Influenza
CDC Yellow Book 2024
Travel-Associated Infections & DiseasesINFECTIOUS AGENT: Influenza virus
ENDEMICITY
Worldwide
TRAVELER CATEGORIES AT GREATEST RISK FOR EXPOSURE & INFECTION
PREVENTION METHODS
Influenza is a vaccine-preventable disease
Practice hand hygiene
Use appropriate personal protective equipment
Use postexposure antiviral medication
DIAGNOSTIC SUPPORT
Infectious Agent
Influenza is caused by infection of the respiratory tract with influenza viruses, RNA viruses of the Orthomyxovirus genus. Influenza viruses are classified into 4 types: A, B, C, and D. Influenza A and B viruses commonly cause illness in humans and seasonal epidemics. Influenza A viruses are classified into subtypes based on the surface proteins hemagglutinin (HA) and neuraminidase (NA). Two influenza A virus subtypes, A(H1N1) and A(H3N2), and 2 influenza B virus lineages, B-Yamagata and B-Victoria, co-circulate in humans worldwide; the distribution of these viruses varies year to year and between geographic areas and time of year. Information about circulating seasonal viruses in various regions can be found on the Centers for Disease Control and Prevention (CDC) website or World Health Organization website.
Influenza type C infections generally cause mild illness and are not thought to cause human influenza epidemics. Influenza D viruses primarily affect cattle and are not known to infect or cause illness in people.
Novel influenza refers to viruses with a subtype different from seasonal influenza, and usually is caused by influenza A viruses that circulate among animals. Notably, avian influenza A(H5N1), A(H5N6), A(H7N9), and A(H9N2) viruses, and swine-origin variant viruses A(H1N1)v, A(H1N2)v, and A(H3N2)v have resulted in novel human influenza infections globally. An influenza virus that normally circulates in swine (but not people) but then is detected in a person is called a variant virus and is denoted with the letter v.
Transmission
Influenza viruses spread from person to person, primarily through respiratory droplets (e.g., when an infected person coughs or sneezes near a susceptible person). Transmission generally occurs via large particle droplets that require close proximity (≤6 feet) between the source and the recipient, but airborne transmission via small particle aerosols can occur within confined air spaces. Indirect transmission occurs when a person touches their face after touching a virus-contaminated surface (fomite).
The incubation period is usually 1–4 days after exposure. Most adults ill with influenza shed the virus in the upper respiratory tract and are infectious from the day before symptom onset to ≈5–7 days after symptom onset. Infectiousness is greatest within 3–4 days of illness onset and is correlated with fever. Children, immunocompromised people, and severely ill people might shed influenza virus for ≥10 days after symptom onset. Those who are asymptomatic can still shed the virus and infect others. Seasonal influenza viruses are rarely detected in blood or stool.
Influenza A virus transmission from animals to humans is rare but possible. Infected birds shed influenza virus in their droppings, mucus, and saliva, and transmission to humans can occur from direct contact with an animal (by touching an infected animal or by droplet spread) or contact with a sick animal’s environment (by inhalation of airborne viruses or through fomite transmission). See CDC’s Avian Influenza A Virus Infection in Humans website for more details. Infected swine shed the virus in nasal secretions and can transmit viruses to humans in the same way seasonal influenza viruses spread among people. For more information, see CDC’s website What People Who Raise Pigs Need to Know about Influenza.
Epidemiology
Seasonal Influenza
Influenza seasonality varies geographically. The risk for influenza exposure during travel depends on the time of year and destination. In temperate regions, influenza epidemics are more common during cooler months, October–March in the Northern Hemisphere and April–September in the Southern Hemisphere. In subtropical and tropical regions, seasonal influenza epidemics follow a similar pattern, but influenza illnesses can occur throughout the year. During the coronavirus disease 2019 pandemic in 2020 and 2021, there was a sharp decrease in global influenza activity. Although causality has not confirmed, the decrease has been attributed, in part, to community and personal implementation of nonpharmaceutical interventions to mitigate severe acute respiratory syndrome coronavirus 2 transmission.
