Volume 17, Number 5—May 2011
Research
Plasmodium knowlesi Malaria in Children
Abstract
Plasmodium knowlesi can cause severe malaria in adults; however, descriptions of clinical disease in children are lacking. We reviewed case records of children (age <15 years) with a malaria diagnosis at Kudat District Hospital, serving a largely deforested area of Sabah, Malaysia, during January–November 2009. Sixteen children with PCR-confirmed P. knowlesi monoinfection were compared with 14 children with P. falciparum monoinfection diagnosed by microscopy or PCR. Four children with knowlesi malaria had a hemoglobin level at admission of <10.0 g/dL (minimum lowest level 6.4 g/dL). Minimum level platelet counts were lower in knowlesi than in falciparum malaria (median 76,500/µL vs. 156,000/μL; p = 0.01). Most (81%) children with P. knowlesi malaria received chloroquine and primaquine; median parasite clearance time was 2 days (range 1–5 days). P. knowlesi is the most common cause of childhood malaria in Kudat. Although infection is generally uncomplicated, anemia is common and thrombocytopenia universal. Transmission dynamics in this region require additional investigation.
The simian malaria parasite Plasmodium knowlesi is increasingly recognized as a frequent cause of potentially fatal human malaria in adults in Malaysian Borneo (1–4). The infection has also been reported in peninsular Malaysia (5) and in other Southeast Asian countries, including Thailand (6,7), Myanmar (8,9), Vietnam (10), the Philippines (11), Indonesian Borneo (12–14), and Singapore (15,16). Until recently, P. knowlesi had been almost uniformly misdiagnosed by microscopy as P. malariae because of its morphologic similarities, leading to underestimations of prevalence (1,17). Accurate diagnosis therefore requires molecular methods.
The clinical and laboratory features of P. knowlesi infections in adults have been described in Kapit, Sarawak, where 107 (70%) of 152 adults with malaria were infected with P. knowlesi (3). Although P. knowlesi malaria was diagnosed in 8 children, the clinical and laboratory features were not described. All previously reported P. knowlesi infections that caused clinical disease have been in adults (1,2,6,8,11–13,15,18–20). In malaria caused by P. falciparum (21) and P. vivax (22), the 2 species that cause the greatest number of human malaria cases, well-described differences exist between adults and children in terms of the clinical epidemiology, disease spectrum, and laboratory manifestations of disease. We report the demographic, clinical, and laboratory features of P. knowlesi infection in children in Kudat, Sabah, a rural coastal farming area with little remaining primary rainforest, an epidemiologic setting that contrasts with the previously described forested areas of Sarawak.
Study Setting
The study was conducted at Kudat District Hospital (KDH) on the northeast tip of Sabah, Malaysia, a coastal rural area which has been largely deforested. KDH services 5 subdistricts (Tigapapan, Dualog, Matunggung, Tambuluran, and the island of Banggi), with a total population of 85,000 persons. The Rungus are the most common ethnic group on the mainland, and minority groups include ethnic Chinese, Bajaus, Dusuns, and Balabaks.
Ministry of Health policy in Sabah requires that all patients with a blood film result that indicates malaria be admitted to the hospital and discharged only after smear results are negative for 2 consecutive days. Since January 2009, in response to increasing reports of P. knowlesi infections and the difficulties of diagnosing this species by microscopy, it became policy at KDH to send slides that had been determined by microscopy to show presence of P. malariae to the Sabah State Reference Laboratory for PCR confirmation. In addition, KDH sends ≈15% of all blood films positive for other Plasmodium spp. for PCR confirmation.
Retrospective Case Review
We retrospectively searched laboratory microscopy records for all blood smear results positive for Plasmodium spp. during January 1–November 30, 2009. The patient’s age, sex, ethnicity, and address were recorded for all positive samples. Microscopy results were matched with PCR results.
