Volume 13, Number 4—April 2007
Dispatch
16S rRNA Methylase–producing, Gram-Negative Pathogens, Japan
Cite This Article
Citation for Media
Abstract
To investigate the exact isolation frequency of 16S rRNA methylase–producing, gram-negative pathogenic bacteria, we tested 87,626 clinical isolates from 169 hospitals. Twenty-six strains from 16 hospitals harbored 16S rRNA methylase genes, which suggests sparse but diffuse spread of pan-aminoglycoside–resistant microbes in Japan.
Broad-spectrum β-lactams and fluoroquinolones have been widely prescribed in the treatment of gram-negative bacterial infections; as a result, resistance to these antimicrobial agents has developed in some species. Although these agents are not immune to an increasing number of resistance mechanisms, they remain relatively potent and continue to be essential antimicrobial drugs for treating life-threatening bacterial infections.
Although the production of aminoglycoside-modifying enzymes is the most common mechanism of resistance in aminoglycosides, the emergence of pan-aminoglycoside–resistant, 16S rRNA methylase–producing, gram-negative bacteria has been increasingly reported in recent years. Five types of plasmid-mediated 16S rRNA methylases (ArmA, RmtA, RmtB, RmtC, and RmtD) have so far been identified in east Asia, Europe, and South America (1–7). RmtA was first identified in 2001 in Japan (3) and has so far been identified exclusively in Pseudomonas aeruginosa (8). RmtC was subsequently identified only in Proteus mirabilis (4). RmtB has been found among various gram-negative bacterial species, including Serratia marcescens, Escherichia coli, Citrobacter freundii, Klebsiella pneumoniae, and K. oxytoca, isolated in Japan, South Korea, and Taiwan (2,5,6,9). Another new 16S rRNA methylase was initially identified in C. freundii in Poland, submitted to European Molecular Biology Laboratory (EMBL)/GenBank in 2002 (accession no. AF550415), and later characterized and assigned as ArmA in K. pneumoniae, E. coli, Enterobacter cloacae, Salmonella enterica, and Shigella flexneri in France, Bulgaria, and Spain (10,11). Moreover, ArmA was also identified in E. coli, K. pneumoniae, E. cloacae, C. freundii and S. marcescens in South Korea, Japan, and Taiwan (5,8,9). This enzyme has also been identified in a glucose nonfermentative Acinetobacter sp. in South Korea and Japan (6,8). Quite recently, RmtD was newly identified in the SMP-1–producing P. aeruginosa strain PA0905, which was isolated in Brazil (7). In Japan, arbekacin, a semisynthetic aminoglycoside, has been approved for treatment of methicillin-resistant Staphylococcus aureus infections, and this agent is also very efficacious for gram-negative bacteria. However, 16S rRNA methylase–producing microbes can adapt to this agent, and its prescription may well be a selective pressure on the kind of microbes in the clinical environment. Thus, this investigation was conducted to determine the exact isolation frequency of 16S rRNA methylase–producing, gram-negative pathogenic bacteria in Japanese medical facilities and assess the possibility of the future prevalence of these hazardous microbes.
From September 1 to October 31, 2004, 169 medical facilities with in-house microbiology laboratories participated in this investigation. Clinical specimens were collected from inpatients and outpatients with suspected infections. Bacterial isolates that belonged to the family Enterobacteriaceae or were nonfermentors of glucose, for example, P. aeruginosa and Acinetobacter spp., were included in this study. A total of 87,626 clinical isolates were collected and analyzed. The results are shown in Table 1.
