Volume 11, Number 11—November 2005
Research
Cryptococcus gattii in AIDS Patients, Southern California
Abstract
Cryptococcus isolates from AIDS patients in southern California were characterized by molecular analyses. Pheromone MFα1 and MFa1 gene fragments were polymerase chain reaction–amplified with fluorescently labeled primers and analyzed by capillary electrophoresis (CE) on DNA analyzer. CE–fragment-length analyses (CE-FLAs) and CE–single-strand conformation polymorphisms (CE-SSCPs) were used to determine Cryptococcus gattii (Cg), C. neoformans (Cn) varieties neoformans (CnVN) and grubii (CnVG), mating types, and hybrids. Corroborative tests carried out in parallel included growth on specialized media and serotyping with a commercial kit. All 276 clinical strains tested as haploid MATα by CE-FLA. CE-SSCP analyses of MFα1 showed 219 (79.3%) CnVG, 23 (8.3%) CnVN, and 34 (12.3%) Cg isolates. CE-FLA and CE-SSCP are promising tools for high-throughput screening of Cryptococcus isolates. The high prevalence of Cg was noteworthy, in view of its sporadic reports from AIDS patients in North America and its recent emergence as a primary pathogen on Vancouver Island, Canada.
The encapsulated basidiomycete Cryptococcus neoformans was recently divided into 2 species, C. neoformans (Cn) and C. gattii (Cg) (1). Cn consists of 2 varieties, grubii (CnVG) and neoformans (CnVN), which are opportunistic pathogens and predominantly infect immunocompromised persons (2,3). CnVG is the major causative agent of cryptococcosis worldwide, except in central Europe, where CnVN infection is most prominent. In contrast, Cg is a primary pathogen, which predominantly infects immunocompetent persons (4). Cg was previously thought to be restricted to tropical and subtropical climates with a special ecologic niche on Eucalyptus trees (5,6). However, the recent outbreak of Cg infection in healthy humans and animals in the temperate climate of Vancouver Island, British Columbia, Canada, and its isolation from several species of trees other than Eucalyptus have raised the strong possibility that this fungus might have broader geographic distribution (7–9).
The mechanisms underlying pathogenic and environmental differences between Cn and Cg are not known. Within Cn species, CnVN infections are more likely to display skin involvement and to afflict older patients, whereas CnVG infections are reported to cause a higher mortality rate (3,10). In contrast, infections caused by Cg result in a lower mortality rate but are frequently complicated by neurologic sequelae and require surgery and prolonged therapy (4,11). Our recent studies with the Cu,Zn SOD (SOD1) and MnSOD (SOD2) knockout mutants of Cg indicated that these antioxidants are crucial for Cg pathogenesis (12,13). In contrast, the antioxidant function of SOD1 in CnVG is less crucial for pathogenesis (14). These observations are the first molecular evidence of a likely divergence in the pathogenic mechanisms used by Cn and Cg.
Both Cn and Cg have a single locus, 2-allele mating system comprising MATα and MATa strains. The MATα strains of Cn and Cg predominate in nature and in clinical settings, and this predominance over MATa strains is linked to high virulence and reproduction by haploid fruiting (3,15). Generally, Cryptococcus strains are haploid, but hybrid strains have also been characterized from both clinical and environmental sources (16–20). Thus, characterizing clinical Cryptococcus isolates to the individual species or varieties and according to mating and hybrid types could be useful for managing cryptococcosis cases and for further understanding the epidemiology of this disease.
Several laboratory typing methods have been used in epidemiologic studies of cryptococcosis, including serotyping, electrophoretic karyotyping, use of mitochondrial DNA probes, use of genomic DNA probes, determination of allelic variations at the URA5 locus, multilocus enzyme typing, measurement of creatinine utilization, polymerase chain reaction (PCR), fingerprinting and amplified fragment-length polymorphism (reviewed in [2]). Previously, we described a PCR–restriction fragment length polymorphism (PCR-RFLP) typing scheme for Cn and Cg pheromone genes, which could be used for characterizing mating types, hybrids, and variety (21). In the present study, we developed a capillary electrophoresis–fragment length analysis (CE-FLA) test, and a CE–single stranded conformation polymorphism (CE-SSCP) test by using the pheromone genes MFα1 and MFa1. These tests were used in parallel with more traditional specialized culture medium and a commercial serotyping kit to characterize Cryptococcus isolates from AIDS patients in southern California.
