Volume 13, Number 3—March 2007
Research
Worldwide Emergence of Extensively Drug-resistant Tuberculosis
Abstract
Mycobacterium tuberculosis strains that are resistant to an increasing number of second-line drugs used to treat multidrug-resistant tuberculosis (MDR-TB) are becoming a threat to public health worldwide. We surveyed the Network of Supranational Reference Laboratories for M. tuberculosis isolates that were resistant to second-line anti-TB drugs during 2000–2004. We defined extensively drug-resistant TB (XDR-TB) as MDR-TB with further resistance to ≥3 of the 6 classes of second-line drugs. Of 23 eligible laboratories, 14 (61%) contributed data on 17,690 isolates, which reflected drug susceptibility results from 48 countries. Of 3,520 (19.9%) MDR-TB isolates, 347 (9.9%) met criteria for XDR-TB. Further investigation of population-based trends and expanded efforts to prevent drug resistance and effectively treat patients with MDR-TB are crucial for protection of public health and control of TB.
Multidrug-resistant tuberculosis (MDR-TB) has been documented in nearly 90 countries and regions worldwide (1); 424,203 cases of MDR-TB were estimated to have occurred in 2004, which is 4.3% of all new and previously treated TB cases (2). Treatment for MDR-TB patients requires use of second-line drugs for ≥24 months. These drugs are more costly, toxic, and less effective than first-line drugs used for routine treatment of TB (3–6). As with other diseases, resistance to TB drugs results primarily from nonadherence by patients, incorrect drug prescribing by providers, poor quality drugs, or erratic supply of drugs (7).
To facilitate treatment of MDR-TB in resource-limited countries, where most TB cases occur (1,2), the World Health Organization (WHO) and its partners developed the Green Light Committee, which helps ensure proper use of second-line drugs, to prevent further drug resistance (8). Nonetheless, the Green Light Committee encountered numerous anecdotal reports of MDR-TB cases with resistance to most second-line drugs. Once a strain has developed resistance to second-line drugs, these new TB strains are even more difficult to treat with existing drugs. Untreated or inadequately treated patients are at increased risk of spreading their disease in the community, which could lead to outbreaks in vulnerable populations and widespread emergence of a lethal, costly epidemic of drug-resistant TB, reminiscent of the MDR-TB outbreaks in the early 1990s (9–13). Therefore, to determine whether these anecdotal reports were isolated events, early evidence of an emerging epidemic, or the occurrence of virtually untreatable forms of drug-resistant TB that had not been described previously in different parts of the world, we characterized and quantified the frequency of second-line–drug resistance in several geographic regions.
We sought to determine the extent to which highly resistant M. tuberculosis strains have been identified by the international laboratories that participate in the Network of Supranational Reference Laboratories (SRLs). The SRL Network consists of 25 highly proficient TB laboratories on 6 continents. These laboratories collaborate with national reference laboratories to strengthen culture and drug-susceptibility testing capacity and to provide quality control for the WHO/International Union Against Tuberculosis and Lung Diseases Global Project on Anti-TB Drug Resistance (14).
Participants
From November 2004 through November 2005, we surveyed the global SRL Network. All SRL directors were invited to participate during the 2004 annual SRL directors meeting, by individual mailings, and by personal phone calls. Drug-susceptibility testing results were requested for M. tuberculosis isolates that had been tested for resistance to first-line drugs and second-line drugs during 2000–2004. Two SRLs were not eligible because they did not test for second-line drugs or tested for <3 classes of second-line drugs.
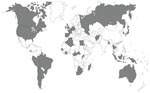
The 14 SRLs that provided data for this study support 112 TB laboratories in 80 countries worldwide (Figure 1). SRLs serve as international reference laboratories to a wide geographic area, performing drug-susceptibility testing that may not be available in a country (e.g., for second-line drugs) and providing quality assurance for first-line–drug testing. Most SRLs also serve as the national reference laboratory for the country in which they are located; they receive varying proportions of isolates from their own and other countries for surveillance, clinical diagnosis, and quality assurance. First-line–drug susceptibility testing is performed on all isolates; second-line–drug susceptibility testing is usually limited to isolates from patients known or suspected to have drug-resistant TB. Of the 14 participating SRLs, not all tested for all 6 classes of second-line drugs, and 4 did not submit data for the entire survey period.
