Volume 5, Number 1—February 1999
Perspective
The Economic Impact of Staphylococcus aureus Infection in New York City Hospitals
Abstract
We modeled estimates of the incidence, deaths, and direct medical costs of Staphylococcus aureus infections in hospitalized patients in the New York City metropolitan area in 1995 by using hospital discharge data collected by the New York State Department of Health and standard sources for the costs of health care. We also examined the relative impact of methicillin-resistant versus -sensitive strains of S. aureus and of community-acquired versus nosocomial infections. S. aureus-associated hospitalizations resulted in approximately twice the length of stay, deaths, and medical costs of typical hospitalizations; methicillin-resistant and -sensitive infections had similar direct medical costs, but resistant infections caused more deaths (21% versus 8%). Community-acquired and nosocomial infections had similar death rates, but community-acquired infections appeared to have increased direct medical costs per patient ($35,300 versus $28,800). The results of our study indicate that reducing the incidence of methicillin-resistant and -sensitive nosocomial infections would reduce the societal costs of S. aureus infection.
Each year approximately two million hospitalizations result in nosocomial infections (1). In a study of critically ill patients in a large teaching hospital, illness attributable to nosocomial bacteremia increased intensive care unit stay by 8 days, hospital stay by 14 days, and the death rate by 35% (2). An earlier study found that postoperative wound infections increased hospital stay an average of 7.4 days (3).
Staphylococcus aureus was the most common cause of nosocomial infections reported in the National Nosocomial Surveillance System between 1990 to 1996 (4). The leading cause of nosocomial pneumonia and surgical site infections and the second leading cause of nosocomial bloodstream infections (4), S. aureus also causes community-acquired infections (e.g., osteomyelitis and septic arthritis, skin infections, endocarditis, and meningitis). More than 95% of patients with S. aureus infections worldwide do not respond to first-line antibiotics such as penicillin or ampicillin (5). Additionally, methicillin-resistant strains of S. aureus (MRSA) are common. First reported in the 1960s (6), MRSA has become increasingly prevalent since the 1980s (7,8) and is now endemic in many hospitals and even epidemic in some, with resistance in approximately 30% of all S. aureus infections (8).
Vancomycin is the only drug that can consistently treat MRSA. However, beginning in 1989, hospitals have reported a rapid increase in vancomycin resistance in enterococci (VRE) (9). Increased vancomycin use helps select for VRE, and even a small increase in incidence of VRE infection could lead to cross-resistance in S. aureus, since genes conferring vancomycin resistance might be transferred from VRE (10). In 1996, Japan reported the first case of S. aureus infection with intermediate resistance to vancomycin (11). In 1997, two unrelated cases of S. aureus infection with intermediate resistance to vancomycin were reported in the United States (Michigan and New Jersey) (12). In both cases, patients had been treated with multiple courses of vancomycin for repeated MRSA infections over the 6 months before the S. aureus infection with intermediate resistance to vancomycin; additionally, VRE colonization had been diagnosed 7 months before the S. aureus infection with intermediate resistance to vancomycin in the New Jersey patient. The emergence of S. aureus infection with intermediate resistance to vancomycin in the United States suggests that S. aureus strains are constantly evolving and full resistance may develop (12).
The various ways of controlling MRSA (13) are still being debated. The elimination of endemic MRSA in hospitals is difficult and costly (14-17). In general, infection control in the United States is less stringent than in Canada and in some European countries, where identification of known carriers, prospective surveillance of patients and hospital workers, and use of nasal mupirocin have helped control drug-resistant S. aureus infection rates (18).
Knowledge of the scope of the problem is helpful for hospital administrators, insurers, and medical personnel who make policy decisions on control measures to prevent the spread of MRSA and the emergence of vancomycin-resistant S. aureus. However, the economic cost of S. aureus infections is not well known. Many studies focus on the cost of nonorganism-specific nosocomial infections (2,19,20). Moreover, the reported cost of a nosocomial infection varies because of the wide range of study populations, sites of infection, and methods used (16,21). The few investigations into the cost of S. aureus infections have focused on the differential cost of MRSA and MSSA infections (22,23) and are case studies of outbreaks in single hospitals. Thus, they do not provide perspective on the scope of the problem for a population over time.