CDC estimates that 9–45 million (symptomatic) illnesses, 4–21 million outpatient visits, 140,000–810,000 hospitalizations, and 12,000–61,000 deaths associated with influenza occur each year in the United States. Globally, annual influenza epidemics result in an estimated 3–5 million cases of severe illness and 290,000–650,000 respiratory deaths.
At-Risk Populations
Certain groups are at increased risk for influenza complications (see Box 5-03). The incidence of influenza illness is greatest among children, especially those aged 0–4 years, and adults aged 50–64 years. Rates of hospitalization (a marker of severe illness) and death due to influenza are typically higher among older adults (≥65 years old) followed by adults aged 50–64 years, children aged <2 years, and people of any age with underlying medical conditions that place them at increased risk for complications.
Box 5-03 Groups at increased risk for influenza complications
- Adults ≥65 years old
- Children <2 years old; although all children <5 years are considered at increased risk for serious influenza complications, the highest risk is for those <2
- Pregnant people and people ≤2 weeks post-partum
- People with certain medical conditions, including asthma, blood disorders, body mass index ≥40, chronic lung disease, endocrine disorders, heart disease, immunocompromise due to disease or medication, kidney disease, liver disorders, metabolic disorders, neurologic and neurodevelopment conditions, and history of stroke
- American Indians and Alaska Natives
- People living in nursing homes and other long-term care facilities
Zoonotic Influenza
Influenza A viruses circulate among animal populations and occasionally infect humans. The primary reservoirs for influenza A viruses are wild birds, like waterfowl, but influenza A viruses are also common in domestic poultry and swine populations. Influenza A viruses can infect other animal species (e.g., bats, cats, dogs, ferrets, horses, sea lions, seals).
In the United States, the last large outbreak of H5 lineage avian influenza virus in birds occurred in 2022. Although 34 different avian influenza subtypes have been reported globally since 2005, 94% of outbreaks reported in birds were caused by H5 lineage viruses. Since 2005, 23,754 outbreaks of H5 lineage avian influenza in animals have been reported from 97 countries (Map 5-09). Avian influenza virus outbreaks do not have to be reported to the World Organisation for Animal Health (OIE) if the virus is endemic in a country; avian influenza A(H5N1) was declared endemic in Indonesia in 2006 and in Egypt in 2008. Swine influenza is not reportable to OIE.
Map 5-09 Global distribution of highly pathogenic avian influenza A (H5Nx) virus1
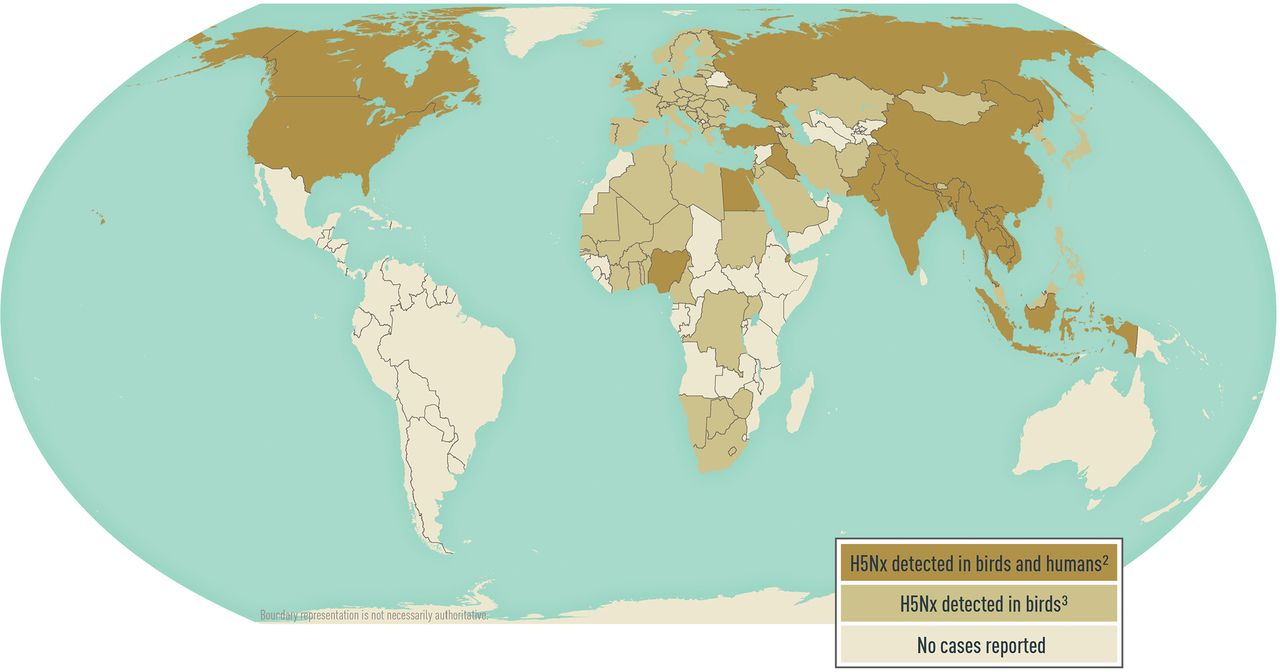
1Disease data source: Animal disease events. World Organisation for Animal Health, World Animal Health Information System (OIE-WAHIS). Available from: https://www.woah.org/en/what-we-do/animal-health-and-welfare/disease-data-collection/.
