Volume 10, Number 12—December 2004
Research
Nonsusceptibility of Primate Cells to Taura Syndrome Virus
Abstract
Taura syndrome virus (TSV), a pathogen of penaeid shrimp and member of the family Dicistroviridae, was recently reported to have the ability to infect primate cells. We independently retested this hypothesis. Three lines of primate cells FRhK-4, MA-104, and BGMK, which are highly susceptible to infection by human picornaviruses, were challenged with TSV. Viral replication was assayed by real time reverse transcription–polymerase chain reaction using cell media samples collected on days 0, 4, and 7 postchallenge. By day 7, genome copy numbers had decreased 25%–99%. No cytopathic effect was observed after 7 days. An in situ hybridization assay, with gene probes specific for detection of TSV, was negative for TSV in challenged cells. The infectivity of residual virus in the cell culture media at day 7 was confirmed by bioassay using TSV-free indicator shrimp (Litopenaeus vannamei). TSV did not infect the primate cells tested, and no evidence of zoonotic potential was found.
The general assumption is that the viral agents that cause disease in penaeid shrimp do not infect vertebrates. Supporting this assumption is the absence of documented cases of any shrimp virus causing disease in any animal species other than crustaceans. In a recent article, Taura syndrome virus (TSV), exclusively a pathogen of penaeid shrimp, was attributed a zoonotic potential because of its reported ability to infect cultured human and monkey cells (1). Aside from the food safety issues raised by this report, we were very interested in confirming those results because of the practical value of this new option for growing TSV in vitro. To date, TSV (or any other of the known viruses of the penaeid shrimp) has not yet been successfully cultured in any invertebrate or vertebrate cell-culture system. Hence, if viable, the use of primate cells for propagation of TSV would prove to be an important advancement in the study of TSV, and perhaps of other crustacean viruses. While the experiment reported in this study did not include all of the cell lines used by Audelo-del-Valle et al. (1), namely human rhabdomyosarcoma (RD), human larynx carcinoma (Hep-2), and Buffalo green monkey kidney (BGMK), the three cell lines that we used are also routinely used for virus isolation and diagnosis of diseases caused by human enteroviruses that belong to the family Picornaviridae (2–7). TSV, is a member of the family Dicistroviridae (closely related to Picornaviridae), genus Cripavirus (8,9). Other than TSV, which only infects penaeid shrimp, members of the genus Cripavirus are known to infect only insects (9). We report the results obtained after performing an experiment to test the hypothesis proposed by Audelo-del-Valle et al. (1).
Source of TSV
Rather than using TSV-infected shrimp originated from shrimp farms as a source, TSV infection was induced under laboratory conditions by injecting specific pathogen free (SPF) shrimp (10) Litopenaeus vannamei with purified TSV reference isolate Hawaii-94 (11). The use of SPF shrimp ensured that no contamination with other viral pathogens would interfere with the experiment. Hemolymph and hepatopancreas were obtained from moribund shrimp during the acute phase of Taura syndrome and used to prepare the viral inocula.
Preparation of Inocula
Hemolymph was drawn from a moribund shrimp with acute-phase Taura syndrome and the hepatopancreas was excised by using aseptic technique. The hemolymph was diluted 1:10 with Eagles Balanced Salts Minimum Essential Medium (EMEM), without fetal bovine sera (FBS), and filtered through a syringe filter of 0.22-μm pore size. The hepatopancreas was homogenized in 10 mL of EMEM without FBS, centrifuged at 125 x g for 2 min to eliminate coarse material and the supernatant filtered with a syringe filter of 0.22-μm pore size. Samples of hemolymph and hepatopancreas from health SPF shrimp were processed in identical manner and used as a negative control.
Cell Culture
The lines and cell cultures used were African green monkey kidney (BGMK), Monkey Rhesus female kidney embryonic (FRhK-4), and Monkey African green kidney (MA-104). Other than the report by Audelo-del-Valle et al. (1), TSV culture or CPE has not been reported in any invertebrate or vertebrate cell line. Hence, no positive control for TSV-induced CPE in cell culture was included in this study.