Cryptococcus Isolates
Two hundred seventy-six Cryptococcus isolates originating from patients with HIV/AIDS were obtained from the Infectious Diseases Laboratory, Los Angeles County Hospital, Los Angeles, California. The isolates were suspended in sterile skim milk and stored at –20°C. The isolates were transferred frozen to the Mycology Laboratory of the Wadsworth Center in Albany, New York, USA, where they were streaked on Niger seed agar plates (3) to check for purity and reconfirmation of their identity; a typical colony was picked for further analysis. The subcultures were placed in long-term storage in sterile 15% glycerol at –70°C. These isolates were further characterized in our laboratory by testing their growth on canavanine-glycine-bromothymol blue (CGB) agar for differentiation of Cryptococcus species (22) and serotyping with Crypto Check Kit (Iatron Laboratories Inc., Tokyo, Japan). Several investigators gave strains to put together a panel of reference isolates that were either currently being used in molecular pathogenesis studies, represented type strains, or were otherwise unique. The details of these 16 reference isolates are listed in Table 1. Six additional A/D hybrid strains, characterized in our earlier study, were also used (18).
Multiplex PCR for Pheromone Genes
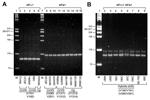
A previous report from this laboratory described the use of specific primers for amplification of MFα1 and MFa1 gene fragments, which could be separated as 100-bp and 117-bp fragments on a specialized agarose gel (21). The primer sets V290/V291, which was earlier designed to amplify MFa1 gene from CnVN, did not amplify similar genes from MATa strains of either CnVG or Cg. Multiple alignment of MFa1 indicated that this gene is highly polymorphic among CnVG, CnVN, and Cg (Figure 1). Therefore, 2 new sets of primers were designed to obtain MFa1 amplicons from CnVG and Cg. These primers are listed in Table 2. A multiplex PCR for simultaneous amplification of MFα1 and MFa1 in a 50-μL reaction volume was performed with 5 μL of 10× PCR buffer with 15 mmol/L MgCl2, 2.5 μL of each of 8 primers (10 μmol/L stock), 3.0 μL dNTP mix (10 μmol/L each), and 2.0 U Taq DNA polymerase (Perkin Elmer, Foster City, CA, USA). The template DNA was 5.0 μL of either a boiled cell suspension or 50 ng genomic DNA. Initial denaturation was conducted at 95°C for 3 min, followed by 30 cycles of denaturation at 94°C for 1 min, annealing at 57.5°C for 1 min, amplification at 72°C for 1 min, and final extension at 72°C for 7 min, in a GeneAmp PCR System 9600 (Perkin Elmer). In preliminary experiments, PCR products (10-μL aliquots) were resolved by electrophoresis on 3.5% MetaPhor agarose (FMC Bio-Products, Rockland, ME, USA) gels in Tris-borate-EDTA (TBE) buffer, and were detected by ethidium bromide staining. The PCR experiments were repeated twice, and identical results were obtained.