2H5Nx lineages include: H5N1, H5N6, and H5N8
3H5Nx lineages include: H5N1, H5N2, H5N3, H5N4, H5N5, H5N6, H5N8, H5N9, and H5Nx
Novel Influenza A Viruses
Human infections with novel influenza A viruses are uncommon, but potentially could cause a pandemic if sustained human-to-human transmission occurs. Human infections with influenza A(H1N1)v, A(H1N2)v, and A(H3N2)v have been identified in the United States; the largest variant influenza outbreak occurred in 2012 and had a total of 309 infections and 1 death associated with an A(H3N2)v virus.
From 2011 through July 2021, 468 human infections with variant influenza viruses were identified in 24 US states. Most people identified with variant virus infections reported contact with swine preceding their illness, suggesting swine-to-human transmission. Limited cases of human-to-human transmission of variant viruses have also been reported. Seasonal human influenza viruses have infected swine, suggesting person-to-swine transmission. Agricultural fairs and swine farms are settings in which humans are exposed to swine. Illnesses associated with variant virus infections are usually mild, with symptoms similar to those of seasonal influenza.
Avian Influenza A(H5) Lineage Viruses
Avian influenza viruses do not commonly infect humans, but cases are reported globally each year. During 2013–2020, 9 countries reported 281 human illnesses caused by avian influenza A(H5) lineage viruses, and a reported case-fatality ratio of ≈37% (Map 5-09). Most disease from A(H5) lineage viruses occurred after direct or close contact with sick or dead infected poultry. A(H5N1) and A(H5N6) viruses are widespread among poultry in some countries in Asia and the Middle East. Egypt and Indonesia account for 79% of A(H5N1) infections, and China accounts for 99% of A(H5N6) infections in humans globally. Instances of limited human-to-human A(H5N1) virus transmission have been reported. In February 2021, Russia reported 7 cases of human, asymptomatic infection with A(H5N8), the first report of influenza A(H5N8) infection in humans.
Avian Influenza A(H7N9) Virus
Avian influenza A(H7N9) virus emerged in China in 2013; as of July 2021, it has caused 1,568 confirmed human infections. Most cases have been identified in mainland China, but several infections have been identified in Hong Kong Special Administrative Region (SAR), Macau SAR, Malaysia, and Taiwan, in travelers who reported exposure in mainland China. In 2014, Canada reported the first known imported influenza A(H7N9) virus infection in North America in a traveler returning from China. Most people with A(H7N9) infection were exposed to infected poultry or contaminated environments (e.g., live bird markets). The virus has been found in poultry and environmental samples collected in China. Most reported human A(H7N9) infections have been severe respiratory illnesses; the reported case-fatality ratio is 40%.