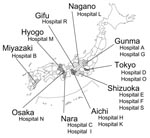
Twenty-nine strains (17 P. aeruginosa, 4 A. baumannii, 3 E. coli, 2 P. mirabilis, 1 E. cloacae, 1 K. pneumoniae, and 1 Enterobacter aerogenes) that grew on LB agar plates supplemented with 500 mg of arbekacin per liter were subjected to the typing of 16S rRNA methylase genes by a multiplex PCR. Primers used for the PCR amplification of bacterial 16S rRNA methylase genes were the following: RMTA-F 5′-CTA GCG TCC ATC CTT TCC TC-3′ and RMTA-R 5′-TTT GCT TCC ATG CCC TTG CC-3′, which amplify a 635-bp DNA fragment within rmtA gene (3); RMTB-F 5′-GCT TTC TGC GGG CGA TGT AA-3′ and RMTB-R 5′-ATG CAA TGC CGC GCT CGT AT-3′, which amplify a 173-bp DNA fragment within rmtB (2); RMTC-F 5′-CGA AGA AGT AAC AGC CAA AG-3′ and RMTC-R 5′-ATC CCA ACA TCT CTC CCA CT-3′, which amplify a 711-bp DNA fragment within rmtC (4); and ARMA-F 5′-ATT CTG CCT ATC CTA ATT GG-3′ and ARMA-R 5′-ACC TAT ACT TTA TCG TCG TC-3′, which amplify a 315-bp DNA fragment within armA (accession nos. AY220558 and AB117519). PCR results and clinical data from these 29 strains are summarized in the Table 2. Genes for 16S rRNA methylases were absent in 3 arbekacin high-level-resistant strains of P. aeruginosa by PCR analyses that used 4 sets of 16S rRNA methylase-specific primers. In these strains, simultaneous production of multiple aminoglycoside-modifying enzymes was suggested as reported previously (12). Twenty-six strains harboring any of the four 16S rRNA methylase genes were identified in 16 hospitals, with no apparent geographic convergence in the locations of the hospitals (Figure 1). In hospital L, 3 different bacterial species (E. coli, E. aerogenes, and K. pneumoniae) harbored the armA gene, which suggests probable conjugal transfer of armA-carrying plasmids among different bacterial species.
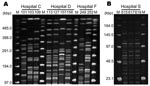
Pulsed-field gel electrophoresis (PFGE) was performed on 9 strains of P. aeruginosa and 3 strains of A. baumannii isolated from 4 separate hospitals where 16S rRNA methylase genes were isolated (Figure 2). Genomic DNA preparations from P. aeruginosa and A. baumannii were digested with SpeI and SmaI, respectively. Clonality was inferred based on the criteria of Tenover et al. (13) Two of 3 rmtA-positive P. aeruginosa strains isolated in hospital C were estimated to be the same clone. Among 4 rmtA-positive P. aeruginosa isolates recovered in hospital D, 2 different clonal lineages were observed. This finding suggests possible conjugal transfers of rmtA-carrying plasmids among genetically different strains of P. aeruginosa. Three armA gene–harboring A. baumannii identified in hospital S were obviously the same clone. These findings imply probable nosocomial transmission of 16S rRNA methylase gene–harboring strains in hospitals C, D, and S, as well as frequent conjugal transfers of plasmids carrying 16S rRNA methylase genes among gram-negative pathogenic bacterial species.
MIC determinations were performed according to the guideline of the CLSI (formerly National Committee on Clinical Laboratory Standards). All 16S rRNA methylase-positive strains were highly resistant (MICs >1,024 mg/L) of all 4,6-disubstituted deoxystreptamine group aminoglycosides (Table 3). In contrast, resistance to streptomycin and neomycin varied. Three16S rRNA methylase gene-negative P. aeruginosa strains were also highly resistant to arbekacin, but the MICs of some of the 4,6-disubstituted deoxystreptamine group aminoglycosides were relatively lower (256–512 mg/L) for these strains than those for 16S rRNA methylase gene–positive strains (>1,024 mg/L). Strains harboring 16S rRNA methylase genes tended to show resistance to oxyimino-cephalosporins such as cefotaxime and ceftazidime as well, but were susceptible to imipenem. As reported for the armA- or rmtB-bearing strains, the presence of β-lactamase genes was suggested in cefotaxime-resistant strains, and indeed the blaCTX-M-14 gene was detected in several rmtB-positive strains tested in our study (data not shown). Some of these strains also demonstrated resistance to fluoroquinolones (Table 3).
