Volume 16, Number 5—May 2010
Dispatch
Rickettsiae in Gulf Coast Ticks, Arkansas, USA
Abstract
To determine the cause of spotted fever cases in the southern United States, we screened Gulf Coast ticks (Amblyomma maculatum) collected in Arkansas for rickettsiae. Of the screened ticks, 30% had PCR amplicons consistent with Rickettsia parkeri or Candidatus Rickettsia amblyommii.
The Centers for Disease Control and Prevention identified Arkansas as a leading state for the incidence of Rocky Mountain spotted fever (causative agent Rickettsia rickettsii) and reported >15 cases per 1,000,000 persons in 2002 (1). Given the known cross-reactivity of serologic testing results for spotted fever group (SFG) rickettsia, it is unclear if cases outside the natural range of the vectors for R. rickettsii are misdiagnosed, if the pathogen is less virulent than previously suggested, or if additional rickettsiae are responsible for pathogenesis (2).
Recently, the Gulf Coast tick (Amblyomma maculatum) was identified as the primary vector of R. parkeri, a newly described pathogen that causes disease symptoms similar to Rocky Mountain spotted fever (3). R. parkeri has previously been identified in A. maculatum tick specimens collected in the southeastern United States (4) and from a human biopsy specimen in Virginia, USA (5). We have identified A. maculatum ticks collected from canids, felids, white-tailed deer, and a cow from locations throughout Arkansas (6). Notably, R. amblyommii has been identified as a potential pathogen and is found in lone star ticks (A. americanum) (7,8). We report the presence of DNA consistent with that of Candidatus Rickettsia amblyommii and R. parkeri in A. maculatum ticks in Arkansas.
We screened 112 A. maculatum ticks collected during March 2006–January 2008 from 22 dogs (Canis lupus familiaris) and 95 A. maculatum ticks collected during the 2008 hunting season from 52 white-tailed deer (Odocoileus virginianus Boddaert) for rickettsial DNA. Collectors removed specimens; stored them in vials containing 100% ethanol; and recorded tick collection date, location, and host (6). Ticks were identified by species, sex, life stage, and engorgement (9). Each sample was bisected longitudinally with a razor blade and subjected to the extraction procedure by using QIAGEN DNeasy (QIAGEN, Valencia, CA, USA) following the manufacturer’s protocols.
Tick DNA extracts were screened for SFG Rickettsia spp. DNA by PCR by using genus-specific primers for the citrate synthase (gltA) (10) and rickettsial outer membrane protein B (rompB) (11) genes. Reaction products were analyzed (12), and positive amplicons for gltA (513 bp) and rompB (578 bp) were sent to the University of North Texas Health Science Center (Fort Worth, TX, USA) for sequence determination. At least 1 amplicon from each host was sequenced to determine the Rickettsia species identity. PCR products were hydrolyzed with ExoSAP-IT (USB Corporation, Cleveland, OH, USA), and sequence determination was performed by using a BigDYE Terminator v.3.1 Cycle Sequencing Kit (Applied Biosystems, Inc., Foster City, CA, USA) followed by capillary electrophoresis on an ABI PRISM 310 Genetic Analyzer (Applied Biosystems, Inc.) (13).
Sequences were edited, aligned, and analyzed with Sequencher 4.7 (Gene Codes Corporation, Ann Arbor, MI, USA) and compared with sequences in GenBank (National Center for Biotechnology Information, Bethesda, MD, USA). BEAST version 1.4.2 software (http://beast.bio.ed.ac.uk/Main_Page) was used to infer phylogenetic relationships and create dendrograms (14). The consensus tree ran for 106 generations with a burn-in of 2 × 104. Established methods were used (12) to conduct parsimony bootstrap and maximum-likelihood analyses. Maximum-likelihood and unweighted parsimony analyses on the alignments were performed by using the branch and bound algorithm of PAUP* 4.0b10 (http://paup.csit.fsu.edu). Outgroup taxa were obtained from GenBank.
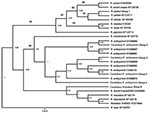
Of the 207 ticks, 62 were positive for Rickettsia spp. DNA by PCR. Nineteen ticks were positive for gltA only, 12 were positive for rompB only, and 31 were positive for both genes (Table). Of the ticks collected from white-tailed deer, 28 were positive, and those amplicons were 100% homologous with Candidatus Rickettsia amblyommii from GenBank (FJ455415, EU7228827, AY388899) (Table, Figure). Of the positive ticks collected from dogs, 3 had sequences with 100% similarity to either rompB (AF123717) or gltA (EF102236) of R. parkeri. A single tick (unengorged male) had a sequence 98% similar to GenBank sequences EF219464 (rompB) and EF451001 (gltA). The remaining 30 ticks collected from dogs that were positive all produced amplicons with 100% sequence identity to Candidatus R. amblyommii gltA (EF450708). However, rompB sequences generated from the same sample set demonstrated greater diversity (Table, Figure).