Other Avian Influenza Viruses
Although uncommon, human infections with other avian influenza viruses, including A(H7N2), A(H7N3), and A(H9N2), have been reported globally in recent years, including 2 cases of A(H7N2) in humans exposed to infected cats in New York in 2016. And even though human infections with avian influenza viruses in the United States are rare, surveillance in domestic birds and people exposed to infected birds abides because of the low, but continued, risk for transmission to humans.
Clinical Presentation
Physical Findings
Uncomplicated influenza illness, the most common presentation of seasonal influenza, is characterized by an abrupt onset of signs and symptoms that include nonproductive cough, fever, headache, malaise, muscle aches, rhinitis, sore throat, and vomiting, and, less commonly, rash. Illness without fever can occur, especially in older adults and infants. Children are more likely than adults to experience nausea, vomiting, or diarrhea when ill with influenza.
Physical findings are predominantly localized to the respiratory tract and include nasal discharge, pharyngeal inflammation without exudates, and occasionally rales on chest auscultation. Influenza illness typically resolves within 1 week for most previously healthy children and adults who do not receive antiviral medication, although cough and malaise can persist for >2 weeks, especially in older adults.
Humans infected with variant influenza viruses have a clinical presentation like seasonal influenza virus infections. Reported human infections with avian influenza A(H5N1) or A(H7N9) viruses often have severe pneumonia or respiratory failure and a high case-fatality ratio. These data might be skewed, however, because people with less severe illness often do not seek care for influenza or get tested for avian origin A(H5) or A(H7) viruses.
Complications
Complications of influenza virus infection include primary influenza viral pneumonia and secondary bacterial pneumonia; also, co-infections with other viral or bacterial pathogens, encephalopathy, exacerbation of underlying medical conditions (e.g., cardiac disease, pulmonary disease), Guillain-Barré syndrome (GBS), myocarditis, myositis, parotitis, seizures, and rarely, death.
Diagnosis
Influenza can be difficult to distinguish from respiratory illnesses caused by other pathogens based on signs and symptoms alone. The positive predictive value of clinical signs and symptoms for influenza-like illness (fever with either cough or sore throat) for laboratory-confirmed influenza virus infection is 30%–88%, depending on host factors (e.g., age, community influenza activity levels).
Diagnostic Testing
Consider diagnostic testing for hospitalized patients with suspected influenza; patients for whom a diagnosis of influenza will inform clinical care decisions, including patients who do not improve on antiviral therapy and those with medical conditions that place them at increased risk for complications; and patients for whom results of influenza testing would affect infection control or management of close contacts, including other patients, such as in institutional outbreaks or other settings (e.g., cruise ships, tour groups).
For clinicians seeking laboratory confirmation of influenza, the Infectious Diseases Society of America recommends the use of rapid molecular assays in outpatients and nucleic acid amplification tests (e.g., reverse transcription PCR [RT-PCR]), in hospitalized patients. For suspected human infection with a novel influenza A virus of animal origin (e.g., avian influenza A virus, swine influenza A virus), contact the local and state health departments to perform RT-PCR for seasonal influenza viruses and novel influenza A viruses.
Other diagnostic tests available for influenza include antigen-based rapid influenza diagnostic tests, immunofluorescence assays, and viral culture. Most patients with clinical illness consistent with uncomplicated influenza in communities where influenza viruses are circulating do not require diagnostic testing for empiric clinical management.
Test Sensitivity
Nucleic acid assays are the most sensitive diagnostic assays. Thus, if infection with these viruses is suspected, contact the state health department in the United States or CDC outside the United States. Do not delay starting antiviral treatment while waiting for confirmatory laboratory testing results.
The sensitivity of antigen-based rapid influenza diagnostic tests varies but is substantially lower than RT-PCR or viral culture. Antigen-based rapid influenza diagnostic tests cannot distinguish between seasonal influenza A virus infections and animal-origin influenza A virus infections, and their sensitivity to detect these animal-origin influenza viruses can vary by test type and virus subtype. Therefore, a negative antigen-based rapid influenza diagnostic test result does not rule out influenza virus infection, and health care providers should not rely on a negative antigen-based rapid influenza diagnostic test result to make decisions about treatment.
