Volume 12, Number 8—August 2006
Research
Streptococcus suis Sequence Type 7 Outbreak, Sichuan, China
Abstract
An outbreak of Streptococcus suis serotype 2 emerged in the summer of 2005 in Sichuan Province, and sporadic infections occurred in 4 additional provinces of China. In total, 99 S. suis strains were isolated and analyzed in this study: 88 isolates from human patients and 11 from diseased pigs. We defined 98 of 99 isolates as pulse type I by using pulsed-field gel electrophoresis analysis of SmaI-digested chromosomal DNA. Furthermore, multilocus sequence typing classified 97 of 98 members of the pulse type I in the same sequence type (ST), ST-7. Isolates of ST-7 were more toxic to peripheral blood mononuclear cells than ST-1 strains. S. suis ST-7, the causative agent, was a single-locus variant of ST-1 with increased virulence. These findings strongly suggest that ST-7 is an emerging, highly virulent S. suis clone that caused the largest S. suis outbreak ever described.
Streptococcus suis, a swine pathogen, is increasing in clinical importance in countries with intensive swine industries (1–4). Infection in humans is considered an occupational disease that affects persons who work in close contact with pigs or pork byproducts (5). Currently, 35 capsular types or serotypes are officially described; S. suis serotype 2 is considered the most prevalent and virulent in pigs and humans (6). The first human case was recorded in 1968, and only 200 cases have been subsequently reported globally through June 2005 (5). In some Asian countries, S. suis may be the second most common cause of adult streptococcal meningitis (7).
However, a larger outbreak due to S. suis serotype 2 emerged in the summer of 2005 in Sichuan Province, China. In total, 215 cases were reported; 38 deaths occurred in 202 villages in 36 counties. New cases were identified in 4 additional provinces in China after the Sichuan outbreak. A striking feature of this outbreak was the unusually high rate of death and streptococcal toxic shocklike syndrome (STSS) as a clinical manifestation. Indeed, reported symptoms included high fever, malaise, nausea, vomiting, and diarrhea, followed by meningitis, subcutaneous hemorrhage, toxic shock, and coma in severe cases. This increased severity of S. suis infections in humans, such as the shorter incubation time, more rapid disease progression, and higher death rate, underscores the need to better understand the factors associated with S. suis infection.
The aim of this study was to characterize and analyze the causative agent of this unusual outbreak with regard to its virulence and evolution. Using multilocus sequence typing (MLST), we classified the isolated strains into a single sequence type (ST), ST-7 of the ST-1 complex. The ST-1 complex is strongly associated with cases of septicemia and meningitis worldwide (8). S. suis ST-7 expressed the proposed virulence markers muramidase-released protein (MRP), extracellular protein factor (EF), and hemolysin (named suilysin) (9–11) and was markedly more cytototoxic to human peripheral blood mononuclear cells (PBMC) than a representative ST-1 strain.
Bacterial Isolation and Identification
All α-hemolytic streptococcal colonies grown on sheep blood agar from specimens obtained from normally sterile sites on humans or diseased pigs were identified as S. suis by API-20 STREP system (Biomerieux, WeTech, Beijing, People's Republic of China) (Table). The specific serotype of the isolates was characterized as S. suis capsular type 2 by using S. suis antisera specific against individual serotypes (provided by the Statens Serum Institut, Copenhagen, Denmark) and confirmed by the coagglutination test, as previously reported (12). PCR assays were used to detect the genes coding for 16S rRNA of S. suis, for the capsule of S. suis serotype 2 (cps2J), and for MRP, suilysin (sly), and EF, as reported (9,10,13). PCR results were confirmed by sequencing the synthesized fragments. The expression of the virulence markers MRP, suilysin, and EF was confirmed at the protein level by Western blotting bacterial culture supernatants as previously described (11).
Pulsed-field Gel Electrophoresis (PFGE) Analysis
The protocol described by Berthelot-Hérault et al., with modification, was used (14). Cells were restricted with 25 U of SmaI (Promega, Sino-American Biotechnology Co, Beijing, People's Republic of China). DNA fragments were resolved by PFGE with 1% SeaKem Gold agarose gels and the CHEF-DR III system (Bio-Rad, Beijing, People's Republic of China). Salmonella enterica serovar Braenderup H9812 restricted with XbaI was used for molecular weight and size determinations (15). Similarities between restriction endonuclease digestion profiles were analyzed by using BioNumerics software (Applied Maths, Kortrijk, Belgium).