Medical records were retrieved for all children <15 years of age who had received a diagnosis of malaria on the basis of microscopy results. Demographic, clinical, and laboratory details were extracted by using standardized data forms, which also included disease response to antimalarial treatment. Parasite clearance time was defined as the number of days until negative smear. Anemia and severe anemia were defined as hemoglobin levels <11 g/dL and <7.1 g/dL (3), respectively.
Laboratory Procedures
Blood films were examined by experienced laboratory microscopists at KDH with the parasite count being classified in most on a scale of 1 to 4 (1 = 4–40 parasites/µL, 2 = 41–400 parasites/µL, 3 = 401–4,000 parasites/µL, 4 = >4,000 parasites/µL), with accurate quantitation per microliter being recorded for most blood films that showed P. knowlesi, but for only a limited number that showed P. falciparum. Hemoglobin level and leukocyte and thrombocyte counts were measured on site by using automated systems (Sysmex XT1800 [Sysmex Corp., Mundelein, IL, USA] and CELL-DYN Sapphire [Abbott Diagnostics, Abbott Park, IL, USA]) At the Sabah State Reference Laboratory, parasite DNA was extracted, and nested PCR was performed for P. falciparum, P. vivax, P. malariae, P. ovale, and P. knowlesi by methods described (1,23).
Statistical Analysis
Data were analyzed by using Stata statistical software, version 10 (StataCorp LP, College Station, TX, USA). Proportions were compared by using the χ2 or Fisher exact test. Normally distributed and non–normally distributed variables were compared by using Student t test and Wilcoxon rank-sum test, respectively. For comparison between P. falciparum and P. knowlesi cases, the analysis included children with PCR-confirmed P. knowlesi infection, and children with P. falciparum infection diagnosed by either microscopy or PCR. Children with mixed Plasmodium infections were excluded from analysis, as were children whose medical records could not be located.
Malaria in All Age Groups
From January 1 through November 30, 2009, 220 patients at KDH were given a diagnosis of malaria on the basis of microscopy results (Figure 1). Of these, 196 (89%) had P. malariae monoinfection or mixed infection. PCR was performed on samples from 157 (80%) of these patients and results were positive for P. knowlesi in 137 (87%); 125 (91%) of these were P. knowlesi monoinfections. For the remaining 20 patients who had been given a diagnosis of P. malariae monoinfection or mixed infection by microscopy, PCR found undifferentiated Plasmodium spp., negative results for Plasmodium spp., and 7 cases of P. falciparum infection.
To estimate the final numbers of malaria species, we used positive PCR results when possible and microscopy results when PCR had not been performed (replacing P. malariae with P. knowlesi). Microscopy results were also used if PCR result was negative (5 P. knowlesi infections and 1 P. knowlesi/P. falciparum infection) or if PCR result was positive for Plasmodium spp. (7 P. knowlesi infections). Using this method, we found 172 (78%) P. knowlesi monoinfections, 29 (13%) P. falciparum monoinfections, and 19 (9%) mixed infections (Figure 1). A greater proportion of patients with P. knowlesi malaria were male (123/172 [72%]) than were those with P. falciparum malaria (15/29 [52%]; p = 0.03). Median age was higher for those with P. knowlesi infection (median 32 years, interquartile range [IQR] 19–49 years) than for those with P. falciparum infection (median 11 years, IQR 6–30 years).
Malaria in Children
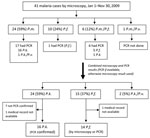
Of 220 patients with positive results by microscopy, 41 (19%) were <15 years of age. Microscopy showed that 24 (59%) children had P. malariae monoinfection, 10 (24%) had P. falciparum monoinfection, and 7 (17%) had mixed infections (Figure 2). Samples from 17 children with P. malariae monoinfection underwent PCR; 16 (94%) showed P. knowlesi, and 1 showed mixed P. knowlesi/P. vivax infection. Again, final numbers of malaria species were estimated by using PCR results (if available) or microscopy (if PCR had not been performed). Accordingly, 24 cases (59%) were P. knowlesi, 15 (37%) were P. falciparum, and 2 were mixed P. knowlesi/P. vivax infections. Children thus represented 14% (24/172) of all cases of P. knowlesi monoinfection.
