Volume 22, Number 2—February 2016
CME ACTIVITY - Research
Sustained Transmission of Pertussis in Vaccinated, 1–5-Year-Old Children in a Preschool, Florida, USA
Cite This Article
Citation for Media
Introduction

Medscape, LLC is pleased to provide online continuing medical education (CME) for this journal article, allowing clinicians the opportunity to earn CME credit.
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint providership of Medscape, LLC and Emerging Infectious Diseases. Medscape, LLC is accredited by the ACCME to provide continuing medical education for physicians.
Medscape, LLC designates this Journal-based CME activity for a maximum of 1.0 AMA PRA Category 1 Credit(s)TM. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
All other clinicians completing this activity will be issued a certificate of participation. To participate in this journal CME activity: (1) review the learning objectives and author disclosures; (2) study the education content; (3) take the post-test with a 75% minimum passing score and complete the evaluation at http://www.medscape.org/journal/eid; (4) view/print certificate.
Release date: January 15, 2016; Expiration date: January 15, 2017
Learning Objectives
Upon completion of this activity, participants will be able to:
• Evaluate the epidemiology and prevention of pertussis
• Assess the efficacy of the pertussis vaccine in preventing clinical pertussis in the current study
• Analyze the attack rate of pertussis in a preschool in the current study
• Distinguish the most common symptoms of pertussis among children in the current study
CME Editor
Thomas J. Gryczan, MS, Technical Writer/Editor, Emerging Infectious Diseases. Disclosure: Thomas J. Gryczan, MS, has disclosed no relevant financial relationships.
CME Author
Charles P. Vega, MD, Clinical Professor of Family Medicine, University of California, Irvine. Disclosure: Charles P. Vega, MD, has disclosed the following financial relationships: served as an advisor or consultant for Lundbeck, Inc.; McNeil Pharmaceuticals; Takeda Pharmaceuticals North America, Inc.
Authors
Disclosures: James Matthias, MPH; P. Scott Pritchard, MPH; Stacey W. Martin, MSc; Cristina Dusek, BSN, RN; Erika Cathey, MPH; Rebecca D’Alessio, BSN; and Marjorie Kirsch, MD, have disclosed no relevant financial relationships.
Abstract
In September 2013, local county health officials in Tallahassee, Florida, USA, were notified of a laboratory-confirmed pertussis case in a 1-year-old preschool attendee. During a 5-month period, 26 (22%) students 1–5 years of age, 2 staff from the same preschool, and 11 family members met the national case definition for pertussis. Four persons during this outbreak were hospitalized for clinical management of pertussis symptoms. Only 5 students, including 2 students with pertussis, had not received the complete series of vaccinations for pertussis. Attack rates in 1 classroom for all students who received the complete series of vaccinations for pertussis approached 50%. This outbreak raises concerns about vaccine effectiveness in this preschool age group and reinforces the idea that recent pertussis vaccination should not dissuade physicians from diagnosing, testing, or treating persons with compatible illness for pertussis.
In the United States, incidence of pertussis is greatest among infants, children 7–10 years of age, and adolescents (1). During 2000–2012, reported pertussis cases increased >6-fold from 7,867 cases to 48,277 cases (2). One potential contributing factor for increased incidence of pertussis is waning immunity after acellular pertussis vaccination (3,4).
In September 2013, the Florida Department of Health in Leon County (DOH–Leon, Tallahassee, FL, USA) was notified of a PCR result positive for Bordetella pertussis for a 1-year-old vaccine-exempt preschool student. Treatment, chemoprophylaxis, and pertussis education were provided to household close contacts, classmates, parents of classmates, and staff associated with the classroom for this student. The investigation identified a 3-year-old sibling who had illness clinically compatible with pertussis before onset of pertussis in the 1-year-old student. This sibling did not attend the preschool.
On December 11, DOH–Leon received a report that a 1-month-old infant had a PCR result positive for pertussis. A public health investigation determined that the vaccinated 3-year-old sibling and mother of this infant had illnesses clinically compatible with pertussis and disease onset before that of the infant. The sibling attended the same preschool as the initial 1-year-old student, and the mother was a substitute teacher at the preschool. Less than 1 week later, another 3-year-old child who attended the preschool showed a PCR result positive for pertussis.
