Volume 19, Number 8—August 2013
Research
Comparison of 2 Assays for Diagnosing Rotavirus and Evaluating Vaccine Effectiveness in Children with Gastroenteritis
Figure 2
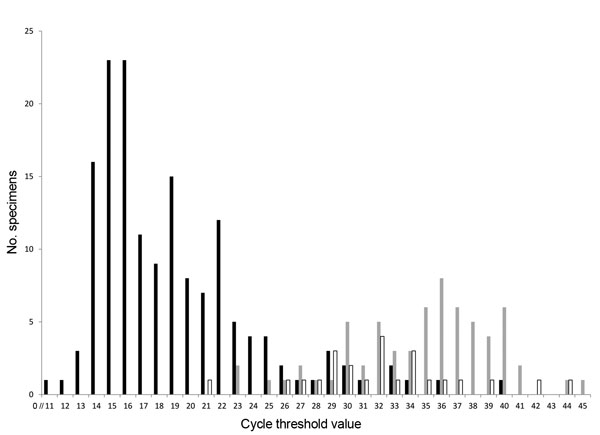
Figure 2. . . . Frequency distribution of Ct values for specimens in which rotavirus was detected by qRT-PCR, 3 New Vaccine Surveillance Network sites (USA), October 2008–October 2009. For 1 (1%) acute gastroenteritis EIA+ specimen, 425 (87%) acute gastroenteritis EIA− specimens, and 476 (95%) healthy control specimens, no virus was detected by qRT-PCR. Ct, cycle threshold; qRT-PCR, semiquantitative reverse transcription PCR; EIA, enzyme immunoassay; +, positive; −, negative. Black bars indicate acute gastroenteritis patients with EIA+ specimens, n = 157; gray bars indicate acute gastroenteritis patients with EIA− specimens, n = 65; white bars indicate healthy controls, n = 24.
Page created: July 02, 2013
Page updated: July 02, 2013
Page reviewed: July 02, 2013
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.