Further analysis was confined to those children with PCR-confirmed P. knowlesi monoinfection and children with P. falciparum monoinfection diagnosed by either microscopy or PCR. Medical records were unavailable for 2 children (1 with P. knowlesi and 1 with P. falciparum infection), leaving 16 children with PCR-confirmed P. knowlesi infection and 14 with P. falciparum infection for comparison (Table 1).
Demographic Characteristics of Children with P. knowlesi Malaria
Half of the children with PCR-confirmed P. knowlesi malaria were male (Table 1). The mean age was 8.9 (95% confidence interval 7.6–10.3) years. The youngest child with P. knowlesi infection was 4 years; all others (15/16; 94%) were 7–14 years of age. In contrast, children with P. falciparum malaria were significantly younger, with a mean age of 5.4 years (95% confidence interval 3.5–7.3; range 9 months–11 years; p = 0.002).
Clinical Features of P. knowlesi and P. falciparum Malaria
All children had fever or a history of fever. The duration of fever before hospital admission appeared shorter with knowlesi malaria than with falciparum malaria (median 4 days vs. 6.5 days), although this difference was not statistically significant. Although documentation of clinical features was limited, no child was in a state of shock, and no child was reported to be dyspneic, to have bleeding complications, or to have any other clinical feature or laboratory results that indicated severe malaria (24). None of the children who had either knowlesi or falciparum malaria died.
Anemia
In 11 (69%) children with P. knowlesi malaria, the parasite density was accurately determined at hospital admission, with a median of 2,240 (IQR 480–7,200) parasites/µL. Only 2 children with P. falciparum malaria had parasite densities accurately determined at admission (200 and 1,600 parasites/µL). Anemia was common with P. knowlesi and P. falciparum infections. At admission, 9 (56%) children with P. knowlesi infection were anemic (hemoglobin level <11.0 g/dL), and 4 (25%) had a hemoglobin level <10 g/dL. All children with P. knowlesi infection had a hemoglobin minimum level <11.0 g/dL, and 10 (63%) had a hemoglobin minimum level <10 g/dL. The lowest hemoglobin level was found in an 8-year-old boy: 8.3 g/dL at admission, 6.4 g/dL on day 3, and 7.2 g/dL on day 6, the day before discharge. His parasite count at admission (14,400 parasites/µL) was the highest recorded in this study.
In addition, severe anemia (hemoglobin 6.0 g/dL at admission; minimum level 4.9 g/dL on day 1 requiring transfusion) occurred in a second child with positive results for P. knowlesi by microscopy (this child was not included in the final analysis because of lack of PCR confirmation). Among children with P. knowlesi infection, a lower hemoglobin minimum level was associated with higher parasite density at admission (p = 0.001). All children with P. falciparum infection had hemoglobin levels at admission <11.0 g/dL; 10 (71%) had hemoglobin levels <10 g/dL, and 5 (36%) had severe anemia (hemoglobin <7.1 g/dL). Severe anemia developed in a sixth child after admission. Four children received transfusions, with hemoglobin levels of 4.8–6.1 g/dL at admission. Children with P. knowlesi malaria took longer to reach their hemoglobin minimum level than did children with P. falciparum malaria (2.6 vs 1.5 days; p = 0.02).
Thrombocytopenia
All children with P. knowlesi malaria had thrombocytopenia. Fifteen children (94%) had a platelet count <150,000/µL at admission, and the remaining child exhibited thrombocytopenia within 1 day. The lowest platelet count recorded was 28,000/µL. Platelet count was not correlated with hemoglobin level. Thrombocytopenia was less common in children with P. falciparum; 7 (50%) had platelet counts <150,000/µL at admission or during hospitalization and the lowest platelet count was 47,000/µL. Children with P. knowlesi malaria had a lower lymphocyte count minimum than those with P. falciparum malaria (1.6 vs. 2.4 × 103/µL; p = 0.01).