In contrast, the SRL in the Republic of Korea serves as the national reference laboratory and routinely performs an extended diagnostic panel of drug-susceptibility testing on isolates from culture-positive TB patients referred from health centers, hospitals, and clinics in the Republic of Korea. This SRL tests all isolates for 6 classes of second-line drugs; thus, data from the Republic of Korea reflect most culture-positive cases and provide a close approximation to a population estimate of prevalence. Because of the large number of isolates received and because sampling for these isolates is systematically different from that at the other SRLs (testing of all TB patients in the Republic of Korea vs testing of patients more likely to have drug-resistant TB in other SRLs), resistance patterns for the Republic of Korea were analyzed separately from those for the other SRLs.
Laboratory Methods
Among participating SRLs, different but internationally accepted methods were used to test for second-line drug resistance (details available upon request). Validation of drug-susceptibility testing results for second-line drugs was not performed as part of this survey, but as part of their role as global reference laboratories, all SRLs participate in international proficiency testing for first-line drugs. Quality assurance procedures for second-line–drug susceptibility testing have not been developed; as a proxy for quality assurance, we examined the accuracy of second-line–drug susceptibility testing among isolates susceptible to the 4 main first-line drugs (isoniazid [INH], rifampin [RIF], ethambutol, and streptomycin). On the basis of known mechanisms of drug resistance, finding an isolate that is susceptible to all first-line drugs and resistant to second-line drugs is unlikely (7).
Procedures and Definitions
A standardized reporting form requested anonymous data for all isolates tested for resistance to ≥3 second-line drug classes during 2000–2004. Data were abstracted from the records, electronic or paper, depending on laboratory practices for data management. Results were submitted for 1 isolate per patient. Because SRLs rarely receive multiple isolates from the same patient, reporting of the same patient more than once was unlikely (B. Metchock and G.H. Bai, pers. comm.). No specimens were collected for this study; we used only data from records of isolates that had already been tested. Limited clinical information about the patient was available with each isolate. Consistent data were available for country of origin and date of drug-susceptibility testing. Data about age and TB treatment history were available for <10% of patients, so analysis was not considered reliable for these variables.
To best compare data for the study samples with data from the Global Drug Resistance Survey and other population-based drug-resistance surveillance, we analyzed first-line–drug resistance patterns according to standard methods used in anti-TB–drug resistance surveys (1). These patterns included any drug resistance, monoresistance (resistance to only the 1 specified drug), polyresistance (resistance to ≥2 first-line drugs, but which drugs not specified), and multidrug resistance (resistance to at least INH and RIF, with or without other drugs).
We defined 6 classes of second-line drugs as follows: aminoglycosides other than streptomycin (e.g., kanamycin and amikacin), cyclic polypeptides (e.g., capreomycin), fluoroquinolones (e.g., ofloxacin, ciprofloxacin, levofloxacin, and moxifloxacin), thioamides (e.g., prothionamide and ethionamide), serine analogs (e.g., cycloserine and terizidone), and salicylic acid derivatives (e.g., para-aminosalicyclic acid).
For this survey we created a consensus definition that incorporates second-line–drug susceptibility results and is based on international guidelines for management of drug-resistant TB (15). The mainstay of an MDR-TB treatment regimen consists of 1 injectible drug (e.g., aminoglycoside or cyclic polypeptide) and a fluoroquinolone; additional drugs from the remaining classes are added until the total reaches 4–6 drugs to which the organism is susceptible. If the infecting organism is resistant to ≥3 second-line drug classes, designing a treatment regimen with sufficient drugs that are known to be effective against TB is difficult. Thus, we defined extensively drug-resistant TB (XDR-TB) isolates as those meeting the criteria established for MDR-TB plus resistance to ≥3 of the 6 classes of second-line drugs.
Second-line–drug resistance patterns were analyzed by geographic region from which the isolate was submitted to the SRL. Regions were grouped into epidemiologically meaningful categories on the basis of prevalence of TB and MDR-TB (1,16). This retrospective survey was evaluated and approved as public health surveillance by the US Centers for Disease Control and Prevention (CDC).