Gene Scan Analysis
The MFα1 sense primer (V190) was labeled with FAM (6-carboxyfluorescein) at the 5´ end, the antisense primer (V191) was labeled with tetrachloro-fluorescein (TET) at the 3´ end, and MFa1 sense primers (V290, V1311, V1313) were labeled with 6-carboxy-2´, 4´, 4´, 5´, 7´, 7´-hexachlorofluorescein (HEX) at the 5´ end. The fluorescent dye–labeled primers were custom ordered (Operon Technologies, Inc., Alameda, CA, USA). FLA and SSCP of the MFα1 and MFa1 PCR amplicons were determined by CE with an ABI PRISM 310 Genetic Analyzer, and the electronic images were analyzed by using GeneScan analysis software (Applied Biosystems Inc., Foster City, CA, USA). The sample preparation for CE consisted of 1 μL MFα1 and MFa1 PCR amplicons, 12 μL highly deionized formamide, and 0.5 μL GeneScan-500 (TAMRA) size standard (Applied Biosystems). The sample mixture was denatured for 5 min at 95°C and was then rapidly cooled on ice before loading on the instrument. For CE-FLA, the samples were analyzed under denaturing conditions (POP-4 polymer [Applied Biosystems] in buffer supplied by manufacturer) at 60°C, and for CE-SSCP, the samples were analyzed under nondenaturing conditions (3% GeneScan polymer in 1× TBE buffer with 10% glycerol) at 30°C. A capillary (47 cm × 50 μm inside diameter) was installed, and POP-4 or 3% polymer was filled according to manufacturer's instructions. The electrophoresis conditions for CE-FLA were 5-s injection time, 15-kV injection voltage, 15-kV electrophoresis voltage, 150-s syringe pump time, 120-s preinjection electrophoresis, and 20-min collection time for each sample, and the run was performed at 60°C. The electrophoresis conditions for CE-SSCP were 5-s injection time, 15-kV injection voltage, 13-kV electrophoresis voltage, 30-s syringe pump time with no preinjection time, and 20-min collection time for each sample, and the run was performed at 30°C. CE-FLA and CE-SSCP standardization experiments were carried out on >4 independent occasions, and unknown sample analyses were repeated at least once.
Multiplex PCR
The 4 sets of primers (MFα1/MFa1) produced reproducible results for control CnVG, CnVN, Cg haploids (Figure 2A), and A/D hybrid strains (Figure 2B). These results validated the robustness of the primers, which had been designed from well within the open reading frames of 2 pheromone genes, to prevent amplification of any nontarget DNA. The latter objective also informed the decision to anchor the 3´ ends of all PCR primers within the characteristic Cys-Val-Ile-Ala (CVIA) motifs; this eliminated any possible amplification of other pheromone genes since this is the only sequence shared among fungal pheromones (18). Even though multiple copies of MFα and MFa genes have been reported in C. gattii and C. neoformans by Southern hybridization and whole genome-sequencing, PCR primers only amplify single amplicons because these genes have identical nucleotide sequences (18,23,24). Although this multiplex method was well suited for identifying mating types and hybrids, further delineation of species and varieties would require restriction digestion with several unique enzymes as we stated previously (21). Therefore, we decided to use CE-FLA and CE-SSCP to further characterize pheromone gene amplicons. These techniques have been successfully used to delineate fragment length as well gene mutations for characterizing various fungal and bacterial isolates (25–27). The SSCP analysis displays migration of the amplified DNA fragment as a function of that fragment's structural conformation. Given that the tertiary structure of a fragment is sensitive to single nucleotide substitutions, this method was shown to be suitable for detecting single nucleotide changes when 100-bp to 300-bp DNA fragments were analyzed (28). Since amplified pheromone fragments yield ≈100- to 120-bp products, they were an ideal substrate for this method of mutant detection.
CE-FLA
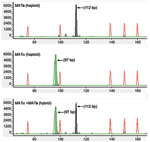
The 16 reference strains of known Cryptococcus species, varieties, mating types, and hybrids were used to establish a robust CE-FLA protocol with denaturing POP-4 polymer at 60°C. The electrophoretic runs with POP-4 polymer produced a 112-bp DNA fragment for MFa1 and 97-bp fragment for MFα1, which were easily distinguished with the GeneScan software by the characteristic peak sizes (Figure 3). CE-FLA allowed Cryptococcus mating types and hybrids to be identified, but not CnVG, CnVN, and Cg.
CE-SSCP
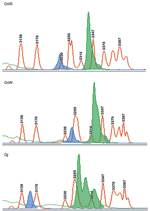
CE-SSCP test under nondenaturing conditions with 3% GeneScan polymer at 30°C allowed characteristic peak patterns to be detected in MFα1 and MFa1 genes because of the individual differences within the nucleotide sequences. Distinct patterns obtained for CnVG, CnVN, and Cg by using MFα1 gene are shown in Figure 4. The sense strand (labeled blue) yielded 1 characteristic peak pattern, while the antisense strand (labeled green) yielded 2–3 characteristic peak patterns. We subsequently decided to label only the sense strand to reduce the cost of the PCR primers as well the complexity of the peak patterns observed with antisense strand. For determining an unknown sample, the instrument analyses needed to yield highly reproducible results. Therefore, a sample of each of the Cryptococcus strains was injected on 4 separate occasions into the same capillary, and the precision of the sizing was calculated. The low standard deviations associated with each mean peak value indicated that the assignment of variety or species for an unknown sample, based on pattern sizing information alone, would be highly reliable (Table 3).
