Volume 20, Number 5—May 2014
Research
Bovine Leukemia Virus DNA in Human Breast Tissue
Figure 4
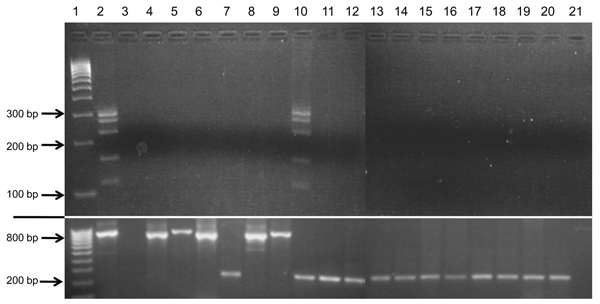
Figure 4. Test results showing lack of cross-reactivity of bovine leukemia virus (BLV)–specific primers with representatives of all mammalian and avian retrovirus subfamilies and human exogenous and endogenous viruses previously identified in human breast tissueNested liquid-phase PCR used primers from 5 BLV genome regions with template DNA from the viruses in lanes 4–10 and 12–21PCR products for each virus, loaded into 1 well, were separated by agarose gel (1.5%) electrophoresis on the basis of size differencesAmplicons were generated only for known BLV-positive cell lines (FLK and Bat2Cl6)Samples in lanes 13–21 were run simultaneously in the same gel in wells below samples in lanes 4–12The section below the white line shows glyceraldehyde 3-phosphate dehydrogenase (GAPDH) amplification of each sample to indicate DNA qualityHuman GAPDH primers were used for human, rhesus monkey, baboon, and bat cell lines (amplicon = 237 bp); murine GAPDH for mouse and rat cell lines (796 bp); and bovine GAPDH for bovine, ovine, and feline cell lines (857 bp)Lane 1, molecular weight marker (HyperLadder IV; Bioline, Taunton, MA, USA), lane 2, fetal lamb kidney cell line, positive control; lane 3, water substituted for DNA template, negative control; lane 4, Rous sarcoma virus; lane 5, murine sarcoma virus; lane 6, mouse mammary tumor virus; lane 7, Mason-Pfizer monkey virus; lane 8, murine leukemia virus; lane 9, feline leukemia virus; lane 10, BLV Bat2Cl6; lane 11, Tb1Lu (known BLV-negative cell line), negative control; lane 12, simian T-cell leukemia virus; lane 13, human T-cell leukemia virus 1; lane 14, human T-cell leukemia virus 2; lane 15, HIV-1; lane 16, HIV-2; lane 17, human papillomavirus 16; lane 18, human papillomavirus 18; lane 19, Epstein-Barr virus; lane 20, human endogenous retrovirus K; lane 21, env of human endogenous retrovirus K.
1Current affiliation: Texas A&M Health Science Center College of Medicine, College Station, Texas, USA.