The overall isolation frequency of 16S rRNA methylase-gene-positive gram-negative bacilli was very low (0.03%) in Japanese medical facilities in 2004, with the highest rates seen in P. aeruginosa and Acinetobacter spp. at 0.08% and 0.12%, respectively. Twenty-six bacterial isolates carrying 1 of the four 16S rRNA methylase genes were recovered from 16 (9.5%) of 169 hospitals that participated in this nationwide investigation. Of the 169 hospitals, 162 hospitals had >200 beds, accounting for 5.9% of all Japanese hospitals of similar scale. This implies that 16S rRNA methylase–producing strains might have been present in >250 Japanese hospitals during the investigation period, which in turn suggests sparse but diffuse spread of 16S rRNA methylase producers in Japan. Since several armA- or rmtB-positive strains have also been isolated in European and Asian countries, and given the potential for further dissemination, nationwide identification and ongoing surveillance of these isolates should be considered by all countries.
According to PFGE typing, nosocomial transmission of 16S rRNA methylase–producing P. aeruginosa and A. baumannii was suspected in 3 hospitals (hospitals C, D, and S). The banding patterns of rmtA-harboring P. aeruginosa isolated in hospitals C, D, and F were diverse, which excluded the possibility of an epidemic P. aeruginosa strain harboring the rmtA gene. Despite the observation of 2 different PFGE profiles among the 4 P. aeruginosa strains isolated in hospital D, they might share the same plasmids carrying the rmtA gene. For further characterization of genetic relations among rmtA-harboring P. aeruginosa strains, comparative analyses of plasmids and mobile elements that carry the rmtA gene (14) should also be pursued.
Nosocomial infections caused by multidrug-resistant, gram-negative bacteria have become a serious problem in clinical facilities. P. aeruginosa and Acinetobacter spp. have been especially efficient at developing multidrug resistance against broad-spectrum β-lactams, fluoroquinolones, and aminoglycosides (3,6,7,9). The identification of armA and rmtB genes in Europe and East Asia in both human (1–11) and livestock (15; EMBL/GenBank accession no. DQ345788) populations suggests that we must pay consistent attention to prevent further global proliferation. If 16S rRNA methylase–positive bacterial isolates disseminate widely and extensively, the high level of pan-aminoglycoside resistance will undoubtedly have an impact on illness, deaths, and costs of care in both clinical and livestock-breeding environments.
Dr Yamane is a research scientist at the National Institute of Infectious Diseases, Japan. His research interests include infection control and the molecular mechanisms of antimicrobial resistance in nosocomial bacteria.
Acknowledgments
We thank all of the cooperating medical institutions for submitting the various bacterial strains to the National Reference Laboratory to allow this study.
