Volume 8, Number 10—October 2002
THEME ISSUE
Bioterrorism-related Anthrax
Bioterrorism-related Anthrax
Bacillus anthracis Aerosolization Associated with a Contaminated Mail Sorting Machine
Cite This Article
Citation for Media
Abstract
On October 12, 2001, two envelopes containing Bacillus anthracis spores passed through a sorting machine in a postal facility in Washington, D.C. When anthrax infection was identified in postal workers 9 days later, the facility was closed. To determine if exposure to airborne B. anthracis spores continued to occur, we performed air sampling around the contaminated sorter. One CFU of B. anthracis was isolated from 990 L of air sampled before the machine was activated. Six CFUs were isolated during machine activation and processing of clean dummy mail. These data indicate that an employee working near this machine might inhale approximately 30 B. anthracis-containing particles during an 8-h work shift. What risk this may have represented to postal workers is not known, but the risk is approximately 20-fold less than estimates of sub-5 micron B. anthracis-containing particles routinely inhaled by asymptomatic, unvaccinated workers in a goat-hair mill.
In the fall of 2001, 22 cases of anthrax were confirmed or suspected throughout the eastern United States as a result of bioterrorist release of spores (1). Ten cases (seven inhalational and three cutaneous) occurred in workers at postal facilities in which envelopes contaminated with Bacillus anthracis spores were processed by high-speed sorting machines. Two contaminated envelopes passed through a sorting machine at the United States Postal Service Processing and Distribution Center in Washington, D.C. (Brentwood mail facility), on the morning of October 12. The facility was closed on October 21 after anthrax infection was diagnosed; four employees were eventually confirmed as having inhalational anthrax (2). During the 9-day period while the facility continued to operate, >2,000 employees processed >60 million pieces of mail. In addition to the primary aerosol to which workers may have been exposed, they may have had continual reexposure to B. anthracis spores during this period.
At the time of the anthrax release in the fall of 2001, little was known about the re-aerosolization potential of B. anthracis spores after initial dispersion. Much of what was known came from studies conducted by the United States and Canadian military biological defense programs, using surrogate biological agents dispersed outdoors at very high concentrations (105–108 agent-containing particles/m2). These studies showed that re-aerosolization can occur, but risk is considered to be low (3,4). No information was available to answer similar questions about re-aerosolization risk in an indoor occupational setting such as a postal facility.
To address the question of continued risk for workers, we conducted an expanded safety evaluation of the partially remediated mail facility. A stamp on one of the two contaminated envelopes indicated that it had passed through Delivery Bar Code Sorter machine no. 17 at the Brentwood mail facility. This sorter, which had been idle for >2 weeks, had been cleaned with 0.5% hypochlorite solution before our testing. We evaluated the potential health risk to workers near that sorter by activating it and conducting surface and air sampling.
Surface Sampling
Two surface sampling techniques were used. Rodac plates (65-mm tryptic soy agar [TSA] plates; Becton-Dickinson, Franklin Lakes, NJ) were pressed onto the surface being sampled. Immediately adjacent to the Rodac sampling site, swab sampling was performed with sterile rayon-tipped swabs moistened with a 0.5-mL solution of phosphate-buffered saline + 0.05% Tween 20 (PBS Tween). An approximately 100-cm2 area was swabbed with sequential vertical, horizontal, and diagonal strokes. The swabs were individually placed in sterile, dry 15-mL conical tubes. Sampling focused on areas in the machine (electrical components, beneath belts, etc.) that were unlikely to have been cleaned with the topical bleach application.