TSV Injection
Each of four 75-cm2 flasks of confluent monolayers of each cell line (BGMK, FRhK-4, and MA-104) was injected with 0.1 mL of either of the four inocula: 1) hemolymph from shrimp with acute-phase Taura syndrome, 2) hepatopancreas from shrimp with acute-phase Taura syndrome, 3) hemolymph from SPF shrimp, or 4) hepatopancreas from SPF shrimp. After injection, the standard volume (15 mL) of fresh EMEM with 2% FBS was added without removing the inoculum. The cells were incubated at 37°C and monitored once a day for 7 days for cytopathic effect (CPE). As an additional negative control, one flask of each of the three cell lines was left untreated but monitored once a day alongside the TSV-injected flasks.
As an additional test to determine if a productive TSV infection occurred, representative samples of cells at day 7 were collected with a sterile pipette and pelleted at 130 x g. The pellet of cells was fixed in Davidson’s AFA (alcohol, formaldehyde, and acetic acid) fixative and processed by using conventional techniques for paraffin embedding and sectioning. Paraffin sections were subjected to in situ hybridization with a mixture of two gene probes, P15 and Q1, specific for detection of TSV (12), according to protocols published elsewhere (13,14).
TSV Quantification
RNA Extraction
A total of 0.2 mL from each of the original inocula (inocula prepared from the hemolymph and hepatopancreas of infected and noninfected shrimp) and 0.2 mL of cell culture media from each of the three different cell line cultures collected at days 0, 4, and 7 postexposure, were subjected to RNA extractions using a High Pure RNA tissue kit (Roche Molecular Biochemicals, Indianapolis, IN), according to the manufacturer’s recommendations. The concentration of extracted RNA was estimated by measuring optical density, OD260nm, with an Eppendorf spectrophotometer.
Real-Time TSV RT-PCR
The real time TSV RT-PCR assays were performed using an ABI GeneAmp 5700 with TaqMan One-Step RT-PCR master mixture (Applied Biosystems, Foster City, CA). The reaction mixture contained no more than 10 ng of extracted RNA, with each primer at a concentration of 0.3 μmol/L, and the TaqMan probe at a concentration of 0.1 μmol/L in a final volume of 25 μL. The cycling consisted of 30 min at 48°C for reverse transcription and 10 min at 95°C, followed by 40 cycles of 95°C for 15 s, and 60°C for 1 min. The data acquisition and analysis were carried out with GeneAmp 5700 Sequence Detector Software (Applied Biosystems). The real-time RT-PCR primers and TaqMan probe for the detection of TSV had been previously designed from ORF1 region of the TSV genomic sequence (15). Serial dilutions from a previously constructed TSV plasmid were used to determine a standard linear relationship for quantification with a correlation of the serial dilutions >0.99.
Confirmation of TSV Infectivity
Bioassay
To confirm the infectivity of the virus, a 6-day bioassay was performed by injecting groups of four to six SPF indicator shrimp (L. vannamei) with approximately 200 μL of either of the following: 1) inoculum prepared from hemolymph of infected shrimp; 2) inoculum prepared from hemolymph of noninfected shrimp; 3) cell media collected at day 7 from TSV-challenged cell culture flasks; or 4) culture media collected at day 7 from SPF shrimp tissue–treated cell culture flasks (negative control). The shrimp were monitored once a day for signs of disease. Moribund shrimp were collected when observed, preserved in Davidson’s fixative and transferred into 70% ethanol after 24 h (14,16). All surviving shrimp at termination of the bioassay (day 6) were preserved in the same manner. Shrimp tissue samples were processed according to conventional techniques for paraffin embedding and sectioning (16). Paraffin sections were stained with Mayer-Bennett’s hematoxylin/eosin-phloxin and subjected to histologic evaluation to determine the presence of TSV diagnostic lesions. Selected specimens were subjected to a confirmatory assay by in situ hybridization (ISH) with a mixture of two gene probes, P15 and Q1, specific for detection of TSV (12–14).
