Volume 14, Number 12—December 2008
Research
Influenza Infection in Wild Raccoons
Abstract
Raccoons (Procyon lotor) are common, widely distributed animals that frequently come into contact with wild waterfowl, agricultural operations, and humans. Serosurveys showed that raccoons are exposed to avian influenza virus. We found antibodies to a variety of influenza virus subtypes (H10N7, H4N6, H4N2, H3, and H1) with wide geographic variation in seroprevalence. Experimental infection studies showed that raccoons become infected with avian and human influenza A viruses, shed and transmit virus to virus-free animals, and seroconvert. Analyses of cellular receptors showed that raccoons have avian and human type receptors with a similar distribution as found in human respiratory tracts. The potential exists for co-infection of multiple subtypes of influenza virus with genetic reassortment and creation of novel strains of influenza virus. Experimental and field data indicate that raccoons may play an important role in influenza disease ecology and pose risks to agriculture and human health.
The primary reservoirs of avian influenza (AI) are wild birds in the orders Anseriformes (ducks, geese, and swans) and Charadriiformes (gulls, terns, and shorebirds). In these hosts, low-pathogenic forms of the virus typically cause little or no apparent disease, however, large quantities of virus are shed in fecal matter. AI virus is relatively stable in water and can remain viable for up to 200 days, depending on temperature and other environmental factors (1). Thus, bodies of water and adjacent shorelines that wild birds use can become potentially contaminated, increasing the likelihood of subsequent exposure of avian and non-avian species to AI virus.
The preference of influenza viruses for different cellular receptors and the presence and distribution of those receptors in the host are important factors involved in determining host range and tissue tropism (2). Humans are not typically infected by AI virus because receptors for this virus are distributed in tissues that are located predominantly in the lower respiratory tract. As such, these receptors are not as accessible as human type receptors found in the upper respiratory tissues and require more intimate contact for transmission. Swine are considered important intermediate hosts between birds and humans because they are frequently infected by avian and human influenza viruses (3). This finding underscores the potential for genetic reassortment that can create new, possibly more virulent subtypes.
Other non-avian hosts of AI virus include mink, harbor seals, pilot whales, dogs, cats, and horses (4). These species were found to be competent hosts only after attracting attention because of severe death or illness (4). Wild mammals often reside in the same habitats as waterfowl, feed in the same agricultural areas, wallow and swim in the same bodies of water, and prey on and scavenge dead birds for food. Therefore, ample opportunities exist for free-ranging wild mammals to be exposed to AI by contact with waterfowl and their environment. Many of these species are highly mobile and have large home ranges that can include agricultural operations, wetlands, and human residences. Humans are frequently unaware of their presence, and wild mammals have the potential to contract AI from waterfowl or their environment and to then transmit AI to domestic animals or humans. To date, no studies have systematically examined wild mammalian species, particularly peridomestic mammals, for exposure to AI, their ability to become infected, and their reassortment potential. This knowledge is critical for accurate risk assessments of low pathogenic and highly pathogenic AI to agriculture and human health.
Raccoons (Procyon lotor) are widespread and common in riparian, wooded, and suburban settings over much of North America (5). Previously, antibodies against AI virus (H4N6) were found in 1 raccoon in Pennsylvania (J. Hall, unpub. data). This finding led us to conduct the present study in which we examined wild populations of raccoons from various regions of the United States for antibodies to influenza virus. Experimental infections of raccoons with avian and human influenza viruses were performed to determine viral shedding, transmission, and immune response. The abundance and distribution of avian or human influenza virus cellular receptors in respiratory tissues was analyzed to determine potential for co-infection and possible reassortment of influenza virus strains in this host. These data provided insight into the complexity of influenza disease ecology and the overlooked, potentially important roles of peridomestic wildlife in transmission cycles.
Field Sample Collection
Blood was opportunistically collected from wild raccoons taken during population control operations in various counties/parishes in Texas, Wyoming, Louisiana, California, and Maryland. Raccoons from northwestern Georgia were sampled as part of the US Department of Agriculture Cooperative National Oral Rabies Vaccination program. Raccoons in Colorado were captured for this, and other studies, in and around Fort Collins, Colorado. Blood samples were obtained by cardiac or jugular puncture, allowed to clot, and centrifuged to separate serum from cellular blood components. Serum was transferred to fresh cryovials and stored frozen (–20°C) until transport to the National Wildlife Research Center in Fort Collins, where they were stored at –80°C until analysis.