Response to Treatment
Most (81%) children with P. knowlesi malaria were given oral chloroquine and primaquine for 3 days, and these children had a median parasite clearance time of 2 days (Table 2). The longest parasite clearance time with chloroquine and primaquine was 5 days in the aforementioned 8-year-old boy with the highest admission parasitemia level (14,400/µL) and the lowest minimum hemoglobin level (6.4 g/dL). Three children with P. knowlesi infection were given oral quinine, and parasites cleared within 2 days. Among the 11 children with P. knowlesi and parasite densities accurately assessed at admission, the correlation between parasite density and parasite clearance time was significant (p = 0.002).
Children with P. falciparum malaria were given a variety of treatment regimens. Most (71%) received a regimen that contained quinine, 7 (50%) children received sulfadoxine/pyrimethamine, 6 (43%) received primaquine, and 5 (36%) were given chloroquine. Only 5 (36%) received artemisinin-based combination therapy. Parasite clearance times were significantly slower among children with P. falciparum malaria, with a median of 5 days until the first negative smear; in 4 children (29%), it took >10 days for parasites be cleared. Children with P. knowlesi malaria had significantly shorter hospital admissions (median 4 days, IQR 4–5 days) than did children with P. falciparum malaria (median 7 days, IQR 5–10 days).
P. knowlesi was the most common cause of malaria in adults and children in the Kudat region in Sabah, Malaysian Borneo. Although those with P. knowlesi infections had an older age distribution than did those with P. falciparum infections, the species still caused 63% of all malaria cases among children <15 years. Although nearly all previous reported cases of knowlesi malaria have been in adults, 14% of all knowlesi cases in Kudat occurred in children. In children, P. knowlesi most often caused uncomplicated malaria, which responded well to chloroquine. Nevertheless, knowlesi malaria was associated with substantial illness in children, with all PCR-confirmed P. knowlesi–infected children being anemic at admission or during hospital stay.
In the only other report of knowlesi malaria in children, a prospective study of adult knowlesi malaria in Sarawak reported the exclusion of 8 children <15 years with P. knowlesi infection, comprising 7% of all knowlesi cases (3). The clinical and laboratory features of P. knowlesi malaria in children have not been described. Consistent with the reported features of P. knowlesi malaria in adults (3), the disease in most children was uncomplicated. In adults with knowlesi malaria, increasing age is a risk factor for severe disease. Although the numbers are small, none of the children had severe manifestations of knowlesi malaria that have been reported in adults (3), such as acute lung injury or acute renal failure. In falciparum malaria, these conditions are also largely confined to adults (21); anemia, coma, and acidosis-related respiratory distress are the major manifestations of severe falciparum malaria in children. No child with knowlesi malaria exhibited coma or respiratory distress; however, anemia developed in all children with knowlesi malaria, with the hemoglobin concentration in 1 patient (6%) falling to <7.0 g/dL. Anemia was more common in children than has been previously described in knowlesi malaria in adults (3).
Thrombocytopenia was found at admission in nearly all (94%) children with P. knowlesi malaria, in contrast to only half of the children with P. falciparum malaria. Although the cause is unclear, thrombocytopenia is also nearly universal in infected adults (3), which makes it a characteristic feature of P. knowlesi infection across all age groups. The role of thrombocytopenia and platelet activation in the pathogenesis of knowlesi malaria requires further investigation.
