Volume 18, Number 12—December 2012
Dispatch
Differentiation of Prions from L-type BSE versus Sporadic Creutzfeldt-Jakob Disease
Figure 2
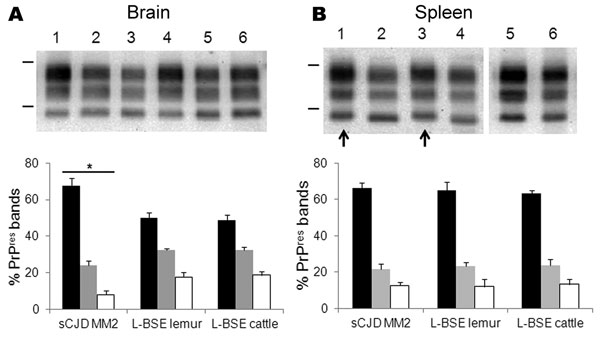
Figure 2. . . . Western blot molecular typing of protease-resistant prion protein (PrPres) in brain and spleen tissues of ovine prion protein–transgenic (TgOvPrP4) mice at second passage. PrPres from mice infected with MM2-cortical subtype sporadic Creutzfeldt-Jakob disease (sCJD), L-type bovine spongiform encephalopathy (L-BSE) from lemur, and L-BSE from cattle (02-2528) were compared in brain (A) and spleen (B) tissues (monoclonal antibody SHa31). Bars to the left of Western blots indicate the 29.0- and 20.1-kDa marker positions. A) Lanes 1, 4, sCJD MM2; lanes 2, 5, L-BSE from lemur; lanes 3, 6, L-BSE from cattle control; B) lanes 1, 3, sCJD MM2; lanes 2, 4, 6, L-BSE from lemur; lane 5, L-BSE from cattle control. C, D) Proportions of PrPres glycoforms in brain (C) and spleen (D) tissues. Error bars indicate SD. *Indicates p<0.0001 when comparing PrPres proportions from mice infected with MM2-cortical sCJD with those infected with L-BSE.