MLST and Phylogenetic Analysis
Seven housekeeping gene loci described by King et al. for MLST analysis of S. suis were used in this study: cpn60, dpr, recA,aroA, thrA, gki, and mutS coding for a 60-kDa chaperonin, a putative peroxide resistance protein, a homologous recombination factor, a 5-enolpyruvylshikimate 3-phosphate synthase, an aspartokinase/homoserine dehydrogenase, a glucose kinase, and a DNA mismatch repair enzyme, respectively (8). PCR products were purified by using QIAquick PCR product purification columns (Qiagen, Gene, Beijing, People's Republic of China) and directly sequenced at both ends with an ABI Prism 3700 DNA analyzer system (Perkin Elmer Applied Biosystems, Wellesley, MA, USA) (8). For each isolate, the alleles at each of the 7 loci defined the allelic profile or ST. MLST information in the S. suis database identified the phylogenetic position of isolates collected in this outbreak investigation (8). eBURST was used to identify clonal complexes within S. suis and to display the overall structure of the population in the MLST database at http://ssuis.mlst.net (16,17). This database contains 92 previously described STs derived from isolates from various sources, including diseased pigs and human patients from different countries. Bayesian maximum likelihood trees were reconstructed with MrBayes version 3.1.1, and maximum likelihood trees were made with the HKY85 model (18).
Cytotoxicity Assays
Bacterial cytotoxicity to PBMCs from healthy donors was determined as previously described (19). PBMCs were seeded at a final concentration of 106 cells/mL in 24-well plates and incubated for 4 h at 37°C in the presence of ≈107 CFU/mL S. suis strain SC84 and SC22 (selected as 2 representative strains from the Sichuan outbreak and isolated from 2 human patients with STSS) or S. suis reference strain 31533 used as a control. Strain 31533, isolated from a pig with meningitis in France, was virulent in pig models of infection and toxic to several types of host cells (19,20). Bacterial cytotoxicity was evaluated by lactate dehydrogenase measurement with Cyto-Tox 96 Cytoxicity Kit (Promega, Madison, WI, USA) according to the manufacturer's instructions. Percentage of cytotoxicity was calculated as (sample optical density [OD]490 – OD0%)/(OD100% – OD0%)×100, where OD0% represents the OD490 of noninfected cells and OD100% represents the OD490 of cells treated with lysis buffer as indicated by the manufacturer. Empty wells with cell culture medium alone served as blanks.
In total, 99 S. suis strains were isolated in this study (Table): 88 from human patients and 11 from diseased pigs. Of the 88 human isolates, 1 each was from Guangdong, Jiangxi, Guizhou, and Guangxi. From the Sichuan outbreak in the summer of 2005, we found 84 human isolates, distributed in 63 administrative villages of 29 counties, and included them in this study. Two strains, 1 from a human patient and 1 from a diseased pig in Jiangsu during the 1998 S. suis outbreak, were also included to determine whether the causative agents for the 2 outbreaks were clonal (21).
Of the 11 pig isolates obtained in 2005, 8 were from Sichuan and 3 were from Jiangxi. One of the 8 isolates from Sichuan, named SC16, originated from 1 diseased pig in a herd. The human patient, who slaughtered and processed the first diseased pig in the same herd, became ill 6 hours later and died 18 hours after the onset of illness. The 3 isolates from Jiangxi were isolated from infected meat samples from a cold storage house. The human patient who processed the meat was infected.
All strains were determined as serotype 2 by using diagnostic antiserum against S. suis and confirmed by the coagglutination method with specific type 2 antiserum (12). The identity of the isolates as S. suis serotype 2 was further confirmed by positive PCR for the genes coding for the 16S rRNA of S. suis and for the capsule of S. suis serotype 2 (cps2J).
PCR showed all isolates to be positive for the virulence genes coding for MRP, suilysin (sly), and EF (9,10). PCR results were confirmed with nucleotide sequencing. Ten selected strains tested positive for suilysin, EF, and MRP by Western blot with specific antibodies, as described previously, which confirmed the expression of these virulence-related genes (11). We saw no indication that S. suis was transmitted between humans, and almost all patients had contact with pigs or infected meat (22).