Cytopathic Effect (CPE)
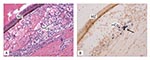
No CPE was observed in any of the three cell lines injected with TSV infected hemolymph, TSV infected hepatopancreas, SPF shrimp hemolymph, or SPF shrimp hepatopancreas (Figure 1). The BGMK cell line showed normal fibroblastic structure throughout the 7-day period of exposure to the different treatments. The BGMK monolayer remained confluent with no evidence of cell detachment or lysis. Similarly, the FRhK-4 and the MA-104 cell lines retained their typical epithelial structure for the 7-day period after exposure to TSV, with confluent monolayers and no evidence of cell detachment or lysis.
Virus Quantification
Approximately 1.3 x 105 to 2.7x106 viral copies/μL were detected at day 0 in the tissue cell flasks exposed to TSV-infected hemolymph. In the case of the tissue cell flasks exposed to TSV-infected hepatopancreas, ≈ 1.2 x 104 viral copies/μL were detected (Table). TSV was not detected by real time RT-PCR in the inoculum prepared from SPF shrimp hemolymph and hepatopancreas, nor in the tissue cell culture flasks exposed to these inocula.
At day 7, real time RT-PCR showed a decrease of 25% to 99% of the TSV genome copy number in the tissue cell culture flasks exposed to TSV (Figure 2), which suggests that no viral replication had occurred but that some residual virus remained from the inoculum.
Bioassay
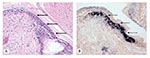
Samples of cell-culture media from tissue culture flasks injected with TSV-infected hemolymph were collected at day 7 and used to inject SPF indicator shrimp L. vannamei to determine the infectivity of the residual virus. Moribund shrimp from these bioassays were examined by conventional hematoxylin/eosin-phloxin histology and by in situ hybridization with the gene probes specific for detection of TSV. TSV infection was diagnosed in all of the moribund shrimp, which indicates that at 7 days after injection, the tissue culture media contained sufficient residual TSV to produce infections in challenged shrimp (Figure 3). Paraffin sections from known TSV-infected and noninfected shrimp were used as ISH positive and negative controls, respectively (results not shown).
During this study, a relationship was observed between the concentration of TSV in the inocula prepared from day 7 tissue culture media (from cells exposed to TSV-infected hemolymph) and the severity of TSV infection in the challenged SPF indicator shrimp. The shrimp that had been injected with tissue cell culture media with the highest TSV concentration (≈ 2.5 x 104 viral copies/μL) developed an acute (overt) infection within 3 days post-challenge, whereas shrimp injected with tissue cell culture media with the lowest viral concentration (≈ 2.3 x 103 viral copies/μL) developed only a subacute (covert) infection. Both overt and covert infections were confirmed by histologic analysis and by ISH with gene probes specific for detection of TSV (Figure 4).
Additional ISH Test.
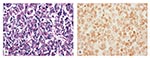
As an additional test to further confirm the absence of viral replication, the accumulation within the cells, or both, a sample of cells at day 7 was obtained from the BGMK cell line and subjected to ISH with TSV-specific gene probes. The BGMK cell line was selected for this assay because, among all three lines, it had the highest initial (day 0) concentration of viral particles and the lowest final concentration at day 7, suggesting either degradation or internalization of the virus. The ISH assay gave negative results (Figure 5), which indicates degradation as the more likely explanation for the reduction in virus content of the cell media. As in previous ISH assays, paraffin sections from known TSV-infected and noninfected shrimp were used as ISH-positive and -negative controls, respectively (results not shown).
