Volume 17, Number 8—August 2011
Research
Incidence of Acute Gastroenteritis and Role of Norovirus, Georgia, USA, 2004–2005
Cite This Article
Citation for Media
Abstract
Approximately 179 million cases of acute gastroenteritis (AGE) occur annually in the United States. However, lack of routine clinical testing for viruses limits understanding of their role among persons seeking medical care. Fecal specimens submitted for routine bacterial culture through a health maintenance organization in Georgia, USA, were tested with molecular diagnostic assays for norovirus, rotavirus, astrovirus, sapovirus, and adenovirus. Incidence was estimated by using national health care utilization rates. Routine clinical diagnostics identified a pathogen in 42 (7.3%) of 572 specimens; inclusion of molecular viral testing increased pathogen detection to 15.7%. Community AGE incidence was 41,000 cases/100,000 person-years and outpatient incidence was 5,400/100,000 person-years. Norovirus was the most common pathogen, accounting for 6,500 (16%) and 640 (12%) per 100,000 person-years of community and outpatient AGE episodes, respectively. This study demonstrates that noroviruses are leading causes of AGE among persons seeking medical care.
Acute gastroenteritis (AGE), defined as diarrheal disease of rapid onset potentially accompanied by nausea, vomiting, fever, or abdominal pain, is a major cause of illness in the United States. Approximately 179 million episodes of AGE occur each year and result in ≈600,000 hospitalizations and 5,000 deaths (1,2). A specific etiology is attributed to only ≈20% of AGE cases, although viruses are recognized as the most common of the known agents (1,3). Noroviruses, in particular, have been estimated to cause ≈21 million cases of AGE annually, including >56,000 hospitalizations and 570 deaths (3). However, these estimates are based on US estimates of AGE and extrapolation of etiologic fractions from studies in other industrialized countries because few laboratory-based data are available on the role of noroviruses in sporadic AGE in the United States.
Development of more precise disease incidence estimates for noroviruses and other viral causes of AGE have been hampered, in part, by the lack of diagnostic assays available in clinical settings. With the exception of an enzyme immunoassay (EIA) for rotavirus, diagnosis of viral AGE in the United States is made largely on the basis of clinical signs and symptoms. Molecular techniques used for definitive diagnosis, specifically PCR, are available mostly in public health laboratories and research settings. Commercial EIA kits for norovirus have been developed but are not widely available in the United States and are not cleared by the US Food and Drug Administration for diagnosis of sporadic AGE cases. Evaluation of viral AGE incidence is further limited by the fact that most AGE patients do not seek medical care, and, of those who do, <20% submit fecal specimens for diagnostics (2). Lastly, general perception is that norovirus gastroenteritis is a self-limiting mild illness that rarely requires medical attention, despite several reports of serious illness and death in various settings (3–7).
Better understanding of the relative role of specific viral causes of AGE among persons seeking medical care is needed to help guide clinical management and ultimately to develop more appropriate AGE prevention strategies. We sought to determine the prevalence of viral pathogens among AGE patients who sought medical care and identify their trends in seasonality and molecular epidemiology. Using a similar strategy as that used for common bacterial and parasitic causes of AGE (3), we further sought to estimate incidence of viral agents of AGE in community and outpatient settings.
Study Population and Sample Selection
Kaiser Foundation Health Plan of Georgia, Inc. (KP) is a health maintenance organization with ≈280,000 members in Georgia, USA, who almost exclusively seek care with KP. One microbiology laboratory serves the entire population of Georgia KP members and receives ≈140 fecal specimens each month from outpatients seeking medical care. Upon order of fecal diagnostics by an outpatient clinician, patients were provided fecal collection kits and instructions for their in-home collection. Patients were instructed to keep specimens refrigerated after collection and to return them as soon as possible to the clinic, typically within 48 hours. Specimens were then transported by same-day courier to the KP microbiology laboratory for processing.
Each week during March 15, 2004–March 13, 2005, a total of 11 specimens from different patients were randomly selected for inclusion in the study. A target sample size of 572 was selected to identify 10% norovirus prevalence with a 95% confidence interval of ± 5%, assuming patients with AGE of short duration were 4× less likely to submit samples than those with AGE of longer duration. Only patients for whom a specimen was submitted after a clinician order for routine culture were eligible for inclusion, although additional diagnostics (e.g., ova and parasites, Clostridium difficile, rotavirus EIA) may have also been ordered for some patients. The following data were obtained for each specimen: days from outpatient visit to receipt at the KP laboratory, week of receipt by the KP laboratory, patient age group in 5-year intervals, patient sex, fecal consistency, and results of any diagnostic tests performed by the KP laboratory. Data and specimens sent to the Centers for Disease Control and Prevention (CDC), Atlanta, GA, USA, were anonymous and had no identifiable information that could be linked to the patient. Thus, our study did not require review by an institutional review board.
Laboratory Testing
All specimens submitted for routine culture were tested for Campylobacter, Shigella, and Salmonella spp. at the KP laboratory. When requested, specimens were also tested for Giardia and Cryptosporidium spp. by using ProSpec T Microplate Assays (Remel, Lenexa, KS, USA), for rotavirus by using the Immunocard STAT Rotavirus Test (Meridian Bioscience Inc., Cincinnati, OH, USA), and for C. difficile toxigenic strains by using the C. difficile Tox A/B 11 EIA (Wampole Laboratories LLC, Princeton, NJ, USA). Specimens submitted in Cary Blair medium (i.e., routine bacterial culture) then underwent molecular testing for norovirus, rotavirus, sapovirus, astrovirus and enteric adenovirus at the CDC laboratory.