Screening for Antibodies to Influenza Virus
Agar gel immunodiffusion is a serologic assay used to detect antibodies to influenza viruses. The antigen used in the assay was derived from the matrix and nucleoproteins of AI and is used to detect antibodies to all subtypes of AI. The procedure has been described by Beard (6) and was performed by using reagents and the protocol provided by the Center for Veterinary Biologics and National Veterinary Services Laboratories (Ames, IA, USA).
Determination of Antibody Subtypes for Influenza Virus
Hemagglutination inhibition and neuraminidase inhibition are used to determine subtype identity of influenza antibodies in sera. These procedures are described by Beard (6) and were performed at the National Veterinary Services Laboratories.
Experimental Infection of Raccoons
Ten wild raccoons were live-trapped in and around Fort Collins, Colorado. These animals were transported to the National Wildlife Research Center and held for a 2-week quarantine period, where they were observed daily and judged to be in good overall health on the basis of food intake, behavior, and absence of clinical signs of disease. After 2 weeks, the animals were anesthetized and moved into a biocontainment level 2 facility. Blood samples and nasal and rectal swabs were collected, and animals were placed in individual cages (5 per room). Four animals in each room were inoculated intranasally with 105 50% egg infectious dose (EID50) of AI virus A/CK/AL/75 (H4N8) diluted in 100 μL of viral transport medium. The fifth animal in each room was uninoculated and monitored to determine transmission between animals. To prevent potential fomite transmission, these controls were always handled and sampled first when technicians entered the rooms, and all food and water bowls were cleaned and sanitized in hypochlorite solution each day. Each animal was provided food and water ad libitum and observed daily for illness, behavior, and general welfare. To avoid excessive handling and anesthesia, serum samples, nasal and rectal swabs, and rectal temperatures were collected from each group of 5 raccoons on alternating days for 14 days. Blood samples were obtained by jugular puncture. Nasal and rectal swabs were collected by using dacron-tipped applicators placed into viral transport media after swabbing. All samples were stored at –80°C until analyzed.
Subsequently, a second cohort of 6 raccoons was captured in Fort Collins, quarantined, and placed into biocontainment. Four raccoons were intranasally inoculated with 105 EID50 of human influenza virus (A/Aichi/2/68 [H3N2]) and were sampled and monitored as described above. Two uninoculated raccoons were housed in cages adjacent to inoculated animals to assess transmission. All animal handling, trapping, and experimental infections were performed following Institutional Animal Care and Use Committee and institutional biosafety protocols, guidelines, and approval.
Reverse Transcription–PCR
Viral RNA was extracted from nasal swabs by using the QIAamp Viral RNA Mini Kit (QIAGEN, Valencia, CA, USA), following manufacturer’s instructions. Viral RNA from rectal swabs was extracted by using the same procedure with addition of half of an Inhibitex tablet (Stool Extraction Kit; QIAGEN) to remove PCR inhibitors. Real-time reverse transcription–PCR (RT-PCR) was performed following the procedure of Spackman et al. (7). Samples were compared with standard curves generated from known concentrations of AI, extracted, and amplified by using the same procedures. Results are expressed as EID50 equivalents. These procedures were used to analyze environmental samples (feces and water) as part of the national surveillance for highly pathogenic AI (8).
Detection of Influenza Virus Cellular Receptors in Raccoon Respiratory Tissues
Airway tissue sections were collected from 5 adult raccoons (humanely killed in a different study) from 7 standardized locations (nasal mucosa; larynx; upper, middle, and lower trachea; bronchus; lung), fixed in formalin, and embedded in paraffin. Airway tissues were cut into 5 μm–thick sections, mounted on 3-aminopropyltrethoxy-silane–coated slides, deparaffinized in xylene, and rehydrated in alcohol. For detection of sialic acids (SAs), sections were stained with SAα2,3Gal- and SAα2,6Gal-specific lectins. Briefly, sections were incubated overnight with 250 μL of Western blocking solution (Roche Biochemicals, Indianapolis, IN, USA), washed 3× in Tris-buffered saline, pH 7.6, and incubated with 250 μL of fluorescein isothiocyanate–labeled Sambucus nigra lectin (Vector Laboratories, Burlingame, CA, USA) and biotinylated Maackia amurensis lectin (Vector Laboratories) overnight at 4°C. After 3 washes in Tris-buffered saline, sections were incubated with Alexa Fluor 594–conjugated streptavidin (Molecular Probes, Inc., Eugene, OR, USA) for 2 hours at room temperature. The sections were washed, counterstained with 4′,6-diamino-2-phenylindole dihydrochloride (Molecular Probes), washed again, and mounted on cover glass. Sections were examined with a fluorescence microscope (Carl Zeiss, Inc., Oberkochen, Germany). Because previous research has demonstrated that equine tracheal epithelial cells predominantly express SAα2,3Gal residues and pig tracheal cells express SAα2,3Gal and SAα2,6Gal residues (2), sections of equine and porcine trachea were included as positive controls for each staining procedure.