We estimated the incidence, death rate, and cost of S. aureus infections associated with hospitalization in the New York City metropolitan area in 1995. We selected this geographic region because of its high prevalence of multidrug-resistant infections (24,25). We also compared the relative contributions of nosocomial versus community-acquired infections and methicillin-sensitive (MSSA) versus methicillin-resistant S. aureus.
Data
The 1995 Statewide Planning and Research Cooperative System (SPARCS) Administratively Releasable File was the primary source of data (26). SPARCS is a database of all hospital discharges in New York state, as reported by hospitals to the State of New York Department of Health, and the Administratively Releasable File contains discharge information on hospital location, patient characteristics (age, sex, race, ethnicity), and visit characteristics (primary diagnosis, secondary diagnoses, primary procedure, secondary procedures, length of stay, total charges, patient status, and disposition). We analyzed data for hospitals in the following New York City metropolitan area counties: Bronx, Dutchess, Kings, Manhattan, Nassau, Orange, Putnam, Queens, Richmond, Rockland, Suffolk, Ulster, and Westchester. Data on infection incidence or resource use not in SPARCS were obtained through a comprehensive literature search or estimated by a clinical panel consisting of four physicians specializing in infectious disease. Other sources for cost information were the 1995 Medicare Fee Schedule (27) for physician fees and the 1995 Red Book (28) for outpatient pharmaceutical average wholesale prices.
Definitions
We identified patients with the most common types of hospital-associated S. aureus infections: pneumonia, bacteremia, endocarditis, surgical site infections, osteomyelitis, and septic arthritis (Table 1) from SPARCS, which uses the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnosis codes (29). With the exception of ICD-9-CM code 482.4 (staphylococcal pneumonia) and 038.1 (staphylococcal septicemia), these codes are not organism-specific.
To identify S. aureus infections, we used the nonorganism–specific codes in conjunction with an additional ICD-9-CM code to identify the bacterial agent (i.e., 041.11 bacterial infection due to S. aureus in conditions classified elsewhere and of unspecified site). Patients with multiple infections were counted only once in the overall incidence rate. Their primary or first occurrence of a diagnosis of interest was used.
Because source of infection (nosocomial versus community-acquired) is not reported in SPARCS, we assumed that specific types of disease were either nosocomial or community-acquired on the basis of the clinical panel opinion (Table 2).
Modeling the Incidence Rate
ICD-9-CM code 041.11 (bacterial infection due to S. aureus) is not widely used by reporting hospitals. Therefore, the incidence of S. aureus infections based on the counts of 041.11 in SPARCS would underestimate the number of cases. We estimated the incidence of S. aureus infections (except pneumonia) as follows (Table 3): the total incidence of each type of infection (e.g., endocarditis) in SPARCS was multiplied by the estimated percentage attributable to S. aureus (determined by research or clinical panel opinion) to give the total number of infections due to S. aureus. The incidence of pneumonia was equated with the occurrence of the ICD-9-CM code 482.4 (staphylococcal pneumonia). For ICD-9-CM code 038.1 (staphylococcal septicemia), we assumed that only 50% of infections were attributable to S. aureus (with the remainder attributable to S. epidermidis) (30).
Modeling Death Rates
The death rates attributable to bacteremia, endocarditis, or community-acquired pneumonia were assumed to be equal to the death rates found when these infections were coded as a primary diagnosis in SPARCS and 041.11 was used as a secondary diagnosis. For nosocomial pneumonia, however, we assumed that the attributable death rate was a percentage of the actual death ratefor ventilator-associated pneumonia patients, death rate is a function of both the severity of underlying disease and the pneumonia. A series of matched-cohort studies have demonstrated that the death rate attributable to ventilator-associated pneumonia is 0% to 57% of the actual death rate (36-39). On the basis of this research and expert panel judgment, nosocomial pneumonia in ventilator-associated pneumonia patients (identified by ICD-9-CM V46.0 or V46.1) was assumed to have an attributable death rate of 50% of the death rate found in SPARCS (30,40). We assumed that the attributable death rate of nonventilator-associated pneumonia was the death rate found in SPARCS. On the basis of the low death rate found in SPARCS (approximately 2%), we assumed that no deaths were attributable to osteomyelitis, septic arthritis, or surgical site infections.