In response to these reports, an outbreak investigation was initiated. On December 19, the local DOH staff conducted a site visit to the preschool to prevent further transmission and determine the incidence of pertussis among students, household close contacts, and staff.
The preschool had 117 students 10 months–6 years of age and 26 staff. On December 19, the preschool director and staff were queried about any additional students or staff members with a cough illness. At this time, the preschool instituted school wide cough illness surveillance and reported any new cough illnesses to DOH. For newly identified case-patients and their contacts, treatment and prophylaxis were administered according to guidelines from the Centers for Disease Control and Prevention (CDC; Atlanta, GA, USA). All symptomatic persons, or their parents if they were <18 years of age, were interviewed by using a standardized case report form to record onset dates, demographics, symptoms, healthcare visits, laboratory testing, treatment, and vaccination status. Pertussis vaccination status for case-patients and all students was cross-referenced in the Florida immunization registry and with preschool vaccination records.
The Council of State and Territorial Epidemiologists (CSTE) 2013 case definition for pertussis was used to classify cases, with the exception that serologic analysis performed at CDC was considered a confirmatory laboratory test (5). In addition, persons with only school-based epidemiologic links to a laboratory-confirmed case were classified as probable cases, rather than confirmed cases. All other epidemiologic links, such as household links, were classified as confirmed per CSTE case definition. One residual nasopharyngeal specimen that showed a PCR result positive for pertussis at a commercial laboratory and 3 serum samples collected retrospectively from consenting case-patients (adult) were forwarded to CDC for confirmation of B. pertussis infection.
On January 7–8, 2014, DOH staff administered an onsite cough illness questionnaire to student and staff households (completion rate 98%). The questionnaire sought to capture any cough illness, classic symptoms of pertussis, and duration of illness since December 1, 2013. All but 3 student households and 1 staff member household completed the cough illness questionnaire. Case data were analyzed for several factors, including age, classroom, number of vaccinations, duration from most recent vaccination to symptom onset, and case classification status.
Vaccine effectiveness was calculated as (1 – relative risk) × 100 for the cohort of children attending the preschool (6). Relative risk was defined as the attack rate (AR) in fully vaccinated children divided by the AR in children whose vaccination status was not up to date. All children attending the preschool were age-eligible to have received >3 doses of pertussis vaccine (DTaP). Children <18 months of age were considered fully vaccinated if they had received 3 doses of DTaP. All other children were considered fully vaccinated if they had received >4 doses of DTaP.
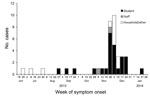
Eleven cases were detected during September 2013–January 2014 and classified as confirmed: 5 laboratory confirmed (3 by PCR specific for IS481; 1 by PCR specific for IS481, HIS1001, PIS1001I, ptxS1, and RNaseP; and 1 by serologic testing at CDC) and 6 epidemiologically linked household contacts (Figure). Twenty-eight cases were classified as probable (total of 39 confirmed and probable cases). Twenty-six students 1–5 years of age (AR 22%) and 2 staff (AR 7%) were identified as having pertussis (Table). The remaining 11 case-patients were linked to the preschool: 9 were household contacts and 2 were camp counselors who had contact with a sibling of a laboratory-confirmed case-patient who attended the preschool.
Four case-patients 1 month–2 years of age, including 2 students, were hospitalized. Only 1 of the hospitalized case-patients had received >3 doses of pertussis vaccine (2 children were too young for 3 doses and 1 child who had received 1 dose was on a delayed schedule). Lengths of hospitalization ranged from 1 to 5 days, and duration of cough ranged from 14 to 50 days. Three of the four hospitalized case-patients had posttussive vomiting, 2 had paroxysmal cough, 2 had inspiratory whoop, and 1 had stridor.
All 39 case-patients had a cough illness for >2 weeks, which is consistent with the CSTE case definition (5). The average duration of illness for all case-patients was estimated to be 23 days, which is an underestimate because 25 case-patients were still symptomatic at time of last interview. Fourteen (54%) of 26 students and 11 (85%) of 13 case-patients who were not students had additional symptoms consistent with pertussis (Table). A paroxysmal cough (56%) was the most common additional symptom identified.