Air Sampling
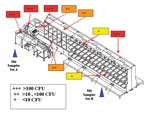
The ventilation system in the mail facility was turned off when the facility closed, and the system remained off during testing. We were unable to simulate the “blow-down” procedure (used to clean the sorter) during testing because the air compressors for the air hoses had lost power. Machine operators typically use high-pressure hoses several times a day to clean accumulated dust and debris between mail-sorting runs. Two banks of 10-slit samplers were placed on two postal trolleys (approximately 5 feet above the floor) and connected to a vacuum pump. The slit sampler intake ports were approximately 10 inches above the trolley. Each of the samplers was loaded with 150-mm TSA plates. Slit sampler set “A” (SSSA) was placed next to the operator’s station (at a location and height where workers would spend most of their time), and slit sampler set “B” (SSSB) was placed at the opposite end of the sorter (Figure 1). To measure the temporal patterns of re-aerosolization, the samplers operated sequentially; the intake port of slit sampler no.1 was opened, allowed to run for either 1 min (SSSA) or 2 min (SSSB), and then closed. Then slit sampler no.2 in the set was activated, and so on, until all 10 slit samplers in the set had been sequentially activated. The rate of air flow through each of the slit samplers was 33 L/min.
SSSB was activated and ran for 20 min while the sorter was turned off. SSSA was then activated and ran for 10 min. The plates were removed from all slit samplers and new plates were loaded. The SSSA and SSSB design characteristics determined the duration of sampling.
Both sets of slit samplers were activated simultaneously while the sorter was inactive. Approximately 1 min later, the sorter was started, and clean dummy mail was processed. After several false starts, continuous operation was achieved in approximately 2 min. The operation of the sorter was interrupted several times by jammed envelopes and quickly restarted each time, until the machine was turned off 1 min before the end of the 20-min sampling period. Postal officials reported that false starts, jamming, and restarting are common during routine operation of the machine.
As with the previous sampling, SSSA ran for 10 min and SSSB ran for 20 min. The plates were removed from the slit samplers and sealed in plastic bags. The bagged plates were taken out of the facility and the exteriors of the bags were decontaminated with 0.5% hypochlorite solution.
Mask Filters
The sampling team was outfitted with Canadian military C4 respirators with C7 canisters. The mask was equipped with 37-mm glass fiber collection filters mounted on the inlet port of the C7 canister, so that the entire inspirational volume of the investigators was sampled. The masked team members were located near the sorter to provide additional point sampling of respirable aerosol during the experiment. Team members were stationed at different work sites along the sorter and elsewhere in the facility to serve as point detectors. All mask filters were worn for at least 2 h.
Sample Handling and Processing
Environmental swabs and TSA plates from the Rodac plates and slit samplers were stored at 4°C until shipped and processed. All specimens were shipped at room temperature overnight to the Centers for Disease Control and Prevention. Swabs were placed in 1.5 mL PBS-Tween and vortexed for approximately 1 min. The solution was heat-shocked at 65°C for 30 min, and 100 µL was plated onto a sheep blood agar (SBA) plate. Rodac and slit sampler plates were incubated for approximately 12 h, and CFU were counted by visual inspection. All colonies suspected to be Bacillus spp. were subcultured on SBA plates. Identification and confirmatory testing of B. anthracis were done according to standard microbiologic procedures (5). The mask filters were removed in the facility and placed in sterile glass tubes. After transport, they were suspended in 3 mL of heart infusion broth and incubated at 35°C for 36 h, after which 10 µL of broth was plated onto SBA plates. The filters and remaining broth were heat-shocked at 65°C for 30 min, and 10 µL was plated onto SBA plates.
Statistical Analysis
Numbers of colonies detected in air sampling before and after the machine was activated were assessed with a one-sided one-sample test for difference in rates from a binomial distribution by using StatExact 4 v. 4.0.1. (Cytel Software Corp., Cambridge, MA).
Surface Sampling
Surface sampling was done by two methods, Rodac plates and premoistened swabs, to establish that the machine was still contaminated with viable B. anthracis spores. Ten Rodac plate samples and 10 swab samples were taken on the sorter surfaces. Both the Rodac plates and swabs yielded growth of CFUs that were too numerous to count at 7 of the 10 sites. Two additional Rodac plates were positive with low levels of contamination (1 and 3 CFUs); these locations were negative by the swab method (Figure 1).
Air Sampling
A single colony of B. anthracis was identified on one of the SSSA plates (Figure 2) during 10 min of sampling before the sorter was activated. No B. anthracis was identified on any of the SSSB plates during the 20 min of sampling.