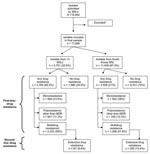
We received data for 18,462 patients from 14 (61%) of 23 eligible SRLs. We excluded those patients tested before 2000 (n=223), tested after 2004 (n = 14), or tested for resistance to <3 classes of second-line drugs (n = 535). Our final study sample consisted of 17,690 patients whose isolates were tested for resistance to ≥3 second-line drugs during 2000–2004 (Figure 2). Of these, 11,939 (67.5%) patients were from the Republic of Korea and 5,751 (32.5%) were from the remaining SRLs.
First-line–Drug Susceptibility
Among isolates from patients from the 13 SRLs other than the Republic of Korea, 3,765 (65.5%) were resistant to ≥1 first-line TB drug (Table 1). Of these, 3,305 (58.5%) were resistant to at least INH and 2,345 (41.5%) were resistant to at least RIF. Among isolates from the Republic of Korea patients, 2,508 (21%) had resistance to any drug; most (n = 2,196; 18.4%) were resistant to INH.
Single-drug resistance was found for isolates from 884 (15.4%) patients from the 13 SRLs; 456 (8.1%) of these were resistant to INH and 99 (1.8%) to RIF. Among isolates from patients from the Republic of Korea, 952 (8%) displayed single-drug resistance, 666 (5.6%) to INH and 148 (1.2%) to RIF.
Polyresistance other than MDR-TB was seen for isolates from 651 (11.5%) patients from the 13 SRLs and 258 (2.2%) from the Republic of Korea SRL. Not all SRLs routinely tested for resistance to pyrazinamide.
Multidrug resistance (i.e., MDR-TB) was present in isolates from 2,222 (39.4%) patients from the 13 SRLs and 1,298 (10.9%) from the Republic of Korea. Resistance to all first-line drugs tested (i.e., MDR-TB with additional resistance to ethambutol and streptomycin) was found in isolates from 1,017 (18.6%) patients from the 13 SRLs and 233 (2%) from the Republic of Korea SRL.
Second-line–Drug Susceptibility
Among patients from the 13 SRLs, resistance to aminoglycosides was detected in 489 (8.7%) isolates and to fluoroquinolones in 298 (5.3%) (Table 2). Among isolates from Republic of Korea patients, resistance was most commonly seen to fluoroquinolones (n = 524, 4.4%) and thioamides (n = 259, 2.2%).
From all SRLs, isolates that were resistant to at least INH and RIF (i.e., MDR-TB; n = 3,520) and tested for susceptibility to ≥3 second-line drugs were combined for analysis of second-line–drug resistance patterns. Resistance to ≥1 class of second-line drug was present in 1,542 (43.8%) MDR-TB patients (Table 3). The most commonly observed patterns were resistance to aminoglycosides (n = 630, 18.3%), fluoroquinolones (n = 673, 19.3%), and thioamides (n = 605, 19.3%).
MDR-TB patients whose isolates had further resistance to ≥3 classes of second-line drugs were classified as XDR-TB (Table 3). A total of 347 (9.9%) MDR-TB patients met criteria for XDR-TB. Among XDR-TB patients, combination drug-resistance patterns included 90 (3.4%) with resistance to aminoglycosides, capreomycin and fluoroquinolones; 102 (3.4%) with resistance to aminoglycosides, fluoroquinolones, and thioamides; and 94 (3.8%) with resistance to fluoroquinolones, thioamides, and para-aminosalicyclic acid. Nearly half (n = 167, 48.1%) of all XDR-TB isolates were resistant to all 4 first-line drugs, bringing the total to ≥7 drugs to which the isolate was resistant.
The proportion of XDR-TB patients by region is shown in Table 4. Among the group of industrialized nations, 53 (6.5%) MDR-TB patients met criteria for XDR-TB. Among patients from Russia and Eastern Europe, 55 (13.6%) MDR-TB patients met criteria for XDR-TB. Among patients from the Republic of Korea, 200 (15.4%) MDR-TB patients, who accounted for 1.7% of all M. tuberculosis isolates tested, met criteria for XDR-TB. According to the revised Global XDR-TB Task Force definition (www.who.int/mediacentre/news/notes/2006/np29/en/index.html), 234 (6.6%) isolates met criteria for XDR-TB.
In evaluating the accuracy of second-line–drug susceptibility testing, we found that 7 (0.1%) of 11,426 patients fully susceptible to all first-line drugs were resistant to 2 second-line drugs, and 109 (1%) were resistant to 1 second-line drug. Most of these patients were resistant to fluoroquinolones.
