Hepatitis C
CDC Yellow Book 2024
Travel-Associated Infections & DiseasesINFECTIOUS AGENT: Hepatitis C virus
ENDEMICITY
Worldwide
Regions of high prevalence in Africa, central, southern, and eastern Asia, and eastern Europe
TRAVELER CATEGORIES AT GREATEST RISK FOR EXPOSURE & INFECTION
PREVENTION METHODS
Practice bloodborne pathogen precautions
Use safe injection practices and syringe service programs
DIAGNOSTIC SUPPORT
Infectious Agent
Hepatitis C virus (HCV) is a spherical, enveloped, positive-strand RNA virus. Seven distinct HCV genotypes and 67 subtypes have been identified, the distribution of which vary geographically worldwide.
Transmission
HCV transmission is bloodborne and most often involves exposure to contaminated needles or syringes, or receipt of blood or blood products that have not been screened for HCV. Although infrequent, HCV can be transmitted through other procedures that involve blood exposure (e.g., tattooing, during sexual contact, perinatally from mother to child).
Epidemiology
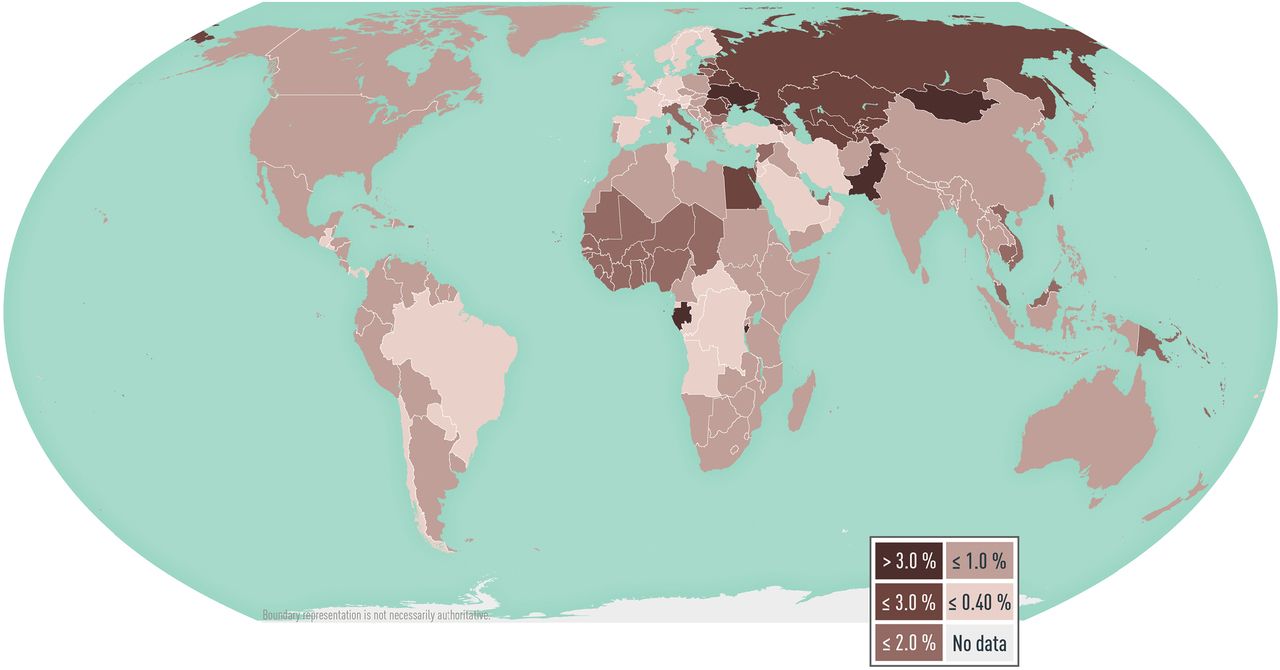
Globally, an estimated 62 million people were living with HCV infection (chronically infected) in 2019. Although the quality of epidemiologic data and prevalence estimates vary widely across countries and within regions, the most recent global estimates from 2019 indicate that the viremic prevalence of HCV infection (prevalence of HCV RNA) is <1.0% in most developed countries, including the United States (Map 5-08). HCV prevalence is considerably higher in some countries in eastern Europe (3.1% in Ukraine, 2.9% in Russia, 2.9% in Moldova, 2.5% in Romania, 2.1% in Latvia) and certain countries in Africa (5.9% in Gabon, 3.6% in Burundi, 2.1% in Egypt), the Middle East (1.6% in Syria), and the South Caucasus and Central Asia (3.1% in Georgia, 3.0% in Uzbekistan, 2.7% in Tajikistan, 2.7% in Turkmenistan).