After the sorter was activated, SSSA and SSSB ran for 10 min and 20 min, respectively. A single colony of B. anthracis was identified during minute 10 of sampling by SSSA (Figure 2; Table). SSSB identified a single colony of B. anthracis at each of minutes 1, 5, 7, 8, and 19.
Five investigators wore mask filters while the sorting machine was inactive; four investigators changed to new mask filters while the sorting machine was active. All mask filters were negative by culture.
New questions have arisen as public health authorities have investigated and responded to the intentional release of B. anthracis in the United States. Studies by Canadian investigators with a sophisticated preparation of Bacillus globigii have shown that a contaminated envelope may, even unopened, cause a substantial primary aerosol event (6). In light of this new appreciation, we investigated whether, after a remote contamination event and initial decontamination, a Delivery Bar Code Sorter machine could be a continual source of aerosolized B. anthracis spores and, if so, whether the particle concentration in the air could be estimated.
Initial reports indicated that no specific remediation had yet been undertaken on the contaminated machine. Subsequently, we learned that the surface of the sorter implicated in processing the contaminated envelopes had been cleaned with 0.5% hypochlorite solution. We proceeded with testing because the expectation was that topical cleaning would provide only fractional decontamination of a contaminated machine. By focused sampling, we found that, despite topical cleaning, the sorter remained contaminated with B. anthracis. By either swab technique or Rodac plates, 9 of 10 sites on the machine were positive and 4 sites produced B. anthracis colonies that were too numerous to count.
Air sampling detected B. anthracis before and after the sorter was activated. Before the sorter was turned on, the samplers detected a single B. anthracis-containing particle (0.0010 agent-containing particles per liter of air [ACPL]). Six colonies of B. anthracis (0.0061 ACPL) were identified in the 990 L of air sampled after sorter activation. The difference between the number of B. anthracis-containing particles detected by the samples collected as background and those collected after the sorter was activated was not significant at the 0.05 level (p=0.06); however, analysis suggests a trend toward a significant increase.
Environmental surface sampling done shortly after the Brentwood mail facility was closed found widespread contamination of the facility with B. anthracis (7). Both aerosolization of B. anthracis spores and direct cross-contamination of surfaces were considered likely mechanisms for contamination. Approximately 30 h of air sampling with open-faced 37-mm mixed cellulose ester filters (0.8-µm pore size) was negative. The previous report of negative air sampling despite extensive testing suggests our detection of airborne B. anthracis while the sorter was inactive may have been spurious and possibly related to investigator activities while the experiment was being set up.
Based on these concentrations and assuming 100% sampler collection efficiency, the estimated number of B. anthracis–containing particles that a worker might inhale near this activated sorter can be calculated. If we assume a normal ventilation rate (10 L/min), during 8 h working around this partially cleaned, but still contaminated sorter, a worker might be expected to inhale approximately 30 B. anthracis-containing particles. This finding of very low-level airborne B. anthracis contamination is supported by the negative testing of the mask filters. If all the airborne particles are assumed to be of optimal size for inhalation, this estimate is approximately 100-fold less than the lower boundary of the 50% lethal dose estimates for inhalational anthrax in nonhuman primate studies (8). This number is also approximately 20-fold less than estimates of the number of routinely inhaled B. anthracis–containing particles from a 1960 study of asymptomatic, unvaccinated workers in a goat-hair mill in Pennsylvania (9). In that study, investigators calculated that, in an 8-h workday, workers inhaled >1,300 viable B. anthracis-containing particles, 510 of which were <5 μm in size. Thus, although detected in the Brentwood facility, airborne contamination was at a relatively low level.
The comparison of this type of exposure with nonhuman primate anthrax data and historical industrial anthrax data is problematic for several reasons. Our understanding of human infection risk at very low-dose B. anthracis exposures is limited, as illustrated by the death from inhalational anthrax of an elderly Connecticut woman for whom no exposure could be determined, despite extensive environmental testing of her home and areas she frequented (1). A well-known contributor to the rate of alveolar deposition of a bioaerosol is the particle size distribution; because the slit sampling method does not measure the aerodynamic particle size distribution, we were unable to measure this attribute. Finally, historical comparisons to goat-hair mill workers are limited by the unknown contributions of prior host immunity, incomplete surveillance, and the lack of additional environmental sampling data other than the study from Pennsylvania.