Modeling Direct Medical Costs
Direct medical costs were defined as hospital costs attributable to S.aureus infection, professional fees incurred during hospitalization, and costs of other infection-related medical services provided after discharge. For each infection, total direct medical costs were calculated by multiplying the average direct medical cost per patient by the incidence of disease. Average hospital costs attributable to S. aureus per patient were assumed to be equal to the average hospital charge from SPARCS when the infection (e.g., pneumonia, bacteremia) was coded as a primary diagnosis and 041.11 was used as a secondary diagnosis. Professional fees incurred during hospitalization include physician visits and consultations for evaluation and management, as well as radiologic, surgical, and anesthesiologic costs. The average frequency of physician services per patient was based on clinical panel estimates. Costs of these services were based on 1995 Medicare Payment Rates for the Long Island, New York, area as an intermediate point between New York City costs and those of outlying counties. Costs of medical services after discharge include those of postdischarge complications (e.g., abscesses, aneurysms) requiring rehospitalization, home-based intravenous antibiotic therapy, and outpatient oral antibiotic therapy. The average frequency of other medical services provided per patient was based on clinical panel estimates. Costs of hospital readmission were based on SPARCS charges; costs of home-based intravenous therapy were based on literature estimates (40,41); and costs of outpatient medications were based on average wholesale prices (25).
Modeling MRSA and MSSA S. aureus Infections
SPARCS does not identify MRSA or MSSA infections, and a code for infection with a drug-resistant organism (V09) is rarely used. Therefore, we modeled the comparative incidence, death rate, and cost of MRSA and MSSA. We computed the incidence of MRSA and MSSA infections by using the estimate that 29% of infections were due to MRSA (8). The clinical panel estimated that 10% of community-acquired infections were due to MRSA (includes infections acquired at long-term care facilities). The number of deaths for MRSA and MSSA infections was estimated as follows: the clinical panel estimated a risk ratio for death rates of MRSA and MSSA infections, and deaths due to MRSA and MSSA infections were calculated from the estimated risk ratio and the overall number of deaths due to S. aureus infection. We estimated the direct medical cost per patient for MRSA and MSSA infections as follows: differences in resource use for those with MRSA and MSSA infections were identified by the clinical panel; these differences were converted into differences in cost using a method similar to that described above for modeling direct medical costs; and average costs for MRSA and MSSA infections were calculated by using the average cost for an S. aureus infection and the average difference in cost between MRSA and MSSA infections.
S. aureus Infection
Of 1,351,362 nonobstetrical hospital discharges in SPARCS for New York City in 1995, an estimated 13,550 (1.0%) were discharges of patients with S. aureus infections (Table 4). The total direct medical costs incurred by these patients was an estimated $435.5 million—average length of stay nearly 20 days, direct cost of infection, $32,100 (Table 4). The number of deaths was estimated at 1,400 (a 10% death rate). In contrast, the hospital charges for the average hospital stay in SPARCS (for all nonobstetrical discharges) were $13,263—average length of stay 9 days, death rate 4.1%. Thus, patients with S. aureus infection had approximately twice the cost, length of stay, and death rate of a typical hospitalized patient.
Pneumonia and bacteremia represented most S. aureus infections and accounted for 60% of the total direct medical costs and 97% of the number of deaths. Endocarditis caused the longest stay (26 days) and highest direct cost per patient ($47,200); surgical site infection caused the shortest stay (14 days) and lowest direct cost per patient ($21,810). Hospital charges were an average of $29,000 (90% of the total costs); professional fees were an average of $2,300 (7%); and postdischarge costs represented $800 (3%) (Table 5).