PFGE effectively detected relationships between genetic background, virulence traits, and epidemiologic implications of many bacterial pathogens. Vela et al. analyzed 302 S. suis clinical isolates of various serotypes from various countries and identified 60 different pulse types, of which 50% corresponded to a single pulse type. One pulse type represented 46.3% of the swine isolates that may be related to a higher pathogenic potential or to a wider environmental distribution (23). In this study, chromosomal DNA digested with SmaI was analyzed in the 101 isolates described in the Table. Results showed 2 pulse types. Pulse type I was found in 100 of the 101 isolates, including those from human patients and diseased pigs from Sichuan and 4 other provinces: Jiangxi, Guangdong, Guangxi, and Jiangsu (Figure 1). This finding indicates that pulse type I was the primary cause of this human outbreak and that the causative agent was clonal. Only 1 isolate, GZ1, isolated in August 2005 and having a slightly different PFGE profile, was from a human patient in Guizhou Province (Figure 1).
Although the PFGE-based network, PulseNet International, is used worldwide for molecular epidemiology, no database is available for S. suis to enable comparison of genetic relatedness among isolates from various origins. MLST defines strains by using sequences at 7 housekeeping loci and has become the method of choice for addressing questions related to epidemiology, population, and evolutionary biology. The MLST network, http://www.mlst.net, allows laboratories to quickly characterize their strains, relate them to those submitted by others, and compare them with the pathogen populations as a whole through the Internet (16,24). The selected 7 housekeeping gene loci from all 101 S. suis isolates were amplified and sequenced. DNA sequences from the 7 loci of all isolates were determined (DQ205243-51), and allelic profiles were assigned and submitted to the MLST database for S. suis (http://ssuis.mlst.net) (8). In our study, 99 of 101 isolates were identified as ST-7, including all 92 isolates from human patients and diseased pigs collected during the Sichuan outbreak in 2005, as well as the 2 isolates from the Jiangsu outbreak in 1998 (1 from a human patient and 1 from a diseased pig). In addition, the isolates from Jiangxi (1 human and 3 pig isolates) and Guangdong (1 human isolate) obtained during summer 2005 were also assigned to ST-7. One human isolate from a patient in Guizhou (strain GZ1) and 1 human isolate from Guangxi (strain GX1) were identified as ST1 (DQ205251). Strain GZ1 showed a PFGE profile that differed slightly from that of the Sichuan isolates (Figure 1, pulse type II). However, strain GX1 had a pulse type I profile similar to that of the Sichuan isolates. These results indicate that MLST is a more powerful discriminatory and epidemiologic tool.
To gain information on the origin and relationships between ST-7 and other STs, we evaluated the population genetic diversity of the entire S. suis MSLT database with eBURST, which divides an MLST dataset of any size into groups of related isolates and clonal complexes, predicts the ancestral genotype for each clonal complex, and computes the bootstrap relatedness value. Using this technique, we showed bacterial clonal diversity in S. suis and provided evidence for the emergence of new clones of particular clinical relevance (17). The population snapshot of all isolates of S. suis generated by eBURST, including our 101 isolates along with all 294 isolates with available MLST data, showed 6 major clusters of related STs and numerous unlinked STs. Of unlinked STs, we noted 6 ST complexes with single-locus variants and some with double-locus variants (8,17). Interspersed among these clonal complexes were minor groups, typically joined doublets and individually unlinked STs that were not single-locus variants of any other STs in the database (Figure 2).
The 6 major clonal complexes with single- and double-locus variants were ST-1, ST-17, ST-27, ST-61, ST-29, and ST-87 (Figure 2). The major clonal complex of S. suis, ST-1, contained isolates from humans and pigs that had invasive disease or were healthy carriers and included, besides serotype 2, isolates of serotypes 1, 1/2, 3, 8, 14, and 1/14. ST-1, original strain of the ST-1 complex (with 100% bootstrap support), includes pig isolates from England (83), Spain (44), the Netherlands (3), France (4), and Hong Kong (5); it is the predominant ST responsible for most swine infections worldwide.
ST-7 may be derived from ST-1, the primary strain of the clonal ST-1 complex, and it may have caused the human infections in Sichuan Province. ST-7 (ST profile 1,1,1,1,1,1,3) (DQ205243-50) and ST-1 (ST profile 1,1,1,1,1,1,1) (DQ205243-51) may have diversified by a stepwise accumulation of point mutations. Furthermore, ST-7 may have only very recently diverged, since it showed a high level of similarity in terms of both allelic profiles and sequences to their nonidentical alleles. Thus, the closely related ST-1 and ST-7 share 6 identical loci and have 1 locus, thyA, differing only at a single nucleotide (Figure 2). Because the housekeeping genes are recognized as the stable core of the bacterial genome, this divergence further shows the emergence of a new virulent ST.