The most frequent mode of transmission in most high-, middle-, and low-income countries is sharing of drug preparation and drug-injection equipment. In countries where infection-control practices are poor, a predominant transmission mode is from unsafe injections and other health care exposures. Travelers’ risk of contracting HCV infection is generally low, but they should exercise caution and avoid non-urgent dental or medical procedures, particularly in high-prevalence areas. Activities that can result in blood exposure include receiving blood transfusions that have not been screened for HCV; undergoing dental or medical procedures; participating in activities in which equipment has not been adequately sterilized or disinfected, or in which contaminated equipment is reused (e.g., acupuncture, injection drug use, shaving, and tattooing); and working in health care fields (dental, laboratory, or medical) that entail direct exposure to human blood.
Map 5-08 Worldwide prevalence of hepatitis C viremia, 2019
Disease data source: 2019 estimates of hepatitis C virus disease burden. CDA Foundation Polaris Observatory. Available from https://cdafound.org/polaris-countries-distribution/.
Clinical Presentation
HCV infection is a major cause of cirrhosis and liver cancer and is the leading reason for liver transplantation in the United States. Most (80%) people with acute HCV infection have no symptoms. When they occur, symptoms are indistinguishable from other forms of acute viral hepatitis and could include abdominal pain, anorexia and nausea, fatigue, jaundice, and dark urine. Among infected people, over half will remain chronically infected unless treated with antiviral medications. Cirrhosis develops in ≈10%–20% of people after 20–30 years of chronic infection, and progression is often clinically silent; evidence of liver disease might not occur until late in the course of the disease.
Diagnosis
In the United States, hepatitis C is a nationally notifiable disease. Hepatitis C testing is required for diagnosis. Testing is not routinely performed in many countries, however, and most HCV-infected people are unaware of their infection. Two types of tests are available: IgG assays for HCV antibodies, and nucleic acid amplification tests to detect HCV RNA in blood (viremia). Both tests are commercially available in the United States and most countries. IgM assays, to detect early or acute infection, are not available. Because a positive HCV antibody test cannot discriminate between a previously infected person who resolved or cleared the infection and someone with current infection, be certain that HCV RNA testing follows a positive HCV antibody test to identify people with current (recent and chronic) HCV infection.
In 2020, CDC updated recommendations to include ≥1 hepatitis C screening test for all adults ≥18 years of age during a lifetime, and hepatitis C screening for all pregnant women during each pregnancy. See information on how to obtain hepatitis C diagnostic support from the Centers for Disease Control and Prevention (CDC), including contact information, which samples to send, and how to send samples is available at or by calling 800-CDC-INFO (800-232-4636).
Treatment
Since 2014, several new all-oral direct-acting antiviral agents have been approved for use to treat hepatitis C. These new regimens require only 8–12 weeks of treatment, have few side effects, and eliminate HCV infection in ≈95% of people who complete treatment, regardless of HCV genotype, prior treatment status, HIV co-infection, and the presence of cirrhosis. See treatment guidelines, which are updated frequently.
Prevention
No vaccine or postexposure prophylaxis is available to prevent HCV infection, nor does immune globulin provide protection. Travelers should check with their health care provider to understand the potential risk for infection and any precautions they should take. If seeking dental or medical care, travelers should be alert to the use of instruments, tools, and other equipment that has not been adequately sterilized or disinfected; reuse of contaminated equipment; and unsafe injection practices (e.g., reuse of disposable needles and syringes). People who travel to undergo dental, medical, or surgical procedures should be cognizant of potential HCV exposure (see Sec. 6, Ch. 4, Medical Tourism).
HCV and other bloodborne pathogens can be transmitted when medical instruments are not sterile or providers do not follow proper infection-control procedures (e.g., washing hands, using latex gloves, cleaning and disinfecting surfaces and instruments). In some parts of the world (e.g., parts of sub-Saharan Africa), blood donors might not be screened for HCV infection. Advise travelers to avoid body piercing, tattooing, being shaved by a barber, or having an elective dental or medical procedure in destinations where adequate sterilization or disinfection practices might not be used. Furthermore, instruct travelers to seek testing for HCV infection upon return if they received blood transfusions or sustained blood exposures for which they could not assess the risks, and to seek immediate medical care if they have signs or symptoms of acute hepatitis.
CDC website: Hepatitis C
The following authors contributed to the previous version of this chapter: Philip Spradling