Treatment
Antiviral Treatment
Early antiviral treatment (see Table 5-13) can shorten the duration of fever and other symptoms and reduce the risk for complications from influenza. Antiviral treatment is recommended as early as possible for any patient with confirmed or suspected influenza who is hospitalized; has severe, complicated, or progressive illness; or who is at increased risk for influenza-associated complications.
Treatment is most effective if it can be initiated ≤48 hours of symptom onset. For hospitalized patients, those with severe illness, or those at higher risk for complications, antiviral therapy might still be beneficial if started >48 hours after illness onset. Four antiviral agents are approved by the US Food and Drug Administration (FDA) for the treatment and prophylaxis of influenza: oral oseltamivir, available as a generic (or as Tamiflu, Genentech); intravenous peramivir (Rapivab, BioCryst Pharmaceuticals); inhaled zanamivir (Relenza, GlaxoSmithKline); and oral baloxavir (Xofluza, Genentech).
Oseltamivir is the recommended treatment for people of all ages and is the preferred agent to treat patients with severe or complicated influenza illness who can tolerate oral medications. Peramivir is approved and recommended to treat patients aged ≥2 years and might be useful in patients unable to tolerate or absorb oral antiviral therapy. Zanamivir is approved and recommended to treat patients aged ≥7 years and for prophylaxis in people aged ≥5 years. Inhaled zanamivir is not recommended for use in people with underlying chronic respiratory disease. Baloxavir is indicated to treat acute uncomplicated influenza in patients ≥12 years of age who have been symptomatic for ≤48 hours.
Two other FDA-approved influenza antiviral medications, amantadine and rimantadine, are not recommended for treatment or prophylaxis of influenza because of widespread viral resistance. Discuss antiviral treatment options with people at increased risk for complications of influenza before they travel to areas with influenza activity.
Table 5-13 Treatment and prophylaxis for influenza A & B: approved and recommended antiviral medication dosing schedules
|
ANTIVIRAL |
ROUTE |
USE |
PEDIATRIC DOSE |
ADULT DOSE |
|---|---|---|---|---|
|
Oseltamivir |
Oral (PO) |
Treatment1 |
<1 year old: 3 mg/kg PO, 2×/day ×5 days2 |
75 mg PO 2×/day ×5 days |
|
|
|
Prophylaxis1 |
<3 months old: unless the situation is judged critical, oseltamivir is not recommended due to limited data in this age group |
75 mg PO 1×/day ×7 days |
|
Peramivir |
Intravenous (IV) |
Treatment3 |
2–12 years old: 12-mg/kg dose (up to 600 mg maximum) by IV infusion over ≥15 minutes ×1 |
≥13 years old: 600 mg by IV infusion over ≥15 minutes ×1 |
|
|
|
Prophylaxis4 |
Not recommended |
Not approved |
|
Zanamivir |
Inhaled |
Treatment5 |
≥7 years old: 10 mg (2 5-mg inhalations) 2×/day ×5 days |
|
|
|
|
Prophylaxis |
≥5 years old: 10 mg (two 5-mg inhalations) 1×/day ×7 days |
|
|
Baloxavir |
Oral (PO) |
Treatment6 |
≥12 year old (weight-based dosing schedule): |
|
|
|
|
Postexposure prophylaxis |
≥12 year old (weight-based dosing schedule): |
|
1Oseltamivir is approved by the US Food and Drug Administration (FDA) for the treatment of acute uncomplicated influenza ≤48 hours of illness onset. Although not part of the FDA-approved indications, use of oseltamivir to treat influenza in infants <14 days old, and for prophylaxis in infants 3 months to 1 year of age, is recommended by the Centers for Disease Control and Prevention (CDC) and the American Academy of Pediatrics (AAP).
2AAP provides alternative dosing guidelines for infants aged 9–11 months and for premature infants.