Using eBURST analysis of the MSLT database, we also examined isolates of S. suis serotype 2 from human patients or diseased pigs that have been shown experimentally to be highly virulent in porcine models of infection (8,20,25). Six of the 7 so-called highly virulent strains belonged to ST-1, including 1 strain isolated from a patient with meningitis in England (H11/1) and 1 strain isolated from an abattoir worker with meningitis in Hong Kong (87555). Another highly virulent strain (89-1591) isolated from a pig with septicemia was ST-25, a single-locus variant of the ST-29 complex (26).
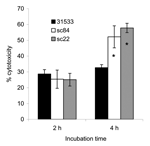
Strains isolated from human patients (including those from the MLST database and this study) can be regrouped in either ST-1, ST-6, ST-7, and ST-84 (ST-1 complex) or ST-25 (ST-29 complex) by MLST. ST-1 includes 10 isolates of S. suis serotype 2 recovered from human infections in France (n = 4), England (n = 1), and Hong Kong. The Hong Kong strains were isolated in 1985 (n = 1), 1995 (n = 3), and 1997 (n = 1). In addition, ST-1 also includes 1 strain of S. suis serotype 14 isolated from a patient in England. ST-25 contains the only 3 serotype 2 strains isolated from human patients in Canada (27,28). ST-6 contains a strain of serotype 14 isolated from a human patient in the Netherlands (29). Before our study, only 1 member of ST-7 had been isolated from the blood of a patient with sepsis in Hong Kong in 1996 (8,30). The ST-1 and ST-29 complexes are highly virulent clones of S. suis populations, and the ST-1 clonal complex is the major complex of S. suis with public health ramifications and is responsible for most human infections. The 3 isolates in ST-25 have none of the recognized virulence genes in S. suis: ef, mrp, and sly. Therefore, we propose that the ST-1 clonal complex is the dominant highly virulent complex and has the potential to cause large human S. suis infection outbreaks. The potential of ST-7 as an emerging virulent clone within the ST-1 complex was further suggested by results obtained by using the cytotoxicity assay with human PBMCs. As shown in Figure 3, live bacterial cell suspensions prepared from S. suis strains SC84 and SC22 (ST-7), isolated from human patients with STSS in Sichuan, were significantly more toxic to PBMCs than the S. suis reference strain 31533 (ST-1) when cells were incubated >2 hours under similar conditions of bacterial growth. Strain 31533 was virulent in mouse and pig models of infection and was toxic to several types of host cells, as demonstrated by in vitro assays (19). Thus, this difference in cytotoxicity is not only significant but also relevant to the fulminant characteristics of the Sichuan outbreak.
This hypothesis is further supported by phylogenetic analysis of the sequences from 92 STs of the 7 S. suis housekeeping genes (Figure 4). To show the phylogenetic relationships between the ST-1 complex and genetic populations of S. suis, we examined the possibility that phylogenetic data were sufficient to construct a relatedness tree among members of STs and, in particular, for the virulent clonal ST-1 complex. Phylogenetic analysis showed 6 lineages within the S. suis population. The previously identified complexes ST-61, ST-27, and ST-87 were placed in lineages 2, 5, and 6, respectively; most of the STs have only a single isolate. The ST-29 complex that contains human isolates of ST-25 is in lineage 5. The clonal ST-1 complex forms a single lineage and appears to be dominant in various locations (8). It has 4 STs for human isolates, ST-1, ST-6, ST-7, and ST-84, which may have evolved from a common ancestor (Figure 4). In this complex, S. suis ST-7 emerged first in Hong Kong in 1996, caused 28 cases in Jiangsu Province in 1998, and was responsible for the largest outbreak of human S. suis infection in history that occurred in Sichuan Province, China, in 2005 and resulted in 215 cases with 38 deaths (8,21,30).
The high death rate in the Sichuan outbreak is of great concern since few human S. suis infections are fatal. This increased virulence may be related to a horizontal gene transfer of a possible new toxin or superantigen consistent with the most relevant clinical manifestation of this outbreak, i.e., STSS. Recently, resistance gene exchanges were reported between porcine S. suis and various human streptococcal species, including S. pyogenes. Thus, gene transfer from porcine to human streptococci and vice versa is possible, albeit at a low frequency (31). Therefore, S. suis ST-7 might be an emerging virulent infectious disease. This unusual S. suis clone may spread further across China and to the rest of the world; spread may become more pronounced with the rapid development of the export business.
