Volume 18, Number 12—December 2012
Dispatch
No Evidence of Prolonged Hendra Virus Shedding by 2 Patients, Australia
Figure
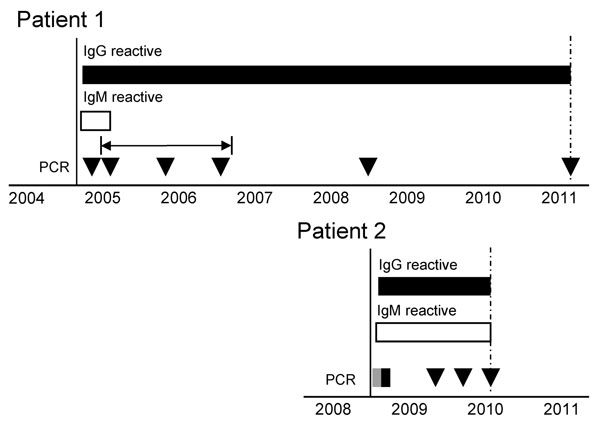
Figure. . Serologic and quantitative reverse transcription PCR (qRT-PCR) results for samples from 2 patients infected with Hendra virus, Australia. Testing was performed from the time at which symptoms first developed (black vertical line) until the most recent sample indicated (dashed vertical arrows). IgG (black bars) and IgM (white bars) reactivity was determined by using a modified microsphere immunoassay (10), and a positive control serum sample to determine the cutoff value. Virus RNA was detected (gray box) or not detected (black box or triangles) by using a qRT-PCR (11). The horizontal arrow indicates 3 samples stored at −80°C and tested retrospectively for Hendra virus RNA. Positive and negative controls were included in all tests and showed expected results.
References
- Field HE, Mackenzie JS, Daszak P. Henipaviruses: emerging paramyxoviruses associated with fruit bats. Curr Top Microbiol Immunol. 2007;315:133–59. DOIPubMedGoogle Scholar
- Goh KJ, Tan CT, Chew NK, Tan PS, Kamarulzaman A, Sarji SA, Clinical features of Nipah virus encephalitis among pig farmers in Malaysia. N Engl J Med. 2000;342:1229–35. DOIPubMedGoogle Scholar
- Tan CT, Goh KJ, Wong KT, Sarji SA, Chua KB, Chew NK, Relapsed and late-onset Nipah encephalitis. Ann Neurol. 2002;51:703–8. DOIPubMedGoogle Scholar
- Chong HT, Chong TT. Relapsed and late-onset Nipah encephalitis, a report of three cases. Neurology Journal of Southeast Asia. 2003;8:109–12.
- O’Sullivan JD, Allworth AM, Paterson DL, Snow TM, Boots R, Gleeson LJ, Fatal encephalitis due to novel paramyxovirus transmitted from horses. Lancet. 1997;349:93–5. DOIPubMedGoogle Scholar
- Wong KT, Robertson T, Ong BB, Chong JW, Yaiw KC, Wang LF, Human Hendra virus infection causes acute and relapsing encephalitis. Neuropathol Appl Neurobiol. 2009;35:296–305. DOIPubMedGoogle Scholar
- Australian veterinary emergency plan. Version 3.3; 2009 [cited 2012 Jun 28]. http://www.animalhealthaustralia.com.au/wp-content/uploads/2011/04/RPB3.3-05FINAL27Jul11.pdf
- Hanna JN, McBride WJ, Brookes DL, Shield J, Taylor CT, Smith IL, Hendra virus infection in a veterinarian. Med J Aust. 2006;185:562–4.PubMedGoogle Scholar
- Playford EG, McCall B, Smith G, Slinko V, Allen G, Smith I, Human Hendra virus encephalitis associated with equine outbreak, Australia, 2008. Emerg Infect Dis. 2010;16:219–23. DOIPubMedGoogle Scholar
- Bossart KN, McEachern JA, Hickey AC, Choudhry V, Dimitrov DS, Eaton BT, Neutralization assays for differential henipavirus serology using Biol-Plex protein array systems. J Virol Methods. 2007;142:29–40. DOIPubMedGoogle Scholar
- Smith IL, Halpin K, Warrilow D, Smith GA. Development of a fluorogenic RT-PCR assay (TaqMan) for the detection of Hendra virus. J Virol Methods. 2001;98:33–40. DOIPubMedGoogle Scholar
- Davis LE, DeBiasi R, Goade DE, Haaland KY, Harrington JA, Harnar JB, West Nile virus neuroinvasive disease. Ann Neurol. 2006;60:286–300. DOIPubMedGoogle Scholar
- Kapoor H, Signs K, Somsel P, Downes FP, Clark PA, Massey JP. Persistence of West Nile virus (WNV) IgM antibodies in cerebrospinal fluid from patients with CNS disease. J Clin Virol. 2004;31:289–91. DOIPubMedGoogle Scholar