3Peramivir is FDA-approved and recommended for treatment of acute uncomplicated influenza ≤48 hours of illness onset. Daily dosing for a minimum of 5 days was used in clinical trials of hospitalized patients with influenza.
4No data for use of peramivir for influenza chemoprophylaxis are available.
5Zanamivir is FDA-approved and recommended for treatment of acute uncomplicated influenza ≤48 hours of illness onset.
6Baloxavir marboxil is FDA-approved and recommended for treatment of acute uncomplicated influenza ≤48 hours of illness onset.
Postexposure Prophylaxis
To complement hand washing, face covering, and social distancing, antiviral drugs can be used for prophylaxis to prevent infection after close contact with a confirmed case. CDC does not, however, recommend routine use of antiviral medications for prophylaxis except as one of multiple interventions to control institutional influenza outbreaks. Initiate postexposure prophylaxis ≤48 hours of exposure, but never >48 hours, because of the risk of treating infection with a subtherapeutic dose. Alternatively, exposed people can monitor for symptoms and initiate antiviral treatment early after symptoms begin.
CDC recommendations for antiviral use for variant influenza virus infections are like those for seasonal influenza virus infection. CDC recommends antiviral treatment for all suspected cases of human infection with avian influenza viruses. See recommendations for postexposure prophylaxis of close contacts of confirmed human infections of avian influenza A(H5N1) and A(H7N9) viruses.
Consider postexposure prophylaxis for anyone exposed to birds infected with influenza A(H5N1), A(H7N9), A(H5N2), and A(H5N8). The decision to initiate prophylaxis, however, should be based on clinical judgment, with consideration given to the type of exposure and whether the exposed person is at increased risk for complications from influenza. If antiviral prophylaxis is initiated for people exposed to avian influenza A viruses, CDC recommends twice daily treatment dosing for oseltamivir or zanamivir instead of once daily prophylaxis dosing.
Prevention
Vaccines
Vaccination is the most effective way to prevent influenza and its complications. In the United States, CDC recommends annual seasonal influenza vaccination for people aged ≥6 months. Several influenza vaccines are approved for use in the United States and can be grouped into 3 categories: inactivated influenza vaccine (IIV) including cell-based, high-dose, and adjuvanted influenza vaccines; live attenuated influenza vaccine (LAIV); and recombinant influenza vaccine (RIV).
See updates and recommendations. For people for whom >1 type of vaccine is approved, clinicians can provide any category of vaccine. Children aged 6 months–8 years who have never received an influenza vaccine, or who have not previously received a lifetime total of ≥2 doses, require 2 doses of age-appropriate influenza vaccine given ≥4 weeks apart during their first season of vaccination to induce sufficient immune response.
Travelers—including people at increased risk for complications of influenza—who did not receive the current seasonal influenza vaccine and who are traveling to parts of the world where influenza activity is ongoing, should consider influenza vaccination ≥2 weeks before departure.
Administration
IIVs are administered by intramuscular injection and labeled for use in people aged ≥6 months, but specific age indications vary by manufacturer and product; follow label instructions. Cell-based inactivated vaccines are licensed for people aged ≥2 years. High-dose and adjuvanted IIV vaccines, which can elicit higher levels of antibodies than standard-dose vaccines, are available for people aged ≥65 years. LAIV is administered as a nasal spray and is labeled for use in people aged 2–49 years who do not have contraindications. RIV is labeled for use in people aged ≥18 years.
Adverse Reactions
Inactivated Influenza Vaccine
The most frequent side effects of vaccination with IIV in adults are soreness and redness at the vaccination site. These local injection-site reactions are slightly more common with high-dose IIV. Reactions generally are mild and rarely interfere with the ability to conduct usual, daily activities. Fever, headache, malaise, myalgia, and other systemic symptoms sometimes occur after vaccination; symptoms might be more frequent in people with no previous exposure to the influenza virus antigens in the vaccine (e.g., young children) and are generally short-lived.