Bacterial strains were collected and characterized by a grant (H16-Tokubetsu-027) from the Ministry of Health, Labor and Welfare, Japan. Precise molecular typing was supported in part by a Grant-in-Aid (no. 16790318) for Young Scientists (B) from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
References
- Galimand M, Courvalin P, Lambert T. Plasmid-mediated high-level resistance to aminoglycosides in Enterobacteriaceae due to 16S rRNA methylation. Antimicrob Agents Chemother. 2003;47:2565–71. DOIPubMedGoogle Scholar
- Doi Y, Yokoyama K, Yamane K, Wachino J, Shibata N, Yagi T, Plasmid-mediated 16S rRNA methylase in Serratia marcescens conferring high-level resistance to aminoglycosides. Antimicrob Agents Chemother. 2004;48:491–6. DOIPubMedGoogle Scholar
- Yokoyama K, Doi Y, Yamane K, Kurokawa H, Shibata N, Shibayama K, Acquisition of 16S rRNA methylase gene in Pseudomonas aeruginosa. Lancet. 2003;362:1888–93. DOIPubMedGoogle Scholar
- Wachino J, Yamane K, Shibayama K, Kurokawa H, Shibata N, Suzuki S, Novel plasmid-mediated 16S rRNA methylase, RmtC, found in a Proteus mirabilis isolate demonstrating extraordinary high-level resistance against various aminoglycosides. Antimicrob Agents Chemother. 2006;50:178–84. DOIPubMedGoogle Scholar
- Park YJ, Lee S, Yu JK, Woo GJ, Lee K, Arakawa Y. Co-production of 16S rRNA methylases and extended-spectrum β-lactamases in AmpC-producing Enterobacter cloacae, Citrobacter freundii and Serratia marcescens in Korea. J Antimicrob Chemother. 2006;58:907–8. DOIPubMedGoogle Scholar
- Lee H, Yong D, Yum JH, Roh KH, Lee K, Yamane K, Dissemination of 16S rRNA methylase-mediated highly amikacin-resistant isolates of Klebsiella pneumoniae and Acinetobacter baumannii in Korea. Diagn Microbiol Infect Dis. 2006;56:305–12. DOIPubMedGoogle Scholar
- Doi Y, de Oliveira Garcia D, Adams J, Paterson DL. Co-production of novel 16S rRNA methylase RmtD and metallo-β-lactamase SPM-1 in a panresistant Pseudomonas aeruginosa isolate from Brazil. Antimicrob Agents Chemother. 2007;51:8552–6.
- Yamane K, Wachino J, Doi Y, Kurokawa H, Arakawa Y. Global spread of multiple-aminoglycoside-resistance genes. Emerg Infect Dis. 2005;11:951–3.PubMedGoogle Scholar
- Yan JJ, Wu JJ, Ko WC, Tsai SH, Chuang CL, Wu HM, Plasmid-mediated 16S rRNA methylases conferring high-level aminoglycoside resistance in Escherichia coli and Klebsiella pneumoniae isolates from two Taiwanese hospitals. J Antimicrob Chemother. 2004;54:1007–12. DOIPubMedGoogle Scholar
- Galimand M, Sabtcheva S, Courvalin P, Lambert T. Worldwide disseminated armA aminoglycoside resistance methylase gene is borne by composite transposon Tn1548. Antimicrob Agents Chemother. 2005;49:2949–53. DOIPubMedGoogle Scholar
- Gonzalez-Zorn B, Catalan A, Escudero JA, Dominguez L, Teshager T, Porrero C, Genetic basis for dissemination of armA. J Antimicrob Chemother. 2005;56:583–5. DOIPubMedGoogle Scholar
- Doi Y, Wachino J, Yamane K, Shibata N, Yagi T, Shibayama K, Spread of novel aminoglycoside resistance gene aac(6')-Iad among Acinetobacter clinical isolates in Japan. Antimicrob Agents Chemother. 2004;48:2075–80. DOIPubMedGoogle Scholar
- Tenover FC, Arbeit RD, Goering RV, Mickelsen PA, Murray BE, Persing DH, Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J Clin Microbiol. 1995;33:2233–9.PubMedGoogle Scholar
- Yamane K, Doi Y, Yokoyama K, Yagi T, Kurokawa H, Shibata N, Genetic environments of the rmtA gene in Pseudomonas aeruginosa clinical isolates. Antimicrob Agents Chemother. 2004;48:2069–74. DOIPubMedGoogle Scholar
- Gonzalez-Zorn B, Teshager T, Casas M, Porrero MC, Moreno MA, Courvalin P, armA and aminoglycoside resistance in Escherichia coli. Emerg Infect Dis. 2005;11:954–6.PubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 13, Number 4—April 2007
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Yoshichika Arakawa, Department of Bacterial Pathogenesis and Infection Control, National Institute of Infectious Diseases, 4-7-1 Gakuen, Musashi-Murayama, Tokyo 208-0011, Japan;
Top