Dr Ye is a microbiologist. Her main research interest is in emerging infectious diseases. She is a group leader for the study of S. suis infection in China.
Acknowledgments
We thank A.M. Whatmore and S.J. King for assistance with MLST analysis.
This work was supported by grants (2005CB522904 and 2003BA712A02 to J.G.X.) from Ministry of Science and Technology, People's Republic of China.
References
- Halaby T, Hoitsma E, Hupperts R, Spanjaard L, Luirink M, Jacobs J. Streptococcus suis meningitis, a poacher's risk. Eur J Clin Microbiol Infect Dis. 2000;19:943–5. DOIPubMedGoogle Scholar
- Juncal AR, Pardo F, Rodriguez I, Perez del Molino ML. Meningitis by Streptococcus suis [article in Spanish]. Enferm Infecc Microbiol Clin. 1997;15:120–1.PubMedGoogle Scholar
- Staats JJ, Feder I, Okwumabua O, Chengappa MM. Streptococcus suis: past and present. Vet Res Commun. 1997;21:381–407. DOIPubMedGoogle Scholar
- Dupas D, Vignon M, Geraut C. Streptococcus suis meningitis. A severe noncompensated occupational disease. J Occup Med. 1992;34:1102–5. DOIPubMedGoogle Scholar
- Gottschalk M, Segura M. The pathogenesis of the meningitis caused by Streptococcus suis: the unresolved questions. Vet Microbiol. 2000;76:259–72. DOIPubMedGoogle Scholar
- Suankratay C, Intalapaporn P, Nunthapisud P, Arunyingmongkol K, Wilde H. Streptococcus suis meningitis in Thailand. Southeast Asian J Trop Med Public Health. 2004;35:868–76.PubMedGoogle Scholar
- King SJ, Leigh JA, Heath PJ, Luque I, Tarradas C, Dowson CG, Development of a multilocus sequence typing scheme for the pig pathogen Streptococcus suis: identification of virulent clones and potential capsular serotype exchange. J Clin Microbiol. 2002;40:3671–80. DOIPubMedGoogle Scholar
- Wisselink HJ, Reek FH, Vecht U, Stockhofe-Zurwieden N, Smits MA, Smith HE. Detection of virulent strains of Streptococcus suis type 2 and highly virulent strains of Streptococcus suis type 1 in tonsillar specimens of pigs by PCR. Vet Microbiol. 1999;67:143–57. DOIPubMedGoogle Scholar
- Staats JJ, Plattner BL, Stewart GC, Changappa MM. Presence of the Streptococcus suis suilysin gene and expression of MRP and EF correlates with high virulence in Streptococcus suis type 2 isolates. Vet Microbiol. 1999;70:201–11. DOIPubMedGoogle Scholar
- Berthelot-Herault F, Morvan H, Keribin AM, Gottschalk M, Kobisch M. Production of muraminidase-released protein (MRP), extracellular factor (EF) and suilysin by field isolates of Streptococcus suis capsular types 2, 1/2, 9, 7 and 3 isolated from swine in France. Vet Res. 2000;31:473–9. DOIPubMedGoogle Scholar
- Higgins R, Gottschalk M. An update on Streptococcus suis identification. J Vet Diagn Invest. 1990;2:249–52. DOIPubMedGoogle Scholar
- Marois C, Bougeard S, Gottschalk M, Kobisch M. Multiplex PCR assay for detection of Streptococcus suis species and serotypes 2 and 1/2 in tonsils of live and dead pigs. J Clin Microbiol. 2004;42:3169–75. DOIPubMedGoogle Scholar
- Berthelot-Herault F, Marois C, Gottschalk M, Kobisch M. Genetic diversity of Streptococcus suis strains isolated from pigs and humans as revealed by pulsed-field gel electrophoresis. J Clin Microbiol. 2002;40:615–9. DOIPubMedGoogle Scholar
- Hunter SB, Vauterin P, Lambert-Fair MA, van Duyne MS, Kubota K, Graves L, Establishment of a universal size standard strain for use with the PulseNet standardized pulsed-field gel electrophoresis protocols: converting the national databases to the new size standard. J Clin Microbiol. 2005;43:1045–50. DOIPubMedGoogle Scholar