Based on our success with MFα1 sense primer in the detection of characteristic peaks for CnVG, CnVN, and Cg, we labeled MFa1 sense strands and analyzed MATa strains. In this case, we had to use individual sets of MFa1 primers because of the substantial polymorphism observed at the 5´ and 3´ end of this gene between Cg and Cn, and within Cn vatieties (Figure 1). Again, the MFa1 peak pattern was unique to each Cn variety and Cg (Table 3). Overall, our results indicated that either MFα1 or MFa1 gene products yielded unique SSCP patterns and could be used for identifying Cn varieties and Cg strains.
California Isolates
We used standardized CE-FLA and CE-SSCP techniques to analyze 276 isolates of Cryptococcus that were obtained from AIDS patients and that were stored at the Infectious Diseases Laboratory, Los Angeles County Hospital, Los Angeles, California. The investigations were fully compliant with Los Angeles County Hospital–University of Southern California (USC) Institutional Review Board guidelines (proposal #924008). CE-FLA showed that all 276 isolates were MATα strains, and no MATa or hybrid strains were found in our samples. CE-SSCP found that among the total 276 clinical isolates, 219 (79.3%) were CnVG, 23 (8.3%) were CnVN, and 34 (12.3%) were Cg. For corroborations, all of these isolates were also tested by growth on CGB agar, and by serotyping with the Crypto Check Kit, both of which yielded results in agreement with those obtained with pheromone typing.
The relatively high prevalence of Cg in this survey is noteworthy for several reasons (Table 4). First, we believe it is the first instance in which a large number of Cg clinical isolates from AIDS patients have been identified in the United States. Second, Cg has never been considered a substantial cause of cryptococcosis among US AIDS patients, including those in the southern California. Third, the presence of Cg in HIV-AIDS patient samples in the USC collection is similar to the prevalence recently reported from some countries in Central and South America (29), and it contrasts with the rare occurrence of Cg in immunocompromised patient populations in Australia, Southeast Asia, and Africa (30–32). The prevalence of CnVN (8%) in our samples closely matches its recently reported prevalence in New York City (33). Thus, cryptococcosis due to CnVN in AIDS patients is not a rare clinical entity in the United States, and its pattern of distribution on the East and West Coasts does not differ. The absence of MATa strains in our samples is not surprising, in view of the rare occurrence of this mating type among clinical and environmental specimens (34). This finding is consistent with the results of other recent clinical and environmental surveys in the United States, Europe, and Australia. The Cg outbreak on Vancouver Island also yielded only MATα isolates (8,35). Overall, our inferences are based on limited data, since most published studies on cryptococcosis do not include detailed characterization of Cryptococcus strains. Future epidemiologic studies will likely yield a more complete picture of the causative varieties or species of Cryptococcus across the United States.
As previously noted, CnVG infections are predominant in AIDS patients around the world, except in Europe, where CnVN is seen in sizable numbers. One explanation for this phenomenon is that CnVG is best adapted for the colonization of soil and pigeon droppings. However, CnVN may dominate the same ecologic niche in parts of Europe for undetermined causes (10). The lower incidence of Cg infection in AIDS patients could be due to the paucity of regions around the world in which Cg is endemic and the reported association of Cg disease with the flowering of Eucalyptus camaldulensis trees in certain areas (6). The unprecedented outbreak of Cg infection in Vancouver Island already comprises 66 human and 50 animal cases of cryptococcosis. Ongoing investigation in Vancouver Island indicate that the numbers of human and animal cases are increasing (130 human cases and >200 animal cases), which led to the recent change in the definition of Cg outbreak to Cg endemicity in this region (7). Additionally, Cg was isolated from swab samples from the bark of trees of many species (alder, arbutus, bitter cherry, cedar, fir, garry oak, maple, spruce), as well as from soil and air samples near these trees (7). These investigations have added a new dimension to our understanding of Cg ecology and suggest that this pathogen is neither restricted in its geographic distribution nor to its presumed natural host, Eucalyptus trees.
