proc Natl Acad Sci U S A. 1992;89:8847–51. DOIPubMedGoogle Scholar
- Al-Sunaidi M, Williams CH, Hughes PJ, Schnurr DP, Stanway G. Analysis of a new human parechovirus allows the definition of parechovirus types and the identification of RNA structural domains. J Virol. 2007;81:1013–21. DOIPubMedGoogle Scholar
- Benschop KS, Schinkel J, Luken ME, van den Broek PJ, Beersma MF, Menelik N, Fourth human parechovirus serotype. Emerg Infect Dis. 2006;12:1572–5.PubMedGoogle Scholar
- Boivin G, Abed Y, Boucher FD. Human parechovirus 3 and neonatal infections. Emerg Infect Dis. 2005;11:103–5.PubMedGoogle Scholar
- Oberste MS, Maher K, Pallansch MA. Complete sequence of echovirus 23 and its relationship to echovirus 22 and other human enteroviruses. Virus Res. 1998;56:217–23. DOIPubMedGoogle Scholar
- Watanabe K, Oie M, Higuchi M, Nishikawa M, Fujii M. Isolation and characterization of novel human parechovirus from clinical samples. Emerg Infect Dis. 2007;13:889–95.PubMedGoogle Scholar
- Stanway G, Joki-Korpela P, Hyypia T. Human parechoviruses—biology and clinical significance. Rev Med Virol. 2000;10:57–69. DOIPubMedGoogle Scholar
- Baumgarte S, de Souza Luna LK, Grywna K, Panning M, Drexler JF, Karsten C, Prevalence, types, and RNA concentrations of human parechoviruses, including a sixth parechovirus type, in stool samples from patients with acute enteritis. J Clin Microbiol. 2008;46:242–8. DOIPubMedGoogle Scholar
- Wolthers KC, Benschop KS, Schinkel J, Molenkamp R, Bergevoet RM, Spijkerman IJ, Human parechoviruses as an important viral cause of sepsislike illness and meningitis in young children. Clin Infect Dis. 2008;47:358–63. DOIPubMedGoogle Scholar
- Benschop KS, Schinkel J, Minnaar RP, Pajkrt D, Spanjerberg L, Kraakman HC, Human parechovirus infections in Dutch children and the association between serotype and disease severity. Clin Infect Dis. 2006;42:204–10. DOIPubMedGoogle Scholar
- de Souza Luna LK, Baumgarte S, Grywna K, Panning M, Drexler JF, Drosten C. Identification of a contemporary human parechovirus type 1 by VIDISCA and characterisation of its full genome. Virol J. 2008;5:26. DOIPubMedGoogle Scholar
- Benschop KS, Williams CH, Wolthers KC, Stanway G, Simmonds P. Widespread recombination within human parechoviruses: analysis of temporal dynamics and constraints. J Gen Virol. 2008;89:1030–5. DOIPubMedGoogle Scholar
- Boonyakiat Y, Hughes PJ, Ghazi F, Stanway G. Arginine-glycine-aspartic acid motif is critical for human parechovirus 1 entry. J Virol. 2001;75:10000–4. DOIPubMedGoogle Scholar
- van der Sanden S, de Bruin E, Vennema H, Swanink C, Koopmans M, van der Avoort H. Prevalence of human parechovirus in the Netherlands, 2000 to 2007. J Clin Microbiol. 2008;46:2884–9. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 15, Number 2—February 2009
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Christian Drosten, Institute of Virology, University of Bonn Medical Centre, 53127 Bonn, Germany;
Top