Most children with P. knowlesi malaria had an adequate response to a 3-day regimen of treatment with chloroquine and primaquine, although the mean parasite clearance time of 2 days was longer than the 90% parasite clearance time of 10.3 hours that was recently reported in adults (25). One child who received chloroquine and primaquine, and had a high parasite count at admission, required 5 days to clear parasites. Standard Ministry of Health pediatric dosing regimens of chloroquine are used in Kudat; however, posttreatment vomiting or inadequate blood concentrations could not be excluded. Prospective studies that evaluate the response to chloroquine and artemisinin-based combination therapy in pediatric knowlesi malaria are required. Children with P. falciparum malaria received many different treatment regimens and took significantly longer to clear their parasites. Less than half received the recommended artemisinin-based combination therapy, and only after alternative treatments failed. Children with P. falciparum malaria had significantly longer hospital stays, likely related at least in part to suboptimal treatment regimens. This finding highlights the importance of increasing the usage of artemisinin-based combination therapy for falciparum malaria in district hospitals in Sabah and elsewhere.
Our study had several limitations. First, PCR was only performed for 73% of cases across all age groups, and this limited our ability to accurately determine the true proportion of disease caused by P. knowlesi. Furthermore, although PCR was performed on samples from most children with P. knowlesi malaria, only 1 child with P. falciparum malaria had PCR performed. Our analysis, therefore, compared children with PCR-confirmed knowlesi malaria to children with falciparum malaria diagnosed by either microscopy or PCR. Some of those children with a diagnosis of falciparum malaria may have actually had knowlesi malaria, and the differences found between these 2 species may therefore be minimum estimates. The retrospective design of our study also limited our ability to collect standardized clinical information.
This study demonstrates that P. knowlesi has become the predominant malaria species in the Kudat region, estimated by results of microscopy, PCR, or both, as contributing to 87% of all malaria cases, a higher proportion than that reported elsewhere in Malaysian Borneo. The emerging dominance of P. knowlesi in Malaysian Borneo has been hypothesized to result from the following factors: changing patterns of human exposure to monkeys and vectors (26) because of deforestation, and potentially reduced competition or cross-species protection from P. vivax and P. falciparum as a result of a 40-fold reduction in the prevalence of these species in Sabah and Sarawak during 1960–2006 following intensive malaria control efforts (27). The paucity of P. vivax in Kudat was particularly notable.
Previous reports of adult disease have been from communities adjacent to rainforests (3). Kudat is a coastal rural farming area with varied land use and vegetation patterns and with minimal remaining regrowth forest. Although our retrospective study did not gather detailed travel or exposure histories, it is likely that most pediatric infections were locally acquired and that infections with P. knowlesi did not occur solely in those spending time in forested areas. Macaque monkeys, the natural hosts of P. knowlesi, are widely distributed in different habitats throughout the Kudat area and are frequently domesticated. The major vectors of P. knowlesi in forested areas of Sarawak, Anopheles latens mosquitoes, disappear with deforestation, but vectors capable of transmitting P. knowlesi, An. balabacencis mosquitoes (26), do persist at lower densities in largely deforested areas of Sabah (28,29). In notable contrast to P. falciparum malaria, pediatric knowlesi malaria was restricted to children of school age. Further studies will be required to characterize the transmission patterns, vectors, and risk factors for P. knowlesi in deforested areas of Malaysia.
P. knowlesi is the most common cause of malaria in adults and children in the Kudat region of Sabah, a rural coastal deforested region. Consistent with previous studies in adults (3), we found that P. knowlesi in children most often caused uncomplicated malaria that responded adequately to chloroquine and primaquine. Anemia was common in children and knowlesi infection was associated with moderately severe anemia. Thrombocytopenia was universal and is characteristic of knowlesi malaria across all age groups. Larger prospective clinical studies are needed to describe more fully the epidemiology of P. knowlesi malaria in children, the full spectrum of clinical disease and the transmission patterns in nonforested areas such as Kudat.
Dr Barber is an infectious diseases physician and PhD scholar at Menzies School of Health Research, Darwin, and Clinical Research Centre, Kota Kinabalu, Malaysia. Her primary research interest is P. knowlesi malaria.