Our results indicated that both CE-FLA and CE-SSCP of pheromone genes are amenable to semi-automation and large-scale analyses of pathogenic Cryptococcus species, varieties, mating types, and hybrids. Each step of this analysis, namely, PCR, heat denaturation with formamide, and subsequent loading of samples, can be carried out in 48- or 96-well trays, which allow the use of multichannel or automated pipettors. Both CE-FLA and CE-SSCP individual runs are completed in <20 min, and the instrument can be programmed for multiple runs, thereby giving a high throughput. Thus, analyzing hundreds to thousands of strains is a good possibility, especially in reference laboratories. Moreover, the electrophoretic runs are saved as electronic files for easy portability over the Internet and to facilitate interlaboratory comparisons. This study reports a logical improvement over our earlier published method on pheromone PCR-RFLP for characterizing Cryptococcus isolates. The use of CE-FLA and CE-SSCP allowed us to dispense with running specialized gels as well as the use of unique restriction digestion schemes (21). Thus, CE-FLA alone leads to visualization of size differences in MFα1 versus MFa1 pheromones, which would distinguish mating types and hybrids. The species and varieties could be distinguished by CE-SSCP on the basis of polymorphisms in nucleotide sequences of MFα1 and MFa1 in Cg, CnVG, and CnVN. Thus, 1 typing method had the potential to replace multiple tests, such as specialized media and serotyping kits for species/variety determination, crossing with tester strains on mating agar, and flow cytometry for hybrid determination. The current limitations of this approach include the use of 2 polymers and run temperatures, which makes it necessary to run CE-FLA and CE-SSCP as batch applications on ABI 310 Genetic Analyzer. Since individual electrophoretic runs are completed in ≈20 min, ≈16 hours will be necessary to analyze ≈45 samples (one 48-sample tray) by CE-FLA, followed by change of polymer and run conditions, and another 16 hours to complete CE-SSCP analyses. However, these limitations could be easily overcome in the upgraded model of this instrument (ABI 3130), which has 4–16 capillaries and hands-free, 24-hour operation capabilities for simultaneous analyses of multiple samples, thereby considerably decreasing turnaround time.
Dr Sudha Chaturvedi is a research scientist in the Mycology Laboratory of the Wadsworth Center (New York State Department of Health) and an assistant professor of biomedical sciences, School of Public Health, State University of New York, Albany. Her research interests include fungal and parasite pathogenesis, protein transport and secretion mechanisms, and molecular epidemiology.
Acknowledgments
We thank Birgit Stein and Andrea Doney for technical support and Adriana Verschoor for editorial comments.
This study was funded in part with NIH grants AI-48462 (S.C.), AI-41968 (S.C.), AI-53732 (V.C.) and an educational award from Pfizer Inc. (V.C.).
References
- Kwon-Chung KJ, Boekhout T, Fell JW, Daiz M. Proposal to conserve the name Cryptococcus gattii against C. hondurianus and C. bacillisporus (Basidiomycota, Hymenomycetes, Tremellomycetidae). Taxon. 2002;51:804–6. DOIGoogle Scholar
- Casadevall A, Perfect JR. Cryptococcus neoformans, 1st ed. Washington: ASM Press; 1998.
- Kwon-Chung KJ, Bennett JE. Medical mycology. Philadelphia: Lea & Febiger; 1992.
- Speed BR, Dunt D. Clinical and host differences between infections with two varieties of Cryptococcus neoformans. Clin Infect Dis. 1995;21:28–34. DOIPubMedGoogle Scholar
- Lazera MS, Cavalcanti MA, Londero AT, Trilles L, Nishikawa MM, Wanke B. Possible primary ecological niche of Cryptococcus neoformans. Med Mycol. 2000;38:379–83.PubMedGoogle Scholar
- Bartlet K. Canadian Cryptococcus gattii—temporary visa or landed immigrant? Abstr. S13, p 71. In: Abstracts of the 6th International Conference on Cryptococcus and Cryptococcosis, Boston, MA, USA. Boston: Boston University School of Medicine; 2005.