Only 5 of 117 children in the preschool had not received the complete series of vaccinations. Of these 5 children, 2 were case-patients and both had received >1 dose of pertussis vaccine: the hospitalized 1-year-old child who had received a single dose and a 3-year-old child who had received only 2 doses. The other 3 children were unvaccinated but did not have pertussis.
Of the 33 children who had pertussis, 28 had received >3 pertussis vaccinations, and 23 had received >4 vaccinations. Vaccine effectiveness among children attending the preschool was estimated to be 45.0% (95% CI −70.4% to 82.2%). The average number of days from last vaccination to onset of symptoms for the students was only 667 days (≈22 months), and 7 (27%) children had been vaccinated within the previous year.
ARs among students by classroom ranged from 0% to 47%; 6 classrooms had an AR >20% for students. The 2 classrooms with 3-year-old students had the highest ARs. Moreover, an AR of 48% was identified in 1 of these classrooms in which all 17 students had received the complete series of vaccinations. This classroom had an infectious staff member with laboratory-confirmed pertussis during the outbreak. Children 2–3 years of age were at 2.2 times (95% CI 1.0–4.9 times) greater risk for pertussis infection than children 4–6 years of age. When this analysis was restricted to only children 3 years of age, the risk increased to 2.5 times (95% CI 1.1–5.5 times).
This investigation highlights an outbreak of pertussis in a preschool with few vaccine exemptions. To our knowledge, sustained transmission of pertussis in a vaccinated cohort of 1–5-year-old children has not been reported in the United States. Short-duration vaccine effectiveness estimates for children receiving >3 doses of acellular pertussis vaccines have been described at >80% (4,7,8). Although the small number of nonvaccinated children in the preschool resulted in a vaccine effectiveness that had extremely wide CIs that overlap 0, the low estimate, coupled with documented sustained transmission over a period of months, raises concerns about inadequate protection against pertussis in an age group believed to be well protected by acellular pertussis vaccination.
Poor performance of a vaccine in a defined cohort might suggest a provider-level failure to store, use, and administer the vaccine properly. Although we did not assess vaccine storage and handling practices, children from this investigation were seen by multiple providers in the community. Moreover, no general increase in reported pertussis incidence was observed in the county at the same time as this outbreak.
Although the number of cases confirmed by laboratory testing was low (13%), many of the case-patients had substantial illness consistent with pertussis. In addition, B. pertussis infections were confirmed in persons from 5 households over a 5-month period. Confirmatory laboratory testing at CDC provided further evidence of a B. pertussis outbreak. The 3 nonvaccinated students in whom illness did not develop were not assessed for prior evidence of infection.
The use of a focused cough illness questionnaire for case ascertainment might have captured cough illnesses that met the case definition, but might not have been pertussis, during December 1, 2013–January 7, 2014. However, because the questionnaire focused on this narrow period, to minimize recall bias, additional cases of pertussis before December 1, 2013, might not have been identified.
Over the course of the outbreak, mass prophylaxis was provided only to the classroom with 1-year-old children after laboratory confirmation of the first reported case because of concerns about the risk for severe pertussis in this younger age group. Given the limited laboratory testing early during the outbreak and to be consistent with adherence to CDC guidelines, classroom-wide chemoprophylaxis was not provided for laboratory-confirmed cases in older children. Chemoprophylaxis was provided to household and high-risk contacts. As a result, effects of school-wide or classroom-wide chemoprophylaxis were not assessed. No staff were identified as being pregnant during the outbreak. However, early during the outbreak, the 1-year-old child of a rotating staff member (use of postexposure prophylaxis by this staff member was not known) was hospitalized with laboratory-confirmed pertussis in December, despite use of prophylaxis in September by the class in which the mother worked.
As part of this investigation, it was apparent that many physicians were hesitant to provide a diagnosis of pertussis and did not test for this disease, given the recent vaccination history of the patients and despite reporting of an ongoing laboratory-confirmed pertussis outbreak. The spectrum of illness for pertussis in vaccinated children can vary widely and is often mild, with few classic symptoms of pertussis (9). Hesitation by providers in reporting presumptive pertussis delays public health response to prevent continued transmission of pertussis in the community. Thus, recent pertussis vaccination should not preclude diagnosis, testing, and treatment of presumptive pertussis cases (10).