Acknowledgments
We thank Fread Anderios, Ella Curry, and the laboratory staff at Kudat District Hospital for their help with this study, and Jeffrey Hii for helpful discussions. We also thank the Director General of Health (Malaysia) for permission to publish this article.
National Health and Medical Research Council of Australia (fellowships to T.W.Y. and N.M.A.; scholarship to B.E.B.; and Program Grant 496600)
References
- Cox-Singh J, Davis TM, Lee KS, Shamsul SS, Matusop A, Ratnam S, Plasmodium knowlesi malaria in humans is widely distributed and potentially life threatening. Clin Infect Dis. 2008;46:165–71. DOIPubMedGoogle Scholar
- Cox-Singh J, Hiu J, Lucas S, Divis P, Zulkarnaen M, Chandran P, Severe malaria-a case of fatal Plasmodium knowlesi infection with post-mortem findings. Malar J. 2010;9:10. DOIPubMedGoogle Scholar
- Daneshvar C, Davis TM, Cox-Singh J, Rafa’ee M, Zakaria S, Divis P, Clinical and laboratory features of human Plasmodium knowlesi infection. Clin Infect Dis. 2009;49:852–60. DOIPubMedGoogle Scholar
- William T, Menon J, Rajahram G, Chan L, Ma G, Donaldson S, Severe Plasmodium knowlesi malaria in a tertiary hospital, Sabah, Malaysia. Emerg Infect Dis. In press.
- Vythilingam I, Noorazian YM, Huat TC, Jiram AI, Yusri YM, Azahari AH, Plasmodium knowlesi in humans, macaques and mosquitoes in peninsular Malaysia. Parasit Vectors. 2008;1:26. DOIPubMedGoogle Scholar
- Jongwutiwes S, Putaporntip C, Iwasaki T, Sata T, Kanbara H. Naturally acquired Plasmodium knowlesi malaria in human, Thailand. Emerg Infect Dis. 2004;10:2211–3.PubMedGoogle Scholar
- Putaporntip C, Hongsrimuang T, Seethamchai S, Kobasa T, Limkittikul K, Cui L, Differential prevalence of Plasmodium infections and cryptic Plasmodium knowlesi malaria in humans in Thailand. J Infect Dis. 2009;199:1143–50. DOIPubMedGoogle Scholar
- Zhu HM, Li J, Zheng H. Human natural infection of Plasmodium knowlesi [in Chinese] [PMID: 16866152]. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi. 2006;24:70–1.PubMedGoogle Scholar
- Jiang N, Chang Q, Sun X, Lu H, Yin J, Zhang Z, Co-infections with Plasmodium knowlesi and other malaria parasites, Myanmar. Emerg Infect Dis. 2010;16:1476. DOIPubMedGoogle Scholar
- Van den Eede P, Van HN, Van Overmeir C, Vythilingam I, Duc TN, Hung LX, Human Plasmodium knowlesi infections in young children in central Vietnam. Malar J. 2009;8:249. DOIPubMedGoogle Scholar
- Luchavez J, Espino F, Curameng P, Espina R, Bell D, Chiodini P, Human Infections with Plasmodium knowlesi, the Philippines. Emerg Infect Dis. 2008;14:811–3. DOIPubMedGoogle Scholar
- Figtree M, Lee R, Bain L, Kennedy T, Mackertich S, Urban M, Plasmodium knowlesi in human, Indonesian Borneo. Emerg Infect Dis. 2010;16:672.PubMedGoogle Scholar
- Bronner U, Divis P, Färnert A, Singh B. Swedish traveller with Plasmodium knowlesi malaria after visiting Malaysian Borneo. Malar J. 2009;8:15. DOIPubMedGoogle Scholar
- Sulistyaningsih E, Fitri L, Löscher T, Berens-Riha N. Diagnostic difficulties with Plasmodium knowlesi infection in humans. Emerg Infect Dis. 2010;16:1033.PubMedGoogle Scholar