Nosocomial Infection
Nosocomial infections accounted for 46% of the total incidence of S. aureus infections (6,300 infections), while community-acquired infections accounted for 54% (7,250 infections) (Table 6). Community-acquired pneumonia as a primary diagnosis accounted for 12% (1,500) of the total cases. If community-acquired pneumonia is assumed to be mostly acquired in long-term care facilities, most infections (58%) were acquired institutionally. The cost attributable to community-acquired infections ($35,300) was approximately $6,500 higher on a per patient basis than the cost attributable to nosocomial infections ($28,800). The death rates attributable to community-acquired and nosocomial infections were similar (10.5% and 10.1%).
MRSA Infection
MRSA infections accounted for 21% (2,780) of the total S. aureus infection incidence (29% of 6,300 nosocomial infections plus 10% of 7,250 community-acquired infections), while MSSA infections accounted for 79% (10,770) of total infections (Table 6). The attributable cost of a patient with MRSA was approximately $2,500 higher than the attributable cost of a patient with MSSA ($34,000 versus $31,500). The higher cost of MRSA infections is due to the higher cost of vancomycin, longer hospital stay, and the cost of patient isolation procedures. For nosocomial infections alone, the cost attributable to MRSA was approximately $3,700 higher on a per patient basis than the cost attributable to MSSA infections ($31,400 versus $27,700). The death rate attributable to MRSA infections was estimated at more than 2.5 times higher than that attributable to MSSA infections (21% versus 8%).
Although assumed to be underused in SPARCS, the ICD-9-CM code 041.11 represents a lower boundary of the total incidence of S. aureus infection. In SPARCS, code 041.11 was used 7,366 times associated with a diagnosis of interest (e.g., endocarditis) and represented a total cost of $236.4 million and a death rate (740 deaths) of 2% (Table 7). The upper boundary of the total cost of S. aureus infections was calculated by assuming that all hospital charges and deaths of patients with S. aureus infections were attributable to the infection, representing a total cost of $599 million and a death rate of 14.5% (1,960 deaths). We conducted sensitivity analyses (varying the percentage of nosocomial MRSA; percentage of patients isolated; difference in length of stay between patients with MRSA and MSSA; attributable length of stay for patients with ventilator-associated pneumonia; number of S. aureus catheter infections; and percentage of S. aureus–caused bacteremia, septicemia, and postoperative infections) and found that the difference in cost per case between MRSA and MSSA infections was $1,700 to $5,100.
Our sensitivity analysis shows that we did not vastly over- or underestimate the direct medical costs of S. aureus infections in New York City. However, the study had several limitations; it was retrospective, and the data sources were not validated by other means (e.g., interviews or chart review). Therefore, coding errors in this database may affect the results. The clinical panel estimates we used to model differences between MRSA and MSSA may lead to some inaccuracy in those difference estimates. Thus, our comparison of costs and deaths between MRSA and MSSA should be viewed as a best approximation in the absence of case-control data or a multivariate analysis of a well-defined patient population.
Our estimates of the cost per infection are generally higher than estimates in studies reviewed by Jarvis (19). A major reason may be our focus on New York City, where costs are much higher than in other areas of the United States. In addition, earlier studies have used only hospital costs. Our perspective was societal; therefore, we included physician fees and outpatient costs, as well as hospital charges. Finally, most of these studies focused on nonorganism-specific nosocomial infections; S. aureus infections may have a higher average cost per episode than infections of other organisms (42). On the other hand, we used conservative estimates for certain costs. Medicare prices for professional services are generally lower than commercial rates. Also, we did not account for postdischarge complications that did not lead to hospitalization. Additionally, our societal estimates did not include the cost of dying or lost productivity associated with these illnesses. Despite its limitations, this study shows that hospitalizations associated with S. aureus are serious and have high medical costs and death rates. The average length of stay attributable to S. aureus infection for these patients was very high, 20 days—nearly three times the average for any other type of hospitalization (43). The increased length of stay in turn leads to increases in direct medical costs, with an average cost per case of $32,100 in 1995.