- Aanensen DM, Spratt BG. The multilocus sequence typing network: mlst.net. Nucleic Acids Res. 2005;33:W728–33. DOIPubMedGoogle Scholar
- Feil EJ, Li BC, Aanensen DM, Hanage WP, Spratt BG. eBURST: inferring patterns of evolutionary descent among clusters of related bacterial genotypes from multilocus sequence typing data. J Bacteriol. 2004;186:1518–30. DOIPubMedGoogle Scholar
- Ronquist F, Huelsenbeck JP. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics. 2003;19:1572–4. DOIPubMedGoogle Scholar
- Vanier G, Segura M, Friedl P, Lacouture S, Gottschalk M. Invasion of porcine brain microvascular endothelial cells by Streptococcus suis serotype 2. Infect Immun. 2004;72:1441–9. DOIPubMedGoogle Scholar
- Vecht U, Wisselink HJ, Stockhofe-Zurwieden N, Smith HE. Characterization of virulence of the Streptococcus suis serotype 2 reference strain Henrichsen S 735 in newborn gnotobiotic pigs. Vet Microbiol. 1996;51:125–36. DOIPubMedGoogle Scholar
- Zhu F, Yang H, Hu X, Wang H, Wang G, Song Y, Homogeneity study on the Streptococcus suis isolated from human and swine [article in Chinese]. Zhonghua Liu Xing Bing Xue Za Zhi. 2000;21:427–9.PubMedGoogle Scholar
- Yu H, Jing H, Chen Z, Zheng H, Zhu X, Wang H, Human Streptococcus suis outbreak, Sichuan, China. Emerg Infect Dis. 2006;12:914–20.PubMedGoogle Scholar
- Vela AI, Goyache J, Tarradas C, Luque I, Mateos A, Moreno MA, Analysis of genetic diversity of Streptococcus suis clinical isolates from pigs in Spain by pulsed-field gel electrophoresis. J Clin Microbiol. 2003;41:2498–502. DOIPubMedGoogle Scholar
- Cooper JE, Feil EJ. Multilocus sequence typing–what is resolved? Trends Microbiol. 2004;12:373–7. DOIPubMedGoogle Scholar
- Galina L, Pijoan C, Sitjar M, Christianson WT, Rossow K, Collins JE. Interaction between Streptococcus suis serotype 2 and porcine reproductive and respiratory syndrome virus in specific pathogen-free piglets. Vet Rec. 1994;134:60–4. DOIPubMedGoogle Scholar
- Quessy S, Dubreuil JD, Caya M, Higgins R. Discrimination of virulent and avirulent Streptococcus suis capsular type 2 isolates from different geographical origins. Infect Immun. 1995;63:1975–9.PubMedGoogle Scholar
- Michaud S. Streptococcus suis meningitis: first case reported in Quebec. Can J Infect Dis Med Microbiol. 1996;7:329–31.
- Trottier S, Higgins R, Brochu G, Gottschalk M. A case of human endocarditis due to Streptococcus suis in North America. Rev Infect Dis. 1991;13:1251–2. DOIPubMedGoogle Scholar
- Gottschalk M, Higgins R, Jacques M, Mittal KR, Henrichsen J. Description of 14 new capsular types of Streptococcus suis. J Clin Microbiol. 1989;27:2633–6.PubMedGoogle Scholar
- Kay R, Cheng AF, Tse CY. Streptococcus suis infection in Hong Kong. QJM. 1995;88:39–47.PubMedGoogle Scholar
- Martel A, Decostere A, Leener ED, Marien M, Graef ED, Heyndrickx M, Comparison and transferability of the erm (B) genes between human and farm animal streptococci 6. Microb Drug Resist. 2005;11:295–302. DOIPubMedGoogle Scholar
Figures
Table
Cite This Article1These authors contributed equally to this study.
Table of Contents – Volume 12, Number 8—August 2006
| EID Search Options |
|---|
|
|
|
|
|
|



Please use the form below to submit correspondence to the authors or contact them at the following address:
Jianguo Xu, State Key Laboratory for Infectious Disease Prevention and Control, National Institute for Communicable Disease Control and Prevention, Chinese Center for Disease Control and Prevention, PO Box 5, Changping, Beijing 102206, People’s Republic of China
Top