- Kidd SE, Hagen F, Tscharke RL, Huynh M, Bartlett KH, Fyfe M, A rare genotype of Cryptococcus gattii caused the cryptococcosis outbreak on Vancouver Island (British Columbia, Canada). Proc Natl Acad Sci U S A. 2004;101:17258–63. DOIPubMedGoogle Scholar
- Stephen C, Lester S, Black W, Fyfe M, Raverty S. Multispecies outbreak of cryptococcosis on southern Vancouver Island, British Columbia. Can Vet J. 2002;43:792–4.PubMedGoogle Scholar
- Dromer F, Mathoulin S, Dupont B, Letenneur L, Ronin B. Individual and environmental factors associated with infections due to Cryptococcus neoformans serotype D. Mol Microbiol. 1996;23:91–6.
- Chen S, Sorrell CT, Nimmo G, Speed B, Currie B, Ellis D, Epidemiology and host- and variety-dependent characteristics of infection due to Cryptococcus neoformans in Australia and New Zealand. Australasian Cryptococcal Study Group. Clin Infect Dis. 2000;31:499–508. DOIPubMedGoogle Scholar
- Narasipura SD, Chaturvedi V, Chaturvedi S. Characterization of Cryptococcus neoformans var. gattii SOD2 reveals distinct roles of the two superoxide dismutases in fungal biology and virulence. Mol Microbiol. 2005;55:1782–800. DOIPubMedGoogle Scholar
- Narasipura SD, Ault JG, Behr MJ, Chaturvedi V, Chaturvedi S. Characterization of Cu,Zn superoxide dismutase (SOD1) gene knockout mutant of Cryptococcus neoformans var. gattii: role in biology and virulence. Mol Microbiol. 2003;47:1681–94. DOIPubMedGoogle Scholar
- Cox GM, Harrison TS, McDade HC, Taborda CP, Heinrich G, Casadevall A, Superoxide dismutase influences the virulence of Cryptococcus neoformans by affecting growth within macrophages. Infect Immun. 2003;71:173–80. DOIPubMedGoogle Scholar
- Wickes BL, Mayorga ME, Edman U, Edman JC. Dimorphism and haploid fruiting in Cryptococcus neoformans: association with the α-mating type. Proc Natl Acad Sci U S A. 1996;93:7327–31. DOIPubMedGoogle Scholar
- Boekhout T, Theelen B, Diaz M, Fell JW, Hop WC, Abeln EC, Hybrid genotypes in the pathogenic yeast Cryptococcus neoformans. Microbiology. 2001;147:891–907.PubMedGoogle Scholar
- Brandt MS, Bragg SL, Pinner RW. Multilocus enzyme typing of Cryptococcus neoformans. J Clin Microbiol. 1993;31:2819–23.PubMedGoogle Scholar
- Chaturvedi V, Fan J, Stein B, Behr MJ, Samsonoff WA, Wickes BL, Molecular genetic analyses of mating pheromones reveals intervariety mating/hybridization in Cryptococcus neoformans. Infect Immun. 2002;70:5225–35. DOIPubMedGoogle Scholar
- Lengeler KB, Cox GM, Heitman J. Serotype AD strains of Cryptococcus neoformans are diploid or aneuploid and are heterozygous at the mating-type locus. Infect Immun. 2001;69:115–22. DOIPubMedGoogle Scholar
- Tanaka R, Nishimura K, Miyaji M. Ploidy of serotype AD strains of Cryptococcus neoformans. J Med Mycol. 1999;40:31–4. DOIPubMedGoogle Scholar
- Chaturvedi S, Rodeghier B, Fan J, McClelland CM, Wickes BL, Chaturvedi V. Direct PCR of Cryptococcus neoformans MATα and MATa pheromones to determine mating types, ploidy, and variety: a tool for epidemiological and molecular pathogenesis studies. J Clin Microbiol. 2000;38:2007–9.PubMedGoogle Scholar
- Kwon-Chung KJ, Polacheck I, Bennett JE. Improved diagnostic medium for separation of Cryptococcus neoformans var. neoformans (serotype A and D) and Cryptococcus neoformans var. gattii (serotype B and C). J Clin Microbiol. 1982;15:535–7.PubMedGoogle Scholar