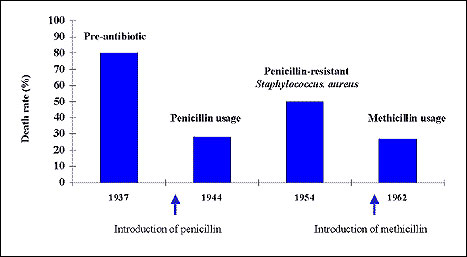
Treating an MRSA infection costs 6% to 10% more than treating an MSSA infection ($2,500 to $3,700 per case). This cost difference does not reflect MRSA's greater virulence; rather, it reflects the increased cost of vancomycin use and isolation procedures (if used). These estimates are slightly lower than the difference of $5,104 found by Wakefield et al. (21), perhaps because they focused on the cost of serious S. aureus infections, while our analysis examined all hospitalizable S. aureus infections. Patients with MRSA infections have a high average attributable death rate of 21% versus 8% for an MSSA infection. Some of the death rate difference may be related to the underlying condition of patients who become infected with MRSA (e.g., older patients, drug users, sicker patients, patients previously exposed to other antibiotics) (44) and to the lack of effectiveness of vancomycin itself in curing MRSA. (Vancomycin has a narrow therapeutic index that allows little room for increasing blood concentration without incurring substantial losses in tolerance [45]). Both MSSA and MRSA infections are associated with high costs and large numbers of deaths in the New York City metropolitan area. The costs and deaths associated with S. aureus infections may dramatically increase if the newly isolated S. aureus infection with intermediate resistance to vancomycin spreads or if VRSA emerges. For example, after penicillin-resistant S. aureus appeared in the 1950s, the death rate of bacteremia increased from 28% to 50% at the University of Minnesota (Figure) (46). After methicillin was introduced, the death rate decreased (47). Efforts should be directed toward reducing the incidence of MRSA and MSSA nosocomial infections to reduce their economic impact on society.
Death rate of staphylococcal bacteremia over time. (Data from 46, 47.)
Acknowledgments
We thank the clinical panel: Drs. Donald Armstrong, Donald Low, James Rahal, and Richard B. Roberts.
This study was sponsored by the Public Health Research Institute in conjunction with the Bacterial Antibiotic Resistance Group and The Rockefeller University. Funders included The New York Community Trust, The Horace W. Goldsmith Foundation, The United Hospital Fund of New York City, The Texaco Foundation, and the U.S. Centers for Disease Control and Prevention. Dr. Rubin, president of The Lewin Group, a Washington-based health-care consulting company, is a clinical professor of medicine at Georgetown University School of Medicine. From 1981-1984, he was assistant surgeon general in the U.S. Public Health Service and assistant secretary for planning and evaluation, U.S. Department of Health and Human Services. Address for correspondence: Robert J. Rubin, The Lewin Group, 9302 Lee Highway, Fairfax, VA 22031-1214, USA; fax: 703-218-5501.
References
- Haley RW, Culver DH, White JW, Morgan WM, Emori TG. The nationwide nosocomial infection rate: a new need for vital statistics. Am J Epidemiol. 1985;121:159.PubMedGoogle Scholar
- Pittet D, Tarara D, Wenzel RP. Nosocomial bloodstream infection in critically ill patients, excess length of stay, extra costs, and attributable mortality. JAMA. 1994;271:1598–601. DOIPubMedGoogle Scholar
- Brachman PS, Dan BB, Haley RW, Hooten TM, Garner JS, Allen JR. Nosocomial surgical infections: incidence and cost. Surg Clin North Am. 1980;60:15–25.PubMedGoogle Scholar
- Centers for Disease Control and Prevention. National Nosocomial Infection Surveillance System report: data summary from October 1986-April 1996. Atlanta (GA): U.S. Department of Health and Human Services; 1996.