This study shows that a mail sorter may remain contaminated, as indicated by surface sampling, many days after processing B. anthracis–contaminated letters and despite topical bleach cleaning. In addition, even after processing >1.2 million subsequent letters, as this sorter did, aerosolized B. anthracis–containing particles can still be detected around a contaminated sorter when active, at a level likely increased over background levels. At the time of our study, the level of B. anthracis–containing particles around this contaminated sorter at Brentwood was low, but any level of aerosolized B. anthracis spores is undesirable in this occupational setting.
Further studies are essential to define the risks of inhalational anthrax in the settings of both primary and secondary aerosolization of B. anthracis spores. In anticipation of potential future B. anthracis exposures, re-aerosolization potential should be evaluated in other environments, such as an office setting. In addition, size stratification of the re-aerosolized portion of a primary release should be part of any testing, to give some guidance as to risk stratification for exposed persons. Finally, better understanding of human health risk of low-dose exposure of B. anthracis spores is critical to guide optimal public health response.
Dr. Dull is an Epidemic Intelligence Service officer in the Meningitis and Special Pathogens Branch, National Center for Infectious Diseases, Centers for Disease Control and Prevention. His recent field epidemiology investigations include febrile illness surveillance in Nepal, meningitis outbreaks in Ethiopia and Montgomery County, Texas, and human cutaneous anthrax associated with an epizootic in North Dakota.
Acknowledgment
The authors acknowledge the extraordinary efforts of Timothy Haney and Louis Higginbotham, United States Postal Services Washington D.C. Processing and Distribution Center, in carrying out the investigation.
References
- Centers for Disease Control and Prevention. Update: investigation of bioterrorism-related anthrax—Connecticut, 2001. MMWR Morb Mortal Wkly Rep. 2001;50:1077–9.PubMedGoogle Scholar
- Centers for Disease Control and Prevention. Update: investigation of bioterrorism-related anthrax and interim guidelines for exposure management and antimicrobial therapy, October 2001. MMWR Morb Mortal Wkly Rep. 2001;50:909–18.PubMedGoogle Scholar
- Chinn KSK, Adams DJ, Carlon HR. Hazard assessment for suspension of agent-contaminated soil. Joint Operational Test & Information Directorate, U.S. Army Dugway Proving Ground. 1990. Publication DPG/JOD-91/002.
- Resnick IG, Martin DD, Larsen LD. Evaluation of the need for detection of surface biological agent contamination. Life Sciences Division, U.S. Army Dugway Proving Ground. 1990. Publication DPG-FR-90-711.
- Logan NA, Turnbull PCB. Bacillus and recently derived genera. In: Murray PR, Baron EJ, Pfaller MA, Tenover FC, Yolken RH, editors. Manual of clinical microbiology, vol. 7. Washington: American Society for Microbiology Press; 1999; p. 357–69
- Kournikakis B, Armour SJ, Boulet CA, Spence M, Barsons B. Risk assessment of anthrax threat letters, Suffield: DRES Technical Report TR 2001-048; 2001 September.
- Centers for Disease Control and Prevention. Evaluation of Bacillus anthracis contamination inside the Brentwood Processing and Distribution Center—District of Columbia, October, 2001. MMWR Morb Mortal Wkly Rep. 2001;50:1129–32.
- Inglesby TV, Henderson DA, Bartlett JG, Ascher MS, Eitzen E, Friedlander AM, Anthrax as a biological weapon: medical and public health management. JAMA. 1999;281:1735–45. DOIPubMedGoogle Scholar
- Dahlgren CM, Buchanan LM, Decker HM, Freed SW, Phillips CR, Brachman PS. Bacillus anthracis aerosols in goat hair processing mills. Am J Hyg. 1960;72:24–31.PubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 8, Number 10—October 2002
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Peter M. Dull, Emory University School of Medicine, Department of Medicine, Division of Infectious Diseases, 69 Butler St., S.E., Atlanta, GA 30303, USA; fax: 404-880-9305;
Top