- Ng OT, Ooi E, Lee C, Lee P, Ng L, Pei S, Naturally acquired human Plasmodium knowlesi infection, Singapore. Emerg Infect Dis. 2008;14:814–6. DOIPubMedGoogle Scholar
- Lee YC, Tang CS, Ang LW, Han HK, James L, Goh KT. Epidemiological characteristics of imported and locally-acquired malaria in Singapore. Ann Acad Med Singapore. 2009;38:840–9.PubMedGoogle Scholar
- Lee K-S, Cox-Singh J, Brooke G, Matusop A, Singh B. Plasmodium knowlesi from archival blood films: further evidence that human infections are widely distributed and not newly emergent in Malaysian Borneo. Int J Parasitol. 2009;39:1125–8. DOIPubMedGoogle Scholar
- Galinski MR, Barnwell JW. Monkey malaria kills four humans. Trends Parasitol. 2009;25:200–4. DOIPubMedGoogle Scholar
- Kantele A, Marti H, Felger I, Muller D, Jokiranta T. Monkey malaria in a European traveler returning from Malaysia. Emerg Infect Dis. 2008;14:1434–6. DOIPubMedGoogle Scholar
- Thuy-Huong T, Ana S, Marwa A, María M, Marta L, Eduardo A, First case of detection of Plasmodium knowlesi in Spain by real time PCR in a traveller from Southeast Asia. Malar J. 2010;9:219. DOIPubMedGoogle Scholar
- Dondorp AM, Lee SJ, Faiz MA, Mishra S, Price R, Tjitra E, The relationship between age and the manifestations of and mortality associated with severe malaria. Clin Infect Dis. 2008;47:151–7. DOIPubMedGoogle Scholar
- Tjitra E, Anstey N, Sugiarto P, Warikar N, Kenangalem E, Karyana M, Multidrug-resistant Plasmodium vivax associated with severe and fatal malaria: a prospective study in Papua, Indonesia. PLoS Med. 2008;5:e128. DOIPubMedGoogle Scholar
- Singh B, Sung L, Matusop A, Radhakrishnan A, Shamsul S, Cox-Singh J, A large focus of naturally acquired Plasmodium knowlesi infections in human beings. Lancet. 2004;363:1017–24. DOIPubMedGoogle Scholar
- World Health Organization. Severe falciparum malaria. World Health Organization, Communicable Diseases Cluster. Trans R Soc Trop Med Hyg. 2000;94:S1–90.PubMedGoogle Scholar
- Daneshvar C, Davis T, Cox-Singh J, Rafa’ee M, Zakaria S, Divis P, Clinical and parasitological response to oral chloroquine and primaquine in uncomplicated human Plasmodium knowlesi infections. Malar J. 2010;9:238. DOIPubMedGoogle Scholar
- Indra V. Plasmodium knowlesi in humans: a review on the role of its vectors in Malaysia. Trop Biomed. 2010;27:1.PubMedGoogle Scholar
- Cox-Singh J, Singh B. Knowlesi malaria: newly emergent and of public health importance? Trends Parasitol. 2008;24:406–10. DOIPubMedGoogle Scholar
- Hii J, Macdonald M, Sang V. The use of CDC light traps for malariometric entomology surveys in Sabah, Malaysia. Trop Biomed. 1986;3:39–48.
- Vythilingam I, Tan C, Asmad M, Chan S, Lee K, Singh B. Natural transmission of Plasmodium knowlesi to humans by Anopheles latens in Sarawak, Malaysia. Trans R Soc Trop Med Hyg. 2006;100:1087–8. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 17, Number 5—May 2011
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Bridget E. Barber, Global Health Division, Menzies School of Health Research, PO Box 41096, Casuarina, Darwin, Northern Territory, Australia
Top