- Barrett FF, McGehee RF, Finland M. Methicillin-resistant Staphylococcus aureus at Boston City hospital. N Engl J Med. 1968;279:441.PubMedGoogle Scholar
- Boyce JM. Increasing prevalence of methicillin-resistant Staphylococcus aureus in the United States. Infect Control Hosp Epidemiol. 1990;11:639–42. DOIPubMedGoogle Scholar
- Panlilio AL, Culver DH, Gaynes RP, Banerjee S, Henderson TS, Tolson JS, Methicillin-resistant Staphylococcus aureus in U.S. hospitals, 1975-1991. Infect Control Hosp Epidemiol. 1992;13:582–6. DOIPubMedGoogle Scholar
- Nosocomial enterococci resistant to vancomycin—United States, 1989-1993. MMWR Morb Mortal Wkly Rep. 1993;42:597–9.PubMedGoogle Scholar
- Recommendations for preventing the spread of vancomycin resistance recommendations of the Hospital Infection Control Practices Advisory Committee. MMWR Morb Mortal Wkly Rep. 1995;44(RR12):1–13.PubMedGoogle Scholar
- Reduced susceptibility of Staphylococcus aureus to vancomycin—Japan, 1996. MMWR Morb Mortal Wkly Rep. 1997;46:624–6.PubMedGoogle Scholar
- Update: Staphylococcus aureus with reduced susceptibility to vancomycin—United States, 1997. MMWR Morb Mortal Wkly Rep. 1997;46:813–5.PubMedGoogle Scholar
- Boyce JM, Jackson MM, Pugliese G, Batt MD, Fleming D, Garner JS, Methicillin-resistant Staphylococcus aureus (MRSA): a briefing for acute care hospitals and nursing facilities. Infect Control Hosp Epidemiol. 1994;15:105–15. DOIPubMedGoogle Scholar
- McManus AT, Mason AD, McManus WF, Pruitt BA. What's in a name? Is methicillin-resistant Staphylococcus aureus just another S. aureus when treated with vancomycin? Arch Surg. 1989;124:1456–9.PubMedGoogle Scholar
- Pittet D, Waldvogel FA. To control or not to control colonization with MRSA…that's the question! QJM. 1997;90:239–41. DOIPubMedGoogle Scholar
- Teare EL, Barrett SP. Stop the ritual of tracing colonised people. BMJ. 1997;314:665–6.PubMedGoogle Scholar
- Cookson B. Controversies: is it time to stop searching for MRSA? Screening is still important. BMJ. 1997;314:664–5.PubMedGoogle Scholar
- Casewell MW. New threats to the control of methicillin-resistant Staphylococcus aureus. J Hosp Infect. 1995;30(Suppl):465–71. DOIPubMedGoogle Scholar
- Jarvis WR. Selected aspects of the socioeconomic impact of nosocomial infections: morbidity, mortality, cost, and prevention. Infect Control Hosp Epidemiol. 1996;17:552–7. DOIPubMedGoogle Scholar
- Haley RW, White JW, Culver DH, Hughes JM. The financial incentive for hospitals to prevent nosocomial infections under the prospective payment system: an empirical determination from a nationally representative sample. JAMA. 1987;257:1611–4. DOIPubMedGoogle Scholar
- Wakefield DS, Pfaller MA, Hammons GT, Massanari RM. Use of the appropriateness evaluation protocol for estimating the incremental costs associated with nosocomial infections. Med Care. 1987;25:481–8. DOIPubMedGoogle Scholar
- Jernigan JA, Clemence MA, Stott GA, Titus MG, Alexander CH, Palumbo CM, Control of methicillin-resistant Staphylococcus aureus at a university hospital. Infect Control Hosp Epidemiol. 1995;16:686–96. DOIPubMedGoogle Scholar
- Wakefield DS, Helms CM, Massanari RM, Mori M, Pfaller M. Cost of nosocomial infection: relative contributions of laboratory, antibiotic and per diem costs in serious Staphylococcus aureus infections. Am J Infect Control. 1988;16:185–92. DOIPubMedGoogle Scholar
- Frieden TR, Fujiwara PI, Washko RM, Hamburg MA. Tuberculosis in New York City—turning the tide. N Engl J Med. 1995;333:229–33. DOIPubMedGoogle Scholar
- Frieden TR, Munsiff SS, Low DE, Willey BM, Williams G, Faur Y, Emergence of vancomycin-resistant enterococci in New York City. Lancet. 1993;342:76–9. DOIPubMedGoogle Scholar
- New York State Department of Health. 1995 Statewide Planning and Research Cooperative System (SPARCS) Administratively Releasable File. Albany (NY): The Department; 1997.
