Volume 17, Number 4—April 2011
Dispatch
Molecular Discrimination of Sheep Bovine Spongiform Encephalopathy from Scrapie
Figure 1
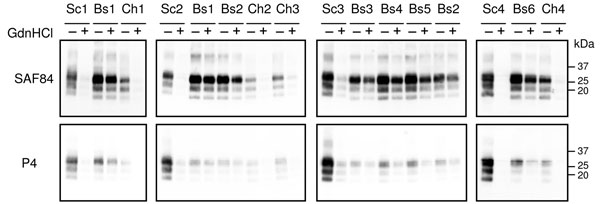
Figure 1. Representative Western blot showing the differential N-terminal proteinase K cleavage (monoclonal antibodies SAF84 vs. P4) and the susceptibility to denaturation of different transmissible spongiform encephalopathy isolates. Samples are indicated according to Table 2: classical scrapie isolates (Sc1, Sc2, Sc3, Sc4); experimental CH1641 (Ch1); CH1641-like isolates (Ch2, Ch3, Ch4); experimental sheep bovine spongiform encephalopathy by intracerebral transmission (Bs1) and oral transmission (Bs2, Bs3, Bs4, Bs5); natural goat isolate (Bs6). All samples were pretreated (+) or not treated (-) with 3.5 mol/L guanidine hydrochloride for 1 h at 37°C and then diluted to a final concentration of 0.35 mol/L guanidine hydrochloride, before digestion with proteinase K according to the Istituto Superiore di Sanità discriminatory method. Replica blots were probed with SAF84 (top) and P4 (bottom). Molecular weights are indicated on the right. GdnHCl, guanidine hydrochloride.