GBS is associated with influenza-like illness and was associated with the 1976 swine influenza vaccine, which had an increased risk of 1 additional case of GBS per 100,000 people vaccinated. None of the studies of influenza vaccines other than the 1976 influenza vaccine have demonstrated a risk for GBS of similar magnitude. The increased risk for GBS after seasonal influenza vaccines generally is small, ≈1–2 additional cases per 1 million people vaccinated, whereas the estimated risk for GBS after influenza is ≈17.2 cases per 1 million patients hospitalized with influenza.
Live Attenuated Influenza Vaccine
The most frequent side effects of LAIV reported in healthy adults include minor upper respiratory symptoms, runny nose, and sore throat, which are generally well tolerated. Some children and adolescents have reported fever, myalgia, vomiting, and wheezing. These symptoms, particularly fever, are more often associated with the first administered LAIV dose and are self-limited.
Children aged 2–4 years who have a history of wheezing in the past year or who have a diagnosis of asthma should not receive LAIV. People 2–49 years of age who have conditions that increase their risk for severe influenza (e.g., immunocompromising conditions, pregnancy) should receive IIV or RIV, not LAIV. To decrease the risk of transmitting live virus to severely immunocompromised people, their caretakers also should not receive LAIV, or should avoid contact for 7 days after receiving LAIV.
Composition
Influenza vaccine composition can be trivalent, protecting against 3 different influenza viruses (2 influenza A subtypes and 1 influenza type B lineage), or quadrivalent, with protection against 4 different influenza viruses (2 influenza A subtypes and 2 influenza type B lineages). Quadrivalent vaccine includes a representative strain from antigenically distinct B-Yamagata and B-Victoria lineages. All influenza vaccines in the United States are quadrivalent vaccines.
Coverage
No information is available about the benefits of revaccinating people before summer travel who were vaccinated during the preceding fall, and revaccination is not recommended. People at increased risk for influenza complications should consult with their health care provider to discuss the risk for influenza or other travel-related diseases before traveling during the summer.
Seasonal influenza vaccines are not expected to provide protection against human infection with animal-origin novel influenza viruses, including influenza A(H5N1) and A(H7N9) viruses. No commercially available influenza vaccines are available to protect against avian or swine viruses.
Precautions & Contraindications
Influenza vaccine is contraindicated in people who have had a previous severe allergic reaction to influenza vaccine, regardless of which vaccine component was responsible for the reaction. Immediate hypersensitivity reactions (e.g., hives, angioedema, allergic asthma, or systemic anaphylaxis) rarely occur after influenza vaccination. These reactions likely result from hypersensitivity to vaccine components, one of which is residual egg protein. People with a history of egg allergy who have experienced only hives after exposure to eggs can receive any licensed and recommended influenza vaccine for their age and health status. Vaccine options are also available for people with a history of egg allergy with a history of severe reaction to egg.
Personal Protective Measures
Measures that can help prevent influenza virus infection and other infections during travel include avoiding close contact with sick people and washing hands often with soap and water. In places where soap and a safe source of water are not available, CDC recommends using an alcohol- based hand sanitizer containing ≥60% alcohol. Face coverings are effective in preventing the spread of respiratory viruses, particularly among people in confined areas, and might have a role in the prevention of contagion during influenza epidemic periods. An ill person can help prevent the spread of illness to others by covering their nose and mouth with their elbow when coughing and sneezing and avoiding close contact with others. If symptomatic people cannot avoid contact with others, consider having them wear a mask when they are in close contact with others.
The best way to prevent infection with animal-origin influenza viruses, including A(H5N1) and A(H7N9), is to follow standard travel safety precautions, including using good hand hygiene, practicing food safety precautions, and avoiding contact with sources of exposure. Most human infections with animal-origin influenza viruses have occurred after direct or close contact with infected poultry or swine. In destinations where avian influenza virus outbreaks are occurring, travelers or those living abroad should avoid live animal markets and farms where animals are raised, avoid contact with sick or dead animals, avoid eating undercooked or raw animal products (including eggs), and avoid eating foods or drinking beverages that contain animal blood.
CDC website: Flu
The following authors contributed to the previous version of this chapter: Katherine Roguski, Alicia Fry