The real time RT-PCR results (Table) show that the number of TSV genome copies did in the cell culture media did not increase for any of the three cell lines challenged with TSV. While differences were observed in the estimated number of viral copies in each flask at day 7, the number of viral copies present was from one to two logs less than that of the day 0 values, which were determined immediately after cells were injected. The apparent plateau of TSV counts at day 7, regardless of the concentration of viral particles in each flask at day 0, may have been due to a protective effect of the cell-culture media, specifically the fetal bovine serum (FBS). This protective effect of FBS on viruses has been documented by other researchers (17–20). Studies on viral transport media for the preservation of virus viability have concluded that the best transport media for specimens at risk of being delayed by long transit times and exposed to significant temperature variations en route, are those that contain 2% FBS (i.e., CVM-Copan Diagnostics, Corana, CA and M4-Multi-Microbe, Micro Test, Inc., Snellville, GA).
To further confirm the absence of viral replication or accumulation within the cells, a sample of cells at day 7 was obtained from the BGMK cell line and subjected to ISH with TSV-specific gene probes. The absence of reaction to the TSV probes and the absence of CPE indicate that viral replication did not occur in the BGMK cells and that if any viral particles were internalized, they were degraded.
The relationship observed between the concentration of TSV in the inocula prepared from day 7 tissue culture media (from cells exposed to TSV-infected hemolymph) and the severity of TSV infection in the challenged SPF indicator shrimp agree with the results obtained during a previous study in the authors’ laboratory, in which it was concluded that a minimum concentration of ≈ 1.0 x 104 viral copies/μL is necessary to induce an acute infection (21). In this case, those shrimp that had been injected with tissue cell culture media with the highest TSV concentration (≈ 2.5 x 104 viral copies/μL) developed an acute (overt) infection within 3 days postchallenge, whereas shrimp injected with tissue cell culture media with the lowest viral concentration (≈ 2.3 x 103 viral copies/μL) developed only a subacute (covert) infection.
BGMK, FRhK-4, and MA-104 cell lines are often used for isolation and research purposes for enteroviral meningitis (3), hepatitis A virus (7), polioviruses, coxsackie A, and coxsackie B (22) because of their marked susceptibility to infection by these members of the Picornaviridae. When exposed to any of these agents, these cell lines develop conspicuous CPE within ≈ 5 days (3,7,22,23). However, no CPE in any of the three cell lines (BGMK, FRhK-4, and MA-104) challenged with TSV was observed in this experiment, even at day 7 postinjection. These results contradict those of Audelo-del-Valle et al. (1), who reported the development of CPE within 19–23 hours. The average incubation time required for CPE development (induced by enteroviruses) in human or monkey cells at 37°C is 5 days, although detection time may be reduced to ≈ 3 days by use of the shell vial method (23). Shorter incubation times of <24 hours for CPE development could be more suggestive of a toxicity problem rather than of virus induced CPE.
As mentioned above, SPF shrimp (L. vannamei) were used to amplify a reference strain of TSV to prepare the inocula. We used SPF shrimp for three reasons. First, pond-reared or wild shrimp may harbor human or other mammalian picornaviruses. Shrimp and other decapod crustaceans have been shown to internalize and passively carry certain fish viruses (24–26) and human enteroviruses (27, C. Gerba, unpub. data). Hence, wild or pond-reared shrimp may be passive carriers of human or other mammalian pricornaviruses or other viruses which could produce CPE in studies such as that reported by Audelo-del-Valle et al. (1). Second, by using a commercially available line of SPF shrimp, the experiment can be standardized; therefore, other researchers can repeat or confirm the present study. Third, the SPF shrimp used are produced in closed biosecure systems with controlled water sources, which preclude chance contamination of the stocks with human or other animal viruses.
BGMK cells were the only cell type in common between our study and that of Audelo-del-Valle et al (1), who also used RD and Hep-2 however, BGMK, FRhK-4, and MA-104 cells were selected for use in our study because these cell types have a marked susceptibility to infection by members of Picornaviridae (28), which makes them as adequate as RD or Hep-2 cells for determining the possible infectivity of TSV to primate cells. We conclude that TSV did not infect the primate cells challenged with TSV in our study.