Although all children in the classroom had received the complete series of vaccinations for pertussis, the classroom with the highest AR was one in which a teacher with a laboratory-confirmed case of pertussis who had not received a Tdap booster vaccination, worked throughout her illness. Three of the students showed symptom onset before the teacher, and 5 students, including 1 laboratory-confirmed case-patient, had symptom onset 6–13 days after the teacher showed symptom onset. Improved efforts toward early diagnosis and appropriate treatment to mitigate transmission and booster vaccinations for adults in situations in which prolonged close contact between children, especially children <1 year of age, and adults occur could be considered (11).
Reports of genetic changes in circulating B. pertussis have raised concern that this organism could be adapting to vaccine-induced immunity (12,13). Given these reports and the increased levels of circulation of pertussis among older age groups with documented waning of immunity, further monitoring of acellular pertussis vaccine performance in preschool-age children is necessary to determine if this outbreak was an isolated finding or possibly identification of an emerging epidemiologic trend.
Mr. Matthias is an epidemiology and surveillance unit manager at the Florida Department of Health, Talahassee, Florida. His research interests include applied public health epidemiology, communicable disease prevention, and outbreak response.
Acknowledgments
We thank the preschool director, staff, and student families for their participation, assistance, and general curiosity, and laboratory staff at CDC, especially Maria Lucia Tondella, Jennifer Liang, Pam Cassiday, and Lucia Pawloski, for their assistance in confirming patient samples and expert guidance on pertussis.
This study was supported by an Epidemiology Laboratory and Capacity Cooperative Agreement with the Centers for Disease Control and Prevention.
References
- Centers for Disease Control and Prevention. 2012 final pertussis surveillance report. April 9, 2014 [cited 2015 Oct 26]. http://www.cdc.gov/pertussis/downloads/pertuss-surv-report-2012.pdf
- Centers for Disease Control and Prevention. Pertussis cases by year (1922–2013). April 9, 2014 [cited 2015 Oct 26]. http://www.cdc.gov/pertussis/surv-reporting/cases-by-year.html
- Tartof SY, Lewis M, Kenyon C, White K, Osborn A, Liko J, Waning immunity to pertussis following 5 doses of DTaP. Pediatrics. 2013;131:e1047–52. DOIGoogle Scholar
- Quinn HE, Snelling TL, Macartney KK, McIntyre PB. Duration of protection after first dose of acellular pertussis vaccine in infants. Pediatrics. 2014;133:e513–9. DOIGoogle Scholar
- Centers for Disease Control and Prevention. National notifiable disease surveillance system (NNDSS): pertussis (whooping cough) (Bordetella pertussis) 2010 case definition. April 9, 2014 [cited 2015 Oct 26]. http://wwwn.cdc.gov/NNDSS/script/casedef.aspx?CondYrID=795&DatePub=1/1/2010%2012:00:00%20AM
- Jekel JF, Katz DL, Elmore JG, Wild DM. Methods of primary prevention: specific protection. In: Epidemiology, biostatistics, and preventive medicine. 3rd ed. Philadelphia: Saunders-Elsevier; 2007. p. 253–66.