This study represents the first assessment of the widespread occurrence of M. tuberculosis with such extensive drug resistance as to be nearly untreatable with currently available drugs, according to international guidelines. We provide data on second-line–drug resistance for the largest sample of patients to date, including >5,000 patients from 47 countries, apart from the Republic of Korea. The definition of XDR-TB in this survey is based on WHO guidelines for the programmatic management of drug-resistant TB; the guidelines recommend treatment with ≥4 drugs known to be effective (15). Therefore, with ≤3 remaining classes of second-line drugs to which the infecting organism is susceptible, treatment of these patients cannot meet international standards. XDR-TB has been detected in all regions of the world. XDR-TB strains in this study also have high rates of resistance to pyrazinamide and ethambutol, thereby severely limiting the treatment options available.
Analysis of combination second-line–drug resistance patterns is critical for clinicians and policymakers who design treatment regimens for these patients. Although limited data exist in the literature about second-line–drug resistance patterns among MDR-TB patients, data from patients undergoing retreatment for TB in Hong Kong showed that 30 (17%) MDR-TB isolates were resistant to ≥3 second-line drugs (17), thereby meeting criteria for XDR-TB. A drug-resistance survey of 447 culture-positive new patients and patients undergoing retreatment in Abkhazia, Republic of Georgia, found that of 63 MDR-TB patients, 2 (3%) had additional resistance to 3 second-line drug classes, consistent with XDR-TB (18). More recently, clusters of XDR-TB have been reported in South Africa and Iran (19,20) and have been associated with HIV infection and rapid and high death rates.
The emergence of new strains of TB that are resistant to second-line drugs, especially in settings where TB control programs have become unable to adequately monitor treatment regimens for MDR-TB, is cause for concern. After the resurgence of TB in industrialized countries during the 1980s and increased awareness of this global problem, implementation of strong TB control programs based on the principles of the global directly observed treatment strategy, short course (DOTS) improved treatment outcomes and reduced TB and MDR-TB incidence in several countries. This framework for DOTS, promulgated by WHO, and the pilot MDR-TB management projects (DOTS-Plus projects) became the basis for programmatic management of MDR-TB, which has demonstrated feasibility and effectiveness in low- and middle-income countries (5,15). However, second-line drugs are available worldwide outside of well-organized TB-control programs (WHO, unpub. data).
Improper treatment of drug-resistant TB, such as using too few drugs, relying on poor quality second-line drugs, and failing to ensure adherence to treatment, will likely lead to increases in XDR-TB. Strengthening basic TB programs and infection control measures is crucial for preventing the selective pressure and environments in which resistant strains are transmitted from person to person. Additionally, MDR-TB programs that rely on quality-assured and internationally recommended treatment regimens according to WHO guidelines must be scaled up and strengthened to stem further second-line–drug resistance and spread of XDR-TB. The Green Light Committee provides a global mechanism to help affected countries achieve these steps. A commentary published in 2000 predicted that “failure to institute [the] entire DOTS-Plus package is likely to destroy the last tools available to combat [TB], and may ultimately result in the victory of the tubercle bacillus over mankind” (21). XDR-TB is an indirect indicator of program failure to adequately diagnose, prevent, and treat MDR-TB.
Documenting the emergence of XDR-TB requires a laboratory-based diagnosis that relies on first- and second-line–drug susceptibility testing. A limitation to accurate detection of XDR-TB is that existing tests for resistance to second-line drugs are not yet standardized and are less reproducible than tests for resistance to INH and RIF. Lack of international recommendations for use, as well as lack of standardization and the historical unavailability of MDR-TB treatment in the public sector, has limited use of second-line–drug susceptibility testing on a wider scale. As access to treatment with second-line drugs increases, standardized methods, improved diagnostics, and quality assurance for second-line–drug susceptibility testing are urgently needed to enable reliable testing and design of appropriate treatment regimens. Although internationally accepted methods were used by all laboratories, the precise methods and drug concentrations used varied among participating SRLs (22). Because these SRLs represent some of the most highly performing laboratories on 6 continents, results of drug-susceptibility testing are credible within the context of stated limitations. Initial studies that standardized different methods for second-line–drug susceptibility testing have been completed (23–26), but more are needed.
Our study has other limitations. The numbers reported for XDR-TB probably represent an underestimate of the true number of cases because not all SRLs and not all national reference laboratories test for all 6 classes of second-line drugs. In the absence of test results for all 6 classes of second-line drugs, we speculate, on the basis of a patient’s TB treatment history and known patterns of drug cross-resistance, that many other unidentified patients are likely to have had and died from XDR-TB. For example, an MDR-TB isolate that is also resistant to an aminoglycoside and a fluoroquinolone but that has not been tested for the other second-line drug classes is very likely to be resistant to an additional second-line drug class for the following reasons: INH and ethionamide have a 15%–20% rate of cross-resistance (27); kanamycin and capreomycin cross-resistance is common, ranging from 20%–60% (CDC, unpub. data) (28,29); and in this study, isolates that were resistant to all 4 first-line drugs as well as an aminoglycoside and a fluoroquinolone were 70%–80% likely to be resistant to at least 1 additional class of second-line drug.
Another limitation is that data from most SRLs were drawn from a convenience sample of isolates and reflect referral bias. Thus, these data can not be considered representative of a patient population or region, and actual denominators are difficult to determine. For this reason, although estimates of prevalence are possible, they cannot be generalized to the local or regional population. However, our study is the first to report XDR-TB patients in multiple geographic regions; future systematic surveys are needed to determine the true extent of this disease. Data from the Republic of Korea reflect a more comprehensive policy for drug-susceptibility testing and provide an estimate of the population prevalence in this setting. However, the 10.9% rate of MDR-TB for the Republic of Korea is higher than rates reported from other national drug resistance surveys and may reflect other unknown referral biases (1).
Lastly, we had limited clinical information about each patient because information submitted to each SRL varied and was not reliably available for inclusion in the analysis. Data about TB treatment history, patient age and sex, or HIV status are not routinely collected by all laboratories. Genotyping data were not available to confirm whether XDR-TB isolates are related to W variant of the Beijing strain, a highly drug-resistant strain of M. tuberculosis responsible for large nosocomial outbreaks in New York in the early 1990s (30).
Despite these limitations, our survey provides the first documentation of the emergence of XDR-TB as a serious worldwide public health threat. XDR-TB was identified on 6 continents and is significantly associated with worse treatment outcomes than MDR-TB (31,32). The emergence of XDR-TB, coupled with the increased use of second-line drugs, suggests that urgent measures are needed to improve rational use of quality-assured second-line drugs. In addition, population-based surveillance for second-line–drug susceptibility testing is needed to better describe the magnitude of XDR-TB worldwide, track trends, and plan a public health response. Indeed, the convergence of XDR-TB with the HIV epidemic may undermine gains in HIV prevention and treatment programs and requires urgent interventions. These interventions include ensuring adherence to recommended international standards of care aimed at promptly and reliably diagnosing TB, ensuring adherence to recommended treatment regimens with demonstrated efficacy, implementing infection control precautions where patients congregate, and improving laboratories’ capacity to accurately and rapidly detect drug-resistant M. tuberculosis isolates so that patients can receive effective treatment (33). Other unmet needs include further development of international standards for second-line–drug susceptibility testing, new anti-TB drug regimens, and better diagnostic tests for TB and MDR-TB. Such measures are crucial if future generations are to be protected from potentially untreatable TB.
Dr Shah is an internist and epidemiologist with Albert Einstein College of Medicine. She is also a guest researcher with the Division of Tuberculosis Elimination at CDC, where she was an Epidemic Intelligence Service Officer at the time of this study. Her research interests focus on TB and HIVcoinfection, drug resistance, and global health.
Acknowledgment
We thank Kenneth G. Castro, Michael F. Iademarco, Mario Raviglione, Paul Nunn, and Ernesto Jaramillo for technical assistance and critical appraisal of the manuscript.
References
- World Health Organization. Anti-tuberculosis drug resistance in the world: Report 3. Document no. (WHO/HTM/TB/2004.343). Geneva: The Organization; 2004.