The lack of CPE in any of the three different cell lines tested, the negative ISH results with TSV specific gene probes assay of TSV challenged BGMK cells, and multilog reduction in TSV number in the cell-culture media as determined by real time RT-PCR indicate that TSV did not replicate in the primate cell lines used in our study. That TSV infection had occurred in SPF indicator shrimp 7 days after injection with media collected from cell culture flasks indicates that sufficient residual TSV remained in the media to infect the challenged shrimp and to cause acute disease or subacute disease as a function of relative concentration of residual TSV present.
Dr. Pantoja is associate research professor at the Aquaculture Pathology Laboratory, University of Arizona (an OIE reference laboratory for Taura syndrome and other diseases of aquatic organisms), where he forms part of a team that provides disease diagnostic services to the international shrimp farming industry and does research on new and emerging diseases of shrimp.
Acknowledgment
Funding for this research was provided by the USDA CSREES Marine Shrimp Farming Program, grant number 2002-38808-01345, and a special grant from the National Fishery Institute.
References
- Audelo-del-Valle J, Clement-Mellado O, Magaña-Hernandez A, Flisser A, Montiel-Aguirre F, Briseño-Garcia B. Infection of cultured human and monkey cell lines with extract of penaeid shrimp infected with Taura syndrome virus. Emerg Infect Dis. 2003;9:265–6.PubMedGoogle Scholar
- Agbalika F, Hatermann P, Foliguet JM. Trypsin-treated Ma-104: a sensitive cell line for isolating enteric viruses from environmental samples. Appl Environ Microbiol. 1984;47:378–80.PubMedGoogle Scholar
- Buck GE, Wiesemann M, Stewart L. Comparison of mixed cell culture containing genetically engineered BGMK and CaCo-2 cells (Suer E-Mix) with RT-PCR and conventional cell culture for the diagnosis of enterovirus meningitis. J Clin Virol. 2002;25:S13–8. DOIPubMedGoogle Scholar
- Kok TW, Pryor T, Payne L. Comparison of rhabdomyosarcoma, Buffalo green money kidney epithelial, A549 (human lung epithelial) cells and human embryonic lung fibroblasts for isolation of enteroviruses from clinical samples. J Clin Virol. 1998;11:61–5. DOIPubMedGoogle Scholar
- Landry M, Garner R. Ferguson. Rapid enterovirus RNA detection in clinical specimens by using nucleic acid sequence based amplification. J Clin Microbiol. 2003;41:346–50. DOIPubMedGoogle Scholar
- Otero JR, Folgueira L, Trallero G, Prieto C, Maldonado S, Babiano MJ, A-549 is a suitable cell line for primary isolation of coxsackie B viruses. J Med Virol. 2001;65:534–6. DOIPubMedGoogle Scholar
- Sanchez G, Pinto RM, Vanaclocha H, Bosch A. Molecular characterization of hepatitis A virus isolates from a transcontinental shellfish-borne outbreak. J Clin Microbiol. 2002;40:4148–55. DOIPubMedGoogle Scholar
- Mayo MA. ICTV at the Paris ICV: results of the plenary session and binomial ballot. Arch Virol. 2002;147:1655–63. DOIPubMedGoogle Scholar
- Mari J, Poulos BT, Lightner DV, Bonami J-R. Shrimp Taura syndrome virus: genomic characterization and similarity with members of the genus Cricket paralysis-like viruses. J Gen Virol. 2002;83:915–26.PubMedGoogle Scholar
- Wyban JA, Swingle JS, Sweeney JN, Pruder GD. Development and commercial performance of high health shrimp using specific pathogen free (SPF) broodstock Penaeus vannamei. In: Wyban J, editor. Proceedings of the Special Session on Shrimp Farming. Baton Rouge (LA): World Aquaculture Society; 1992. p. 254–9.