- Misegades LK, Winter K, Harriman K, Talarico J, Messonnier NE, Clark TA, Association of childhood pertussis with receipt of 5 doses of pertussis vaccine by time since last vaccine dose, California, 2010. JAMA. 2012;308:2126–32. DOIGoogle Scholar
- Bisgard KM, Rhodes P, Connelly BL, Daoling B, Hahn C, Patrick S, Pertussis vaccine effectiveness among children 6 to 59 months of age in the United States, 1998–2001. Pediatrics. 2005;116:e285–94. DOIGoogle Scholar
- Tozzi AE, Ravà L. Ciofi degli Atti ML, Salmaso S; Progetto Pertosse Working Group. Clinical presentation of pertussis in unvaccinated and vaccinated children in the first six years of life. Pediatrics. 2003;112:1069–75. DOIGoogle Scholar
- Deeks S, De Serres G, Boulianne N, Duval B, Rochette L, Déry P, Failure of physicians to consider the diagnosis of pertussis in children. Clin Infect Dis. 1999;28:840–6. DOIGoogle Scholar
- Forsyth KD, Campins-Marti M, Caro J, Cherry JD, Greenberg D, Guiso N, New pertussis vaccination strategies beyond infancy: recommendations by the global pertussis initiative. Clin Infect Dis. 2004;39:1802–9. DOIGoogle Scholar
- Mooi FR, Van Deer Maas NA, De Melker HE. Pertussis resurgence: waning immunity and pathogen adaptation–two sides of the same coin. Epidemiol Infect. 2014;142:685–94. DOIGoogle Scholar
- Pawloski LC, Queenan AM, Cassiday PK, Lynch AS, Harrison MJ, Shang W, Prevalence and molecular characterization of pertactin-deficient Bordetella pertussis in the United States. Clin Vaccine Immunol. 2014;21:119–25. DOIGoogle Scholar
Figure
Table
Follow Up
Earning CME Credit
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions (with a minimum 75% passing score) and earn continuing medical education (CME) credit, please go to http://www.medscape.org/journal/eid. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.org. If you are not registered on Medscape.org, please click on the “Register” link on the right hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider, CME@medscape.net. For technical assistance, contact CME@webmd.net. American Medical Association’s Physician’s Recognition Award (AMA PRA) credits are accepted in the US as evidence of participation in CME activities. For further information on this award, please refer to http://www.ama-assn.org/ama/pub/about-ama/awards/ama-physicians-recognition-award.page. The AMA has determined that physicians not licensed in the US who participate in this CME activity are eligible for AMA PRA Category 1 Credits™. Through agreements that the AMA has made with agencies in some countries, AMA PRA credit may be acceptable as evidence of participation in CME activities. If you are not licensed in the US, please complete the questions online, print the certificate and present it to your national medical association for review.
Article Title: Sustained Transmission of Pertussis in Vaccinated, 1–5-Year-Old Children in a Preschool, Florida, USA
CME Questions
1. You are evaluating a 2-year-old girl for a cough that she has had for the past 2 weeks. The cough is described as a bark, and it may be paroxysmal. The child has had no fever and had some nasal congestion 10 days ago. You consider that this patient may have pertussis. What should you consider regarding the epidemiology and prevention of pertussis?
A. Pertussis is most common among children younger than 4 years, but it is rarely found in school-aged children
B. The prevalence of pertussis increased more than 6-fold from 2000 to 2012
C. Pertussis vaccination is associated with long-term immunity without a waning phenomenon
D. Vaccine efficacy for the pertussis vaccine is 99% to 100%
2. Your patient is up to date with her pertussis vaccine. What should you consider regarding pertussis vaccine data in the current study?
A. Nearly half of children in the affected preschool were not up to date for pertussis vaccination
B. All unvaccinated children had clinical cases of pertussis
C. Less than 5% of children in the affected preschool were vaccinated in the past year
D. The highest attack rate was encountered in the classroom of a teacher who had not received a tetanus-diphtheria-acellular pertussis (Tdap) booster
3. The patient's mother is concerned that her daughter acquired pertussis at her daycare facility. What should you consider regarding the attack rate for pertussis in the current study?
A. The attack rate was less than 20% in all classrooms
B. The highest attack rate was found in the classrooms with 3-year-old students
C. The attack rate was 0% in the classroom with all students up to date on pertussis vaccination
D. The greatest risk for pertussis infection was among 4- to 6-year-old children
4. You initiate testing for pertussis as well as treatment, but the diagnosis is not confirmed. What was the most common symptom besides a prolonged cough in the current study?
A. Paroxysmal cough
B. Apnea
C. Inspiratory whoop
D. Post-tussive vomiting
Activity Evaluation
|
1. The activity supported the learning objectives. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
2. The material was organized clearly for learning to occur. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
3. The content learned from this activity will impact my practice. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
|
4. The activity was presented objectively and free of commercial bias. |
||||
|
Strongly Disagree |
|
|
|
Strongly Agree |
|
1 |
2 |
3 |
4 |
5 |
Related Links
Table of Contents – Volume 22, Number 2—February 2016
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
James Matthias, Bureau of Communicable Diseases, Florida Department of Health, 4042 Bald Cypress Way, Bin A-08, Tallahassee, FL 32399, USA
Top