Serologic Survey of Wild Raccoons for Exposure to AI Virus
We screened 730 wild raccoons from California, Texas, Louisiana, Maryland, Wyoming, and Colorado. Of these, 17 (2.4%) had antibodies to AI virus. Table 1 summarizes the raccoon serosurvey and subtyping results from these states. Four (2.4%) of 168 Maryland raccoons in 2004 had antibodies to AI virus with 3 hemagglutinin subtypes represented. Two of these raccoons had antibodies to 2 subtypes, which indicated multiple exposures to AI virus. Colorado and Wyoming also had seropositive raccoons with prevalences of 12.8% and 25%, respectively. Multiple subtypes were present in both populations, and multiple exposures in individual raccoons were observed. However, none of the raccoons from Georgia, Texas, or California showed serologic evidence of exposure to AI virus. These results indicated that wild raccoons are exposed to a variety of AI virus subtypes and seroconvert on the basis of these exposures.
Experimental Infection of Raccoons with AI Virus
To determine whether raccoons are competent hosts for AI virus infection and are capable of shedding and transmitting virus, raccoons were infected with a specific subtype of AI virus (H4N8) and monitored for symptoms of infection and disease. Two of 10 wild-caught raccoons had antibodies to AI virus (Table 1). These animals were included in the infection study because the AI virus inoculum used was a different subtype, but with potential cross-neutralization as a caveat.
Eight raccoons were inoculated intranasally with 105.0 EID50 of AI virus (H4N8) and monitored for 14 days postinoculation (dpi). Four (50%) of these animals became infected, as shown by nasal shedding of viral RNA detected by RT-PCR. Two of these animals (256 and 275) shed detectable amounts of virus at only 1 time point (1 dpi). Another raccoon (259) shed virus at least up to 6 dpi, and the other infected raccoon (263) shed for the entire 14 days of the study (Table 2). RT-PCR analyses of rectal swabs showed no detectable viral RNA shed by digestive tracts of infected raccoons (data not shown), which is consistent with influenza being primarily a respiratory disease in mammals (2).
One of the 2 uninoculated raccoons housed in cages adjacent (within ≈0.5 m) to inoculated raccoons developed nasal shedding of virus. Every precaution was taken to prevent inadvertent transmission by handling; thus, this animal (262) probably contracted the virus by aerosol from >1 of its infected cohorts. This result indicated that raccoons are capable of transmitting influenza virus from one to another. Given the small amounts of AI virus shed by these raccoons and the timing of infection of this animal, we cannot rule out possible aerosolization of inoculum by adjacent raccoons and transmission by that route.
Three of the 5 raccoons that shed virus developed antibodies to the AI virus (H4N8) isolate, including raccoon 262, which was not inoculated but contracted the virus from adjacent, infected raccoons (Table 3). Raccoon 259 was humanely killed on 8 dpi because of an unrelated physical condition (tooth abscess), presumably before detectable antibodies were produced. Raccoon 256 shed virus only on 1 dpi yet developed detectable antibodies to AI virus (H4N8) by 9 dpi. However, the other raccoon that shed virus on 1 dpi (275) did not develop a detectable immune response, which indicated that virus detected in the swab was probably residual inoculum. Raccoons 263 and 262 had preexisting antibodies to a different subtype of AI virus that did not prevent infection and seroconversion to the other AI virus (H4N8) inoculum.
We observed no overt clinical signs of disease in these animals. Rectal temperatures showed no obvious trends and were probably confounded by stresses of anesthesia and handling. Most of the animals appeared lethargic, possibly because of confinement and manipulations occurring during daytime (raccoons are nocturnal). All other animals ate and drank well and most gained weight over the course of the experiment (data not shown).