Viral nucleic acid was extracted by using RNA spin columns (Omega Bio-Tek, Doraville, GA, USA). TaqMan real-time reverse transcription PCR and PCR were used for initial sample screening for genogroup I (GI) and GII noroviruses, sapoviruses, and adenoviruses, as described (8–10). Conventional PCR was conducted to screen samples for astrovirus and rotavirus group A and to determine the genotype of norovirus-, sapovirus-, and adenovirus-positive samples (11–15). PCR products were purified by using the QIAquick Gel Extraction Kit (QIAGEN, Valencia, CA, USA) and sequenced by using the BigDye Terminator Cycle Sequencing Ready Reaction Kit (Applied Biosystems, Foster City, CA, USA) according to the manufacturers’ instructions. For genotyping, detected virus sequences were compared with sequences in the CDC reference sequence databases.
Disease Incidence Calculations
The prevalence of each pathogen in specimens was used to calculate pathogen-specific incidence rates of acute gastroenteritis among the study population. We used pooled data on self-reported health care utilization practices of persons with acute diarrheal disease obtained from Foodborne Diseases Active Surveillance Network (FoodNet) population surveys in 2000–2001, 2002–2003 (2), and 2006–2007 (CDC, unpub. data). These 3 population-based telephone surveys were conducted in selected sites located throughout the United States, including Georgia, by using a probability sample design.
The weighted proportion of survey respondents with AGE (defined as diarrhea [>3 loose stools in a 24-hour period] beginning within the past month) who sought medical care in person and the proportion of those respondents who submitted a specimen were calculated and stratified by age group (Table 1). Because viral diarrhea is generally of shorter duration than diarrhea of other etiologies (4), data from respondents with diarrhea lasting <3 days at the time of interview were used for estimates of viral etiologies, and data from respondents with diarrhea of any duration were used for estimates of bacterial, parasitic, and other etiologies. These data were used to generate age-group–specific rates of fecal specimen submission among all those in the community with diarrhea and among only those seeking medical care. Respondents with a chronic disease for which diarrhea is a major sign (e.g., celiac disease, Crohn disease, diverticulitis, irritable bowel syndrome, ulcerative colitis) were excluded from analysis.
To adjust for variations in health care utilization practices by age, we weighted age-group–specific FoodNet Population Survey data proportional to the age distribution of persons with specimens positive for each pathogen by using the following age groups: <5, 5–15, 16–25, 26–45, 46–65, and >65 years. These age groups were broadly selected for clinical relevance and consistency in health care utilization rates. Community and outpatient incidence of each pathogen was calculated on the basis of prevalence of that pathogen in sampled specimens (Pi), pathogen-specific fecal specimen submission rates among all respondents with AGE (ComSSi) and among those seeking medical care (OutSSi), the total number of specimens submitted to the KP laboratory during the study period (S = 1,825), and the total Kaiser membership in Georgia (i.e., study catchment population, N = 280,000) (Figure 1).
Thus, incidence was calculated on the basis of extrapolation of pathogen prevalence by using fecal specimen submission and medical care–seeking rates for community estimates and only fecal specimen submission rates for outpatient estimates. A simulation approach was used to generate 90% credible intervals (CIs). For each pathogen or group of pathogens, Pi, ComSSi, and OutSSi were randomly drawn assuming a β distribution for each, and the 2 incidence equations were recalculated. We report 5th and 95th centiles of 100,000 simulations. Incidence estimates weighted for monthly fluctuations in the number of specimens received by the KP laboratory were also evaluated, but because they did not differ appreciably with unadjusted estimates, the simpler unadjusted approach was used. Chi-square tests were used to evaluate trends among categorical variables. Analyses were performed by using SAS version 9.2 (SAS Institute, Cary, NC, USA), Stata version 11.0 (StataCorp LP, College Station, TX, USA), and Epi Info version 3.4.3 (CDC).
A total of 572 specimens were included in the study. Routine bacterial culture and viral PCR were performed on all specimens, and 375 (65.6%) were also tested for ova and parasites, 161 (28.1%) for C. difficile, and 28 (4.9%) for rotavirus by using EIA. Specimens not tested for ova and parasites or C. difficile were considered not positive for those pathogens. Adult patients 26–65 years of age provided 325 (56.8%) of the specimens tested and 316 (55.2%) were from female patients. Clinical diagnostic testing at the KP laboratory identified a pathogen in 42 (7.3%) specimens. Subsequent PCR testing at CDC identified >1 virus in 53 (9.3%) specimens, for a total of 88 (15.4%) specimens with a detected pathogen (includes 5 specimens previously positive for rotavirus by EIA and 2 specimens positive for bacteria and virus). Among these 88 specimens, viruses were detected in 53 (60.2%), bacteria in 30 (34.1%), and parasites in 7 (8.0%). Multiple pathogens were identified in only 4 specimens (Table 2). Norovirus was the most frequently identified pathogen overall, detected in 25 (4.4%) of all specimