Volume 19, Number 5—May 2013
Dispatch
Mapping Environmental Suitability for Malaria Transmission, Greece
Abstract
During 2009–2012, Greece experienced a resurgence of domestic malaria transmission. To help guide malaria response efforts, we used spatial modeling to characterize environmental signatures of areas suitable for transmission. Nonlinear discriminant analysis indicated that sea-level altitude and land-surface temperature parameters are predictive in this regard.
Malaria was eliminated in Greece in 1974 (1,2); however, cases continue to be imported from countries to which malaria is endemic (3) and locally acquired cases have occurred sporadically (4,5). During 2009–2012, health authorities in Greece recorded 267 malaria cases. Although most cases were imported, at least 69 (26%) occurred in patients who did not have travel histories to malaria-endemic regions. A cluster of 6 locally acquired Plamosdium vivax malaria cases occurred during August–October 2009 in the southern Peloponnese (Evrotas Municipality, Lakonia district); in addition, 1 autochthonous case was reported from Marathon Municipality, East Attiki district (2). In 2010, locally transmitted cases were recorded in the same Lakonia district, 1 in East Attiki and 2 in children in central Greece (Viotia district). In 2011, a total of 42 autochthonous cases of P. vivax malaria were reported, representing 44% of the 96 notified cases in 2011. Most (36) of those cases were notified in the Evrotas municipality. In 2012, locally acquired cases appeared to have decreased, with 16 cases representing 21% of the overall number of cases. The ongoing transmission of P. vivax by local anopheline mosquitoes raises concern about reemergence of malaria transmission in Greece in areas that are hospitable to the vector and have permissive environmental and climatic conditions (6).
To guide malaria control efforts, we delineated areas suitable for malaria transmission in Greece using the place of exposure for 69 locally acquired malaria cases. A health officer administered a standardized questionnaire to each malaria case-patient in Greece to determine the origin of infection. Our analysis was restricted to cases classified as locally acquired in persons without travel histories to optimize the specificity of the model because our goal was to describe the environmental suitability of autochthonous malaria transmission in Greece. Thus, our analysis excluded cases that might have been acquired abroad.
We aimed to describe the environmental profile of areas with active transmission cycles during 2009–2012 and then to predict other areas at risk for malaria reemergence in Greece. Our data sources were countrywide georeferenced environmental and climatic information, all acquired from the European Environment and Epidemiology Network data repository and prepared for spatial modeling (7). Variables considered were temperature, vegetation seasonal variations, altitude, land-cover categories, and demographic indicators. Daytime and nighttime Land Surface Temperature and Normalized Difference Vegetation Index were retrieved from the 1 km–resolution long-term Temporal Fourier transformed imagery from Moderate Resolution Imaging Spectroradiometer (8). Altitude values were derived from the Global Land One-km Base Elevation Digital Elevation Model data from the US Geological Survey Earth Resources Observation Systems data center (http://eros.usgs.gov). The coordination of information on the environment (CORINE) land cover from the European Environment Agency provided the framework for land cover patterns exploratory analysis (www.eea.europa.eu/data-and-maps/data/corine-land-cover-2000-clc2000-seamless-vector-database-3). The population density grid of the Joint Research Centre was used as demographic indicator (www.eea.europa.eu/data-and-maps/data/population-density-disaggregated-with-corine-land-cover-2000-1).
The spatial scale for this study was a polygon equivalent to a circular buffer of 3.5 km (40,000 ha). This spatial resolution was considered appropriate to the relatively fine scale at which environmental variability can affect malaria transmission. The construction of the land-cover variables is described in the Technical Appendix. A disease risk map was generated by using nonlinear discriminant analysis available in eRiskMapper version 1.1.4 (www.tala.ox.ac.uk) and is described in detail in the online Technical Appendix.
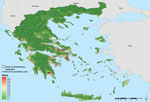
The first 10 best ranked variables comprised sea-level altitude and land-surface temperature (Table). Parameters of nighttime and daytime temperatures were predictive in this model; the annual variation (amplitude) and the mean absolute temperature values scored high in the ranking. Predicted suitability of areas for persistent malaria transmission based on these variables are characterized by low elevation; warmer temperatures (which might enable more rapid mosquito and parasite development); and intensive, year-round irrigated agriculture with complex cultivation patterns (generally requiring a high degree of manual labor) (Figure 1, Appendix). These are probable contributing factors to mosquitoe presence and, possibly, to malaria transmission.
The accuracy statistics of the bootstrap model revealed a sensitivity of 0.98 (where 1 denotes recognition of all actual presences) and specificity of 0.98 (where 1 denotes recognition of all actual absences). Moreover, historical presence of malaria in Greece, before disease elimination, was extracted from several sources and showed a partially coincident distribution pattern with the suitability map: 41% overlap in Peloponnese, 63% along the west coast of central Greece and Epirus, and 39% in eastern central Greece (Figure 2, Appendix). The best overlap was found along the west coast, but in the northern part, the model did not match the historical pattern very well. This discrepancy might be explained by the low rank of landscape pattern variables obtained into nonlinear discriminant analysis modeling.