Influenza Virus Receptors in Raccoons
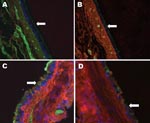
The predominant receptor for AI virus is SA linked α2,3 to galactose. In waterfowl, these receptors are located primarily in intestinal epithelium, which is why AI is primarily a disease of the digestive tract in avian species. In contrast, humans have SA linked α2,6 to galactose that is located predominantly in the respiratory system (2). Tissues from raccoon respiratory tracts were examined for avian and human influenza virus receptors by staining with lectins specific for each type of receptor (Figure). Raccoons have both receptor types in their respiratory systems, similar to swine but with uneven distribution among tissues. In the upper trachea epithelium, the overwhelmingly predominant receptor is the human type SAα2,6 (Figure, panel A). As one examines tissues from deeper in the respiratory tract, increasing amounts of the avian SA α2,3 receptor are found until the 2 types of receptors are in roughly equal amounts in the lungs (Figure, panels B, C, and D).
Experimental Infection of Raccoons with Human Influenza Virus
The presence and distribution of human type receptors in raccoons led us to infect a new cohort of raccoons with human influenza virus (H3N2). Daily monitoring showed that inoculated animals shed virus nasally for up to 8 dpi (Table 4, Figure). The amounts of virus shed were larger than in the AI experimental infection study but no transmission to either co-housed, virus-free raccoon was detected. All 4 inoculated animals subsequently developed antibodies against this virus by 14 dpi (data not shown). One raccoon (272) shed small amounts of virus rectally (0.25 EID50 equivalents) on 5 dpi, but no other rectal shedding of virus was detected. As with AI virus infection, no obvious clinical signs of disease were observed in these animals. Infected raccoons were also capable of shedding moderate amounts of human influenza virus, although no transmission to virus-free animals was observed.
The ecology of AI is complicated. Knowledge of the roles of wild birds and mammals in the epidemiology of the disease and how viral reassortants and variants arise are critical for the planning and preparation of future pandemics, vaccine development, and meaningful human health and agricultural risk assessments (9,10). However, other than a survey of small rodents in Pennsylvania, New Jersey, Maryland, and Virginia after an outbreak of influenza caused by virus subtype H5N2 in 1983–84 (11), no systematic investigation of wild mammals in influenza disease ecology has been performed.
Raccoons can carry a variety of etiologic agents. In Florida, raccoons are known to harbor 132 parasites, disease agents, and environmental contaminants, more than any other species of wild mammal (12). Viral diseases include rabies, canine distemper, pseudorabies, and poxvirus disease. To this list we can add West Nile virus (13,14) and now, from this study, avian and human influenza viruses.
The serologic survey of raccoons for AI virus exposure showed geographic variation in prevalence. AI in wild birds is relatively common; as much as 30% of the local waterfowl population can be infected (15). Raccoons often reside in these areas and can contact AI virus from their food and environment. However, the premise that areas of high waterfowl concentrations promote high antibody prevalence in raccoon populations was not always supported by these data. Raccoons in Georgia were sampled from the northwestern corner of the state, where wild fowl populations are small, and the prevalence of antibodies was 0%. In Maryland, which has one of the highest populations of overwintering and migrating waterfowl on its east coast (16), the prevalence of antibodies was 2.4%. Thus, data from these 2 states were logical on the basis of the waterfowl population size. However, Texas and California, with large seasonal populations of waterfowl, showed no evidence of AI virus exposure in raccoons. Wyoming and Colorado, with relatively small waterfowl populations, had the highest exposure rates of any states examined (25% and 12.8%, respectively). The reasons for higher prevalences in Wyoming and Colorado are unclear but may be related to concentrations of raccoons and waterfowl in riparian corridors in these semi-arid areas.
Wild waterfowl are the primary natural reservoir of AI virus, and different subtypes to which these raccoons were exposed are relatively common in avian populations (17–22). Clearly, raccoons are exposed to AI virus in the wild, and experimental studies confirm they can become infected with this virus and shed virus capable of infecting healthy animals. Also, we showed that raccoons can become infected with human influenza virus and shed moderate amounts of virus. The higher amounts of human influenza virus shed by raccoons than AI virus may indicate that human influenza virus is better adapted to mammalian physiology. The fact that we detected measurable levels of viral shedding with avian and human influenza viruses in infected raccoons is important. If one considers that only 2 uninfected raccoons were available to detect transmission of human influenza virus in this study, the fact that we did not detect transmission does not rule out the possibility that human influenza virus is also capable of being transmitted by raccoons and warrants additional research.
The abundance and distribution of avian and human influenza receptors found in raccoon tissues are similar to those in human respiratory tracts (23,24). The presence of human and AI virus receptors in raccoon respiratory systems creates the possibility of co-infection with multiple types of influenza virus and, as in swine, genetic reassortment and creation of new, possibly highly virulent strains are distinct risks.
Risks associated with wild raccoons and influenza are compounded by several factors. Raccoons are highly mobile with relatively large home ranges that include a variety of ecologic landscapes (5). They routinely travel between wetlands, forests, agricultural operations, and urban and suburban settings. Consequently, a raccoon that acquired AI virus in a marsh from scavenging a diseased bird could easily transport and transmit the virus to poultry and swine operations and to residential areas.
Raccoons apparently are not adversely affected by low pathogenic AI or human influenza viruses and thus remain active and potentially able to transmit virus over large areas. Because of their nocturnal habits, raccoons can be largely invisible to humans but can achieve large population densities. In fact, in some areas more raccoons can inhabit suburban areas than rural areas, reaching >90 raccoons/km2 (25,26).
In summary, the raccoon, a common, peridomestic, wild mammal is capable of becoming infected, transporting, and potentially transmitting avian and human influenza viruses. The risks associated with raccoons and influenza to agriculture and human health are unknown but clearly warrant further research. These results underscore the importance of investigating the roles of other peridomestic species in the disease ecology of influenza.
Dr Hall is a research virologist at the US Geological Survey National Wildlife Health Center, Madison, Wisconsin. His main research interests include ecology of infectious diseases, especially in regard to wildlife, vectors, and interactions and risks to humans.
Acknowledgment
We thank J. Gilman, M. Hudgins, K. Choate, D. Long, D. Emanueli, J. Smith, K. Sullivan, S. Healey, D. Kavanaugh, J. Pedersen, and the staff at the National Veterinary Services Laboratories for outstanding contributions, and the numerous private and public land stewards who made this study possible.
References
- Stallknecht DE, Kearney MT, Shane SM, Zwank PJ. Effects of pH, temperature, and salinity on persistence of avian influenza virus in water. Avian Dis. 1990b;34:412–8. DOIPubMedGoogle Scholar
- Baigent SJ, McCauley JW. Influenza type A in humans, mammals and birds: determinants of virus virulence, host-range and interspecies transmission. Bioessays. 2003;25:657–71. DOIPubMedGoogle Scholar
- Ito T, Nelson J, Couceiro SS, Kelm S, Baum LG, Krauss S, Molecular basis for the generation in pigs of influenza A viruses with pandemic potential. J Virol. 1998;72:7367–73.PubMedGoogle Scholar
- Horimoto T, Kawaoka Y. Pandemic threat posed by avian influenza A viruses. Clin Microbiol Rev. 2001;14:129–49. DOIPubMedGoogle Scholar
- Kaufmann JH. Raccoon and allies. In: Chapman JA Feldhamer GA, editors. Wild mammals of North America. Baltimore: Johns Hopkins University Press; 1982. p. 567–85.
- Beard CW. Demonstration of type-specific influenza antibody in mammalian and avian sera by immunodiffusion. Bull World Health Organ. 1970;42:779–85.PubMedGoogle Scholar
- Spackman E, Senne DA, Myers TJ, Bulaga LL, Garber LP, Perdue ML, Development of a real-time reverse transcriptase PCR assay for type A influenza virus and the avian H5 and H7 hemagglutinin subtypes. J Clin Microbiol. 2002;40:3256–60. DOIPubMedGoogle Scholar
- United States Department of Agriculture. 2006. Wild bird plan: an early detection system for highly pathogenic H5N1 avian influenza in wild migratory birds: U.S. interagency strategic plan. News release no. 0094.06 [cited 2008 Aug 11]. Available from http://www.usda.gov
- Scholtissek C, Koennecke I, Rott R. Host range recombinants of fowl plague (influenza A) virus. Virology. 1978;91:79–85. DOIPubMedGoogle Scholar
- Reid AH, Fanning TG, Hultin JV, Taubenberger JK. Origin and evolution of the 1918 “Spanish” influenza virus hemagglutinin gene. Proc Natl Acad Sci U S A. 1999;96:1651–6. DOIPubMedGoogle Scholar
- Nettles VF, Wood JM, Webster RG. Wildlife surveillance associated with an outbreak of lethal H5N2 avian influenza in domestic poultry. Avian Dis. 1985;29:733–41. DOIPubMedGoogle Scholar
- Forrester DJ. 1992. Parasites and diseases of wild mammals in Florida. Gainesville (FL): University Press of Florida; 1992. p. 123–50.
- Root JJ, Hall JS, McLean RG, Marlenee NL, Beaty BJ, Gansowski J, Serologic evidence of exposure of wild mammals to flaviviruses in the central and eastern United States. Am J Trop Med Hyg. 2005;72:622–30.PubMedGoogle Scholar
- Bentler KT, Hall JS, Root JJ, Klenk K, Schmitt B, Blackwell BF, West Nile virus seroprevalence in North American mesopredators. Am J Trop Med Hyg. 2007;76:173–9.PubMedGoogle Scholar
- Webster RG, Bean WJ, Gorman OT, Chambers TM, Kawaoka Y. Evolution and ecology of influenza A viruses. Microbiol Rev. 1992;56:152–79.PubMedGoogle Scholar
- Stewart RE. Waterfowl of the Chesapeake Bay. Chesapeake Science Supplement. 1972;13:S134–7. DOIGoogle Scholar
- Ito T, Okazaki K, Kawaoka Y, Takada A, Webster RG, Kida H. Perpetuation of influenza A viruses in Alaskan waterfowl reservoirs. Arch Virol. 1995;140:1163–72. DOIPubMedGoogle Scholar
- Stallknecht DE, Shane SM, Zwank PJ, Senne DA, Kearney MT. Avian influenza viruses from migratory and resident ducks of coastal Louisiana. Avian Dis. 1990;34:398–405. DOIPubMedGoogle Scholar
- Graves IL. Influenza viruses in birds of the Atlantic flyway. Avian Dis. 1992;36:1–10. DOIPubMedGoogle Scholar
- Slemons RD, Hansen WR, Converse KA, Senne DA. Type A influenza virus surveillance in free-flying, nonmigratory ducks residing on the eastern shore of Maryland. Avian Dis. 2003;47(Suppl):1107–10. DOIPubMedGoogle Scholar
- Krauss S, Walker D, Pryor SP, Niles L, Chenghong L, Hinshaw VS, Influenza A viruses of migrating wild aquatic birds in North America. Vector Borne Zoonotic Dis. 2004;4:177–89. DOIPubMedGoogle Scholar
- Hanson BA, Swayne DE, Senne DA, Lobpries DS, Hurst J, Stallknecht DE. Avian influenza viruses and paramyxoviruses in wintering and resident ducks in Texas. J Wildl Dis. 2005;41:624–8.PubMedGoogle Scholar
- Shinya K, Ebina M, Yamada S, Ono M, Kasai N, Kawaoka Y. Avian flu: influenza virus receptors in the human airway. Nature. 2006;440:435–6. DOIPubMedGoogle Scholar
- van Riel D, Munster VJ, de Wit E, Rimmelzwaan GF, Fouchier RA, Osterhaus AD, Human and avian influenza viruses target different cells in the lower respiratory tract of humans and other mammals. Am J Pathol. 2007;171:1215–23. DOIPubMedGoogle Scholar
- Riley SP, Hadidian J, Manski DA. Population density, survival, and rabies in raccoons in an urban national park. Can J Zool. 1998;76:1153–64. DOIGoogle Scholar
- Prange S, Gehrt SD, Wiggers EP. Demographic factors contributing to high raccoon densities in urban landscapes. J Wildl Manage. 2003;67:324–33. DOIGoogle Scholar
Figure
Tables
Cite This Article1Current affiliation: US Geological Survey National Wildlife Health Center, Madison, Wisconsin, USA.
Table of Contents – Volume 14, Number 12—December 2008
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Jeffrey S. Hall, US Geological Survey National Wildlife Health Center, 6006 Schroeder Rd, Madison, WI 53711, USA;
Top