- Davidson RC, Moore TD, Odom AR, Heitman J. Characterization of the MFα pheromone of the human fungal pathogen Cryptococcus neoformans. Mol Microbiol. 2000;38:1017–26. DOIPubMedGoogle Scholar
- McClelland CM, Fu J, Woodlee GL, Seymour TS, Wickes BL. Isolation and characterization of the Cryptococcus neoformans MATa pheromone gene. Genetics. 2002;160:935–47.PubMedGoogle Scholar
- Baere TD, Claeys G, Swinne D, Massonet C, Verschraegen G, Muylaert A, Identification of cultured isolates of clinically important yeast species using fluorescent fragment length analysis of the amplified internally transcribed rRNA spacer 2 region. BMC Microbiol. 2002;2:21–8. DOIPubMedGoogle Scholar
- Ghozzi R, Morand P, Ferroni A, Bereti JL, Bingen E, Segond C, Capillary electrophoresis–single strand confirmation polymorphism analysis for rapid identification of Pseudomonas aeruginosa and other gram-negative nonfermenting bacilli recovered from patients with cystic fibrosis. J Clin Microbiol. 1999;37:3374–9.PubMedGoogle Scholar
- Montiel D, Dickinson MJ, Lee HA, Dyer PS, Jeenes DJ, Roberts IN, Genetic differentiation of the Aspergillus section Flavi complex using AFLP fingerprints. Mycol Res. 2003;107:143427–13. DOIPubMedGoogle Scholar
- Sheffield VC, Beck JS, Kwitek AE, Sandstrom DW, Stone EM. The sensitivity of single-strand conformation polymorphism analysis for the detection of single base substitutions. Genomics. 1993;16:325–32. DOIPubMedGoogle Scholar
- Castanon-Olivares LR, Arreguin-Espinosa R, Ruiz-Palacios y Santos G, Lopez-Martinez R. Frequency of Cryptococcus species and varieties in Mexico and their comparison with some Latin American countries. Rev Latinoam Microbiol. 2000;42:35–40.PubMedGoogle Scholar
- Chen SC. Cryptococcosis in Australasia and the treatment of cryptococcal and other fungal infections with liposomal amphotericin B. J Antimicrob Chemother. 2002;49(Suppl 1):57–61. DOIPubMedGoogle Scholar
- Karstaedt AS, Crewe-Brown HH, Dromer F. Cryptococcal meningitis caused by Cryptococcus neoformans var. gattii, serotype C, in AIDS patients in Soweto, South Africa. Med Mycol. 2002;40:7–11.PubMedGoogle Scholar
- Poonwan N, Mikami Y, Poosuwan S, Boon-Long J, Mekha N, Kusum M, Serotyping of Cryptococcus neoformans strains isolated from clinical specimens in Thailand and their susceptibility to various antifungal agents. Eur J Epidemiol. 1997;13:335–40. DOIPubMedGoogle Scholar
- Steenbergen JN, Casadevall A. Prevalence of Cryptococcus neoformans var. neoformans (Serotype D) and Cryptococcus neoformans var. grubii (Serotype A) isolates in New York City. J Clin Microbiol. 2000;38:1974–6.PubMedGoogle Scholar
- Kwon-Chung KJ, Bennett JE. Distribution of α and a mating types of Cryptococcus neoformans among natural and clinical isolates. Am J Epidemiol. 1978;108:337–40.PubMedGoogle Scholar
- Fraser JA, Subaran RL, Nichols CB, Heitman J. Recapitulation of the sexual cycle of the primary fungal pathogen Cryptococcus neoformans var. gattii: implications for an outbreak on Vancouver Island, Canada. Eukaryot Cell. 2003;2:1036–45. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 11, Number 11—November 2005
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Vishnu Chaturvedi, Mycology Laboratory, Wadsworth Center, 120 New Scotland Ave, Albany, NY 12208-2002, USA; fax: 518-486-7811
Top