We assessed environmental and climatic characteristics of the areas with autochthonous spread of P. vivax malaria in Greece during 2009–2012 and delineated similar areas possibly suitable for transmission in Greece. Sea-level altitude and the mean and annual variation of land-surface temperature for daytime and nighttime were predictors in our model.
A major limitation of our study is that it considers only environmental suitability for transmission, not risk for transmission per se. Our approach did not account for certain factors in this regard, for example, presence/absence of malaria vectors under collection in 2012. This partly reflects the lack of knowledge about what mosquito species were responsible for recent outbreaks, although historic work suggests Anopheles sacharovi, An. maculipennis s.s., and An. superpictus (9,10). Indeed, 2 of these species, An. sacharovi and An. superpictus, have been implicated as the probable dominant vectors throughout Greece and adjacent areas (11). Hence, species would seem to be reasonable targets for any mosquito surveillance efforts.
Our model did not highlight areas of northern Greece previously associated with malaria (4,5) as being particularly suitable for transmission. Further refinement of our approach, perhaps including vector information, might address this shortcoming. Further study on the malariogenic potential should be conducted to complete our assessment. Despite these limitations, this spatial analysis can be useful and can help guide the public health response to threats, such as malaria, by directing surveillance and control activities and/or by identifying uncertainties relevant to disease risk mapping and response.
Model accuracy and public health practice can be improved through vigilant disease and vector surveillance with timely case notification (12). Yet, despite these potential limitations, we believe that this spatial analysis is a useful tool; it can help guide response(s), integrated preparedness and response activities, including targeted epidemiologic and entomologic surveillance, vector control activities, and awareness rising among the general population and health care workers, in the areas environmentally suitable for transmission.
Dr Sudre is a scientific officer environmental determinants of communicable diseases at the European Centre for Disease Prevention and Control in Stockholm and an associate researcher at UMR 6249 Chrono-environnement Laboratory (Besançon, France). His research interests include health geography, as well as eco-epidemiology of infectious diseases and its integration in public health.
References
- Sabatinelli G, Ejov M, Joergensen P. Malaria in the WHO European Region (1971–1999). Euro Surveill. 2001;6:61–5.PubMedGoogle Scholar
- Danis K, Baka A, Lenglet A, Van Bortel W, Terzaki I, Tseroni M, Autochthonous Plasmodium vivax malaria in Greece, 2011. Euro Surveill. 2011;16:19993 .PubMedGoogle Scholar
- Vakali A, Patsoula E, Spanakos G, Danis K, Vassalou E, Tegos N, Malaria in Greece, 1975 to 2010. Euro Surveill. 2012;17:20322 .PubMedGoogle Scholar
- Kampen H, Maltezos E, Pagonaki M, Hunfeld KP, Maier WA, Seitz HM. Individual cases of autochthonous malaria in Evros Province, northern Greece: serological aspects. Parasitol Res. 2002;88:261–6. DOIPubMedGoogle Scholar
- Kampen H, Proft J, Etti S, Maltezos E, Pagonaki M, Maier WA, Individual cases of autochthonous malaria in Evros Province, northern Greece: entomological aspects. Parasitol Res. 2003;89:252–8 .PubMedGoogle Scholar
- Sinka ME, Bangs MJ, Manguin S, Chareonviriyaphap T, Patil AP, Temperley WH, The dominant Anopheles vectors of human malaria in the Asia-Pacific region: occurrence data, distribution maps and bionomic precis. Parasit Vectors. 2011;4:89.
- Semenza JC, Menne B. Climate change and infectious diseases in Europe. Lancet Infect Dis. 2009;9:365–75. DOIPubMedGoogle Scholar
- Scharlemann JP, Benz D, Hay SI, Purse BV, Tatem AJ, Wint GR, Global data for ecology and epidemiology: a novel algorithm for temporal Fourier processing MODros Inf Serv. data. PLoS ONE. 2008;3:e1408. DOIPubMedGoogle Scholar
- Alten B, Kempen H, Fontenille D. Malaria in southern Europe: resurgence from the past. In Takken W, Knols BGJ, editors. Emerging pests and vector-borne diseases in Europe. Wageningen (the Netherlands): Wageningen Academic Publishers; 2007. p. 35–58.
- Bruce-Chwatt L, Zulueta J, editors. The rise and fall of malaria in Europe. New York: Oxford University Press; 1980.
- Sinka ME, Bangs MJ, Manguin S, Rubio-Palis Y, Chareonviriyaphap T, Coetzee M, A global map of dominant malaria vectors. Parasit Vectors. 2012;5:69.
- Lindgren E, Andersson Y, Suk JE, Sudre B, Semenza JC. Public health. Monitoring EU emerging infectious disease risk due to climate change. Science. 2012;336:418–9. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 19, Number 5—May 2013
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Jan C. Semenza, European Centre for Disease Prevention and Control, Unit of Scientific Advice, Tomtebodavägen 11A, Stockholm S-171 83, Sweden
Top