- Nunan LM, Poulos BT, Lightner DV. Reverse transcription polymerase chain reaction (RT-PCR) used for the detection of Taura syndrome virus in experimentally infected shrimp. Dis Aquat Organ. 1998;34:87–91. DOIPubMedGoogle Scholar
- Mari J, Bonami J-R, Lightner DV. Taura syndrome of penaeid shrimp: cloning of viral genome fragments and development of specific gene probes. Dis Aquat Organ. 1998;33:11–7. DOIPubMedGoogle Scholar
- Hasson KW, Lightner DV, Mohney LM, Redman RM, Poulos BT, Mari J, The geographic distribution of Taura syndrome virus (TSV) in the Americas: determination by histopathology and in situ hybridization using TSV-specific cDNA probes. Aquaculture. 1999;17:13–26. DOIGoogle Scholar
- Lightner DV. A handbook of shrimp pathology and diagnostic procedures for diseases of cultured penaeid shrimp. Baton Rouge (LA): World Aquaculture Society; 1996.
- Tang KT, Lightner DV. Quantitation of Taura syndrome virus by real-time RT-PCR with TaqMan assay. J Virol Methods. 2004;115:109–14. DOIPubMedGoogle Scholar
- A handbook of normal penaeid shrimp histology. Baton Rouge (LA): World Aquaculture Society; 1998.
- Jensen C, Johnson FB. Comparison of various transport media for viability maintenance of herpes simplex virus, respiratory syncytial virus and adenovirus. Diagn Microbiol Infect Dis. 1994;19:137–42. DOIPubMedGoogle Scholar
- Josephson SL. An update on the collection and transport of specimens for viral culture. Clin Microbiol Newsl. 1997;▪▪▪:57–64. DOIGoogle Scholar
- Dunn JJ, Billetdeaux E, Skodack-Jones L, Cfarroll KC. Evaluation of three Copan viral transport systems for the recovery of cultivable, clinical virus isolates. Diagn Microbiol Infect Dis. 2003;45:191–7. DOIPubMedGoogle Scholar
- Poulos BT, Lightner DV. Analysis of samples from Litopenaeus vannamei during chronic phase Taura syndromevirus (TSV) infection. In: Book of abstracts, Aquaculture America 2003, new frontiers in aquaculture. Baton Rouge (LA): World Aquaculture Society; 2003. p. 237.
- Weng KT, Pryor T, Payne L. Comparison of rhabdomyosarcoma, Buffalo green monkey kidney epithelial, A549 (human lung epithelial) cells and human embryonic lung fibroblasts for isolation of enteroviruses from clinical samples. J Clin Virol. 1998;11:61–5. DOIPubMedGoogle Scholar
- Huang YT, Yam P, Yan H, Sun Y. Engineered BGMK cells for sensitive and rapid detection of enteroviruses. J Clin Microbiol. 2002;40:366–71. DOIPubMedGoogle Scholar
- Bovo G, Ceschia G, Giorgetti G, Vanelli M. Isolation of an IPN-like virus from adult Kuruma shrimp (Penaeus japonicus). Bull Eur Assoc Fish Pathol. 1984;4:21.
- Halder M, Ahne W. Freshwater crayfish Astacus astacus—a vector for infectious pancreatic necrosis virus (IPNV). Dis Aquat Organ. 1988;4:205–9. DOIGoogle Scholar
- Lu Y, Cesar E, Nadala CB, Brock JA, Loh PC. A new virus isolate from infectious hypodermal and hematopoietic necrosis virus (IHHNV)-infected penaeid shrimps. J Virol Methods. 1991;31:189–96. DOIPubMedGoogle Scholar
- Hejkal TW, Gerba CHP. Uptake and survival of enteric viruses in the Blue crab, Callinectes sapidus. Appl Environ Microbiol. 1981;41:207–11.PubMedGoogle Scholar
- Sair AI, D’Souza DH, Jaykus LA. Human enteric viruses as causes of foodborne disease. Comprehensive Reviews in Food Science and Food Safety. 2002;1:73–89. DOIGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 10, Number 12—December 2004
| EID Search Options |
|---|
|
|
|
|
|
|


Please use the form below to submit correspondence to the authors or contact them at the following address:
C.R. Pantoja, University of Arizona, OIE Reference Laboratory for Taura Syndrome, 117 E. Lowell St., Building 90, Room 114, Tucson, Arizona 85721, USA; fax: 520-621-4899
Top