- Health Care Financing Administration. Physician fee schedule (CY 1995); payment policies and relative value adjustments. Fed Regist. 1994;59:63410–635.PubMedGoogle Scholar
- 1995 Drug Topics Red Book. Montvale (NJ): Medical Economics Company; 1995.
- International classification of diseases, 9th revision, clinical modifier: with color symbols: ICD-9-CM. 4th ed. Salt Lake City (UT): Medicode Publications; 1994.
- Lautenschlager S, Herzog C, Zimmerli W. Course and outcome of bacteremia due to Staphylococcus aureus: evaluation of different clinical case definitions. Clin Infect Dis. 1993;16:567–73.PubMedGoogle Scholar
- Espersen F. Identifying the patient risk for Staphylococcus aureus bloodstream infections. J Chemother. 1995;7:11–7.PubMedGoogle Scholar
- Muder R, Brennen C, Wagener M, Goetz A. Bacteremia in a long-term care facility: a five year prospective study of 163 consecutive episodes. Clin Infect Dis. 1992;14:647–54.PubMedGoogle Scholar
- Mandell GI, Bennett JE, Dolin R, eds. Mandell, Douglas and Bennett's principles and practices of infectious diseases. 4th ed. New York: Churchill Livingstone; 1995.
- Lavery LA, Sariaya M, Ashry H, Harkless LB. Microbiology of osteomyelitis in diabetic foot ulcers. J Foot Ankle Surg. 1995;34:61–4. DOIPubMedGoogle Scholar
- Isselbacher KJ, Braunwald E, Wilson JD, Martin JB, Fauci AS, Kasper DL, eds. Harrison's principles of internal medicine. 13th ed. New York: McGraw-Hill, Inc.; 1994.
- Fagon JY, Chastre J, Vuagnat A, Troillet JL, Novara A, Gibert C. Nosocomial pneumonia and mortality among patients in intensive care units. JAMA. 1996;275:866–9. DOIPubMedGoogle Scholar
- Papazian L, Bregeon F, Thirion X, Gregoire R, Saux P, Denis JP, Effect of ventilator-associated pneumonia on mortality and morbidity. Am J Respir Crit Care Med. 1996;154:91–7.PubMedGoogle Scholar
- Fagon JY, Chastre J, Hance AJ, Montravers P, Novara A, Gibert C. Nosocomial pneumonia in ventilated patients: a cohort study evaluating attributable mortality and hospital stay. Am J Med. 1993;94:281–8. DOIPubMedGoogle Scholar
- Leu HS, Kaiser DL, Mori M, Woolson RF, Wenzel RP. Hospital-acquired pneumonia: attributable mortality and morbidity. Am J Epidemiol. 1989;129:1258–67.PubMedGoogle Scholar
- Craven PC. Treating bone and joint infections with teicoplanin: hospitalization vs. outpatient cost issues. Hosp Formul. 1993;28:41–5.PubMedGoogle Scholar
- Allen R. Cost-effectiveness issues for home IV therapy in the United States. Hosp Formul. 1993;28:37–40.PubMedGoogle Scholar
- Arnow PM, Quimosing EM, Beach M. Consequences of intravascular catheter sepsis. Clin Infect Dis. 1993;16:778–84.PubMedGoogle Scholar
- Agency for Health Care Policy and Research. The HCUP-3 Nationwide Inpatient Sample (NIS), Release 2, 1993. Springfield (VA): National Technical Information Service; 1996.
- Bradley SF. Methicillin-resistant Staphylococcus aureus infection. Clin Geriatr Med. 1992;8:853–68.PubMedGoogle Scholar
- McEvoy GK, ed. American hospital formulary service drug information 1997. Bethesda (MD): American Society of Health-System Pharmacists; 1997.
- Spink WW. Staphylococcal infections and the problem of antibiotic-resistant staphylococci. Arch Intern Med. 1954;94:167–96.
- Allen JD, Roberts CE, Kirby WM. Staphylococcal septicemia treated with methicillin: report of twenty-two cases. N Engl J Med. 1962;266:111–6.PubMedGoogle Scholar
Tables
Cite This ArticleTable of Contents – Volume 5, Number 1—February 1999
| EID Search Options |
|---|
|
|
|
|
|
|