In total, 3 ticks collected from 3 different canine hosts produced sequences 100% identical to those of R. parkeri rompB (AF123717) and gltA (EF102236). Candidatus R. amblyommii sequences were identified in 29 ticks collected from 13 dogs and 25 ticks collected from 25 deer. The resulting Bayesian tree showed weak support (consistency index 0.792, tree length 159) (Figure). Neighbor-joining and maximum-likelihood trees supported the GenBank homologies.
We report the identification of SFG rickettsiae in A. maculatum ticks collected from Arkansas, specifically R. parkeri, Candidatus R. amblyommii, and an uncharacterized Rickettsia sp. sequence with high homology to GenBank sequence no. EF219464. Identification of these rickettsiae may be a public health concern given their recent association with cases of spotted fever (4,7,8). The risk for spotted fever transmission to humans is unknown but may be of concern to public health officials in Arkansas because of canid–human relationships and habitat fragmentation that has moved deer ranges closer to human habitation. Additional investigations of the distribution of A. maculatum ticks, the pathogenesis of Rocky Mountain spotted fever, and the ticks’ relationship to human disease should be conducted.
Ms Trout is a PhD candidate in the Department of Entomology at the University of Arkansas. Her dissertation focuses on the spatial identification and genetic characterization of ticks and their relationship with Borrelia and Rickettsia species within Arkansas.
Acknowledgments
We thank the personnel associated with the Arkansas Game and Fish Commission and Arkansas Veterinary Medical Association involved with collecting tick samples.
This research was supported in part by the University of Arkansas, Arkansas Agricultural Experiment Station.
References
- Centers for Disease Control and Prevention. Tickborne rickettsial diseases: statistics. 2008 [cited 2009 Aug 1]. http://www.cdc.gov/ticks/diseases/rocky_mountain_spotted_fever/statistics.html
- Raoult D, Parola P. Rocky Mountain spotted fever in the USA: a benign disease or a common diagnostic error? Lancet Infect Dis. 2008;8:587–9. DOIGoogle Scholar
- Paddock CD, Sumner JW, Comer JA, Zaki SR, Goldsmith CS, Goddard J, Rickettsia parkeri: a newly recognized cause of spotted fever rickettsiosis in the United States. Clin Infect Dis. 2004;38:812–3.Medline DOIGoogle Scholar
- Sumner JW, Durden LA, Goddard J, Stromdahl EY, Clark KL, Reeves WK, Gulf Coast ticks (Amblyomma maculatum) and Rickettsia parkeri, United States. Emerg Infect Dis. 2007;13:751–3.
- Whitman TJ, Richards AL, Paddock CD, Tamminga CL, Sniezek PJ, Jiang J, Rickettsia parkeri infection after tick bite, Virginia. Emerg Infect Dis. 2007;13:334–6. DOIGoogle Scholar
- Trout RT, Steelman CD, Szalanski AL, Loftin K. Establishment of Amblyomma maculatum (Gulf Coast tick) in Arkansas, USA. Fla Entomol. 2010;93:120–2. DOIGoogle Scholar
- Billeter SA, Blanton HL, Little SE, Levy MG, Breitschwerdt EB. Detection of Rickettsia amblyommii in association with a tick bite rash. Vector Borne Zoonotic Dis. 2007;7:607–10.Medline DOIGoogle Scholar
- Apperson CS, Engber B, Nicholson WL, Mead DG, Engel J, Yabsley MJ, Tick-borne diseases in North Carolina: is “Rickettsia amblyommii” a possible cause of rickettsiosis reported as Rocky Mountain spotted fever? Vector Borne Zoonotic Dis. 2008;8:597–606. DOIGoogle Scholar
- Lancaster JL. A guide to ticks of Arkansas. Fayetteville (AR): Agricultural Experiment Station, Division of Agriculture, University of Arkansas; 1973. Bulletin 779.
- Kollars TM Jr, Kengluecha A. Spotted fever group Rickettsia in Dermacentor variabilis (Acari: Ixodidae) infesting raccoons (Carnivora: Procyonidae) and opossums (Marsupialia: Didelphimorphidae) in Tennessee. J Med Entomol. 2001;38:601–2. DOIGoogle Scholar
- Regnery RL, Spruill CL, Plikaytis BD. Genotypic identification of rickettsiae and estimation of intraspecies sequence divergence for portions of two rickettsial genes. J Bacteriol. 1991;173:1576–89.
- Trout RT, Steelman CD, Szalanski AL. Population genetics and phylogeography of Ixodes scapularis from canines and deer in Arkansas. Southwest Entomologist. 2009;34:273–87. DOIGoogle Scholar
- Stromdahl EY, Vince MA, Billingsley PM, Dobbs NA, Williamson PC. Rickettsia amblyommii infecting Amblyomma americanum larvae. Vector Borne Zoonotic Dis. 2008;8:15–24. DOIGoogle Scholar
- Felsenstein J. Confidence limits on phylogenies: an approach using the bootstrap. Evolution. 1985;39:783–91. DOIGoogle Scholar
Figure
Table
Cite This ArticleTable of Contents – Volume 16, Number 5—May 2010
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Rebecca Trout, Department of Entomology, University of Arkansas, Fayetteville, AR 72701, USA
Top