- Zignol M, Hosseini MS, Wright A, Weezenbeek CL, Nunn P, Watt CJ, et al. Global incidence of multidrug-resistant tuberculosis.J Infect Dis. 2006;194:479–85. DOIPubMedGoogle Scholar
- Rajbhandary SS, Marks SM, Bock NN. Costs of patients hospitalized for multidrug-resistant tuberculosis.Int J Tuberc Lung Dis. 2004;8:1012–6.PubMedGoogle Scholar
- Ward HA, Marciniuk DD, Hoeppner VH, Jones W. Treatment outcome of multidrug-resistant tuberculosis among Vietnamese immigrants.Int J Tuberc Lung Dis. 2005;9:164–9.PubMedGoogle Scholar
- Nathanson E, Lambregts-van Weezenbeek C, Rich ML, Gupta R, Bayona J, Blondal K, et al. Multidrug-resistant tuberculosis management in resource-limited settings.Emerg Infect Dis. 2006;12:1389–97.PubMedGoogle Scholar
- Iseman MD. Treatment of multidrug-resistant tuberculosis.N Engl J Med. 1993;329:784–91. DOIPubMedGoogle Scholar
- Pablos-Mendez A, Lessnau K. Clinical mismanagement and other factors producing antituberculosis drug resistance. In: Bastian I, Portaels F, editors. Multidrug-resistant tuberculosis. Dordrecht (the Netherlands): Kluwer Academic Publishers; 2000. p. 59–76.
- Gupta R, Cegielski JP, Espinal MA, Henkens M, Kim JY, Lambregts-Van Weezenbeek CS, et al. Increasing transparency in partnerships for health: introducing the Green Light Committee.Trop Med Int Health. 2002;7:970–6. DOIPubMedGoogle Scholar
- Frieden TR, Sherman LF, Maw KL, Fujiwara PI, Crawford JT, Nivin B, et al. A multi-institutional outbreak of highly drug-resistant tuberculosis.JAMA. 1996;276:1229–35. DOIPubMedGoogle Scholar
- Coronado VG, Beck-Sague CM, Hutton MD, Davis BJ, Nicholas P, Villareal C, et al. Transmission of multidrug-resistant Mycobacterium tuberculosis among persons with human immunodeficiency virus infection in an urban hospital: epidemiologic and restriction fragment length polymorphism analysis.J Infect Dis. 1993;168:1052–5.PubMedGoogle Scholar
- Breathnach AS, de Ruiter A, Holdsworth GM, Bateman NT, O’Sullivan DG, Rees PJ, An outbreak of multi-drug-resistant tuberculosis in a London teaching hospital.J Hosp Infect. 1998;39:111–7. DOIPubMedGoogle Scholar
- CDC. Nosocomial transmission of multidrug-resistant tuberculosis among HIV-infected persons—Florida and New York, 1988–1991.MMWR Morb Mortal Wkly Rep. 1991;40:585–91.PubMedGoogle Scholar
- Edlin BR, Tokars JI, Grieco MH, Crawford JT, Williams J, Sordillo EM, An outbreak of multidrug-resistant tuberculosis among hospitalized patients with the acquired immunodeficiency syndrome.N Engl J Med. 1992;326:1514–21.PubMedGoogle Scholar
- Laszlo A, Rahman M, Espinal M. WHO/IUATLD Network of Supranational Reference Laboratories. Quality assurance program for drug susceptibility testing of Mycobacterium tuberculosis in the WHO/IUATLD Supranational Reference Laboratory Network: five rounds of proficiency testing, 1994–1998.Int J Tuberc Lung Dis. 2002;6:748–56.Raviglione MPubMedGoogle Scholar
- World Health Organization. Guidelines for the programmatic management of drug-resistant tuberculosis [cited 2006 Jan 5]. Geneva: The Organization; 2006. Document no. WHO/HTM/TB/2006.361. Available from http://whqlibdoc.who.int/publications/2006/9241546956_eng.pdf).
- World Health Organization. Global tuberculosis control: WHO report. Geneva: The Organization; 2006. Document no. WHO/HTM/TB/2006.362.
- Kam KM, Yip CW. Surveillance of Mycobacterium tuberculosis susceptibility to second-line drugs in Hong Kong, 1995–2002, after the implementation of DOTS-Plus.Int J Tuberc Lung Dis. 2004;8:760–6.PubMedGoogle Scholar
- Pardini M, Iona E, Varaine F, Karakozian H, Arzumanian H, Brunori L, et al. Mycobacterium tuberculosis drug resistance: Abkhazia[letter]. Emerg Infect Dis. 2005;11:501–3.PubMedGoogle Scholar
- Gandhi NR, Moll A, Sturm AW, Pawinski R, Govender T, Lalloo U, et al. Extensively drug-resistant tuberculosis as a cause of death among patient co-infected with tuberculosis and HIV in a rural area in South Africa.Lancet. 2006;368:1575–80. DOIPubMedGoogle Scholar
- Masjedi MR, Farnia P, Sorooch S, Pooramiri MV, Mansoori SD, Zarifi AZ, et al. Extensively drug-resistant tuberculosis: 2 years of surveillance in Iran.Clin Infect Dis. 2006;43:841–7. DOIPubMedGoogle Scholar
- Lambregts-van Weezenbeek KSB, Reichman LB. DOTS and DOTS-Plus: what’s in a name.Int J Tuberc Lung Dis. 2000;4:995–6.PubMedGoogle Scholar
- Kim SJ. Is second-line anti-tuberculosis drug susceptibility testing reliable?[letter]. Int J Tuberc Lung Dis. 2004;8:1157–8.PubMedGoogle Scholar
- Heifets LB, Cangelosi GA. Drug susceptibility testing of Mycobacterium tuberculosis: a neglected problem at the turn of the century.Int J Tuberc Lung Dis. 1999;3:564–81.PubMedGoogle Scholar
- Krüüner A, Yates MD, Drobniewski FA. Evaluation of MGIT 960-based antimicrobial testing and determination of critical concentrations of first- and second-line antimicrobial drugs with drug-resistant clinical strains of Mycobacterium tuberculosis.J Clin Microbiol. 2006;44:811–8. DOIPubMedGoogle Scholar
- Rüsch-Gerdes S, Pfyffer GE, Casal M, Chadwick M, Siddiqi S. Multicenter laboratory validation of drug susceptibility testing of Mycobacterium tuberculosis against classical second-line drugs and newer antimicrobials by the BACTEC MGIT 960 technique.J Clin Microbiol. 2006;44:688–92. DOIPubMedGoogle Scholar
- Pfyffer GE, Bonato DA, Ebrahimzadeh A, Gross W, Hotaling J, Kornblum J, Multicenter laboratory validation of susceptibility testing of Mycobacterium tuberculosis against classical second-line and newer antimicrobial drugs by using the radiometric BACTEC 460 technique and the proportion method with solid media.J Clin Microbiol. 1999;37:3179–86.PubMedGoogle Scholar
- Canetti G. Present aspects of bacterial resistance in tuberculosis.Am Rev Respir Dis. 1965;92:687–703.PubMedGoogle Scholar
- Maus CE, Plikaytis BB, Shinnick TM. Molecular analysis of cross-resistance to capreomycin, kanamycin, amikacin, and viomycin in Mycobacterium tuberculosis.Antimicrob Agents Chemother. 2005;49:3192–7. DOIPubMedGoogle Scholar
- McClatchy JK, Kanes W, Davidson PT, Moulding TS. Cross-resistance in M. tuberculosis to kanamycin, capreomycin and viomycin.Tubercle. 1977;58:29–34. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Outbreak of multidrug-resistant tuberculosis at a hospital—New York City, 1991.MMWR Morb Mortal Wkly Rep. 1993;42:427–33.PubMedGoogle Scholar
- Centers for Disease Control and Prevention. Emergence of Mycobacterium tuberculosis with extensive resistance to second-line drugs—worldwide, 2000–2004.MMWR Morb Mortal Wkly Rep. 2006;55:301–5.PubMedGoogle Scholar
- Leimane V, Riekstina V, Holtz TH, Zarovska E, Skripconoka V, Thorpe LE, Clinical outcome of individualised treatment of multidrug-resistant tuberculosis in Latvia: a retrospective cohort study.Lancet. 2005;365:318–26.PubMedGoogle Scholar
- Tuberculosis Coalition for Technical Assistance. International standards for tuberculosis care [cited 2006 Jan 5]. The Hague: The Coalition; 2006. Available from http://www.who.int/tb/publications/2006/istc_report.pdf.
Figures
Tables
Cite This ArticleTable of Contents – Volume 13, Number 3—March 2007
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
N. Sarita Shah, Department of Medicine, Division of General Internal Medicine, Albert Einstein College of Medicine, 111 E. 210 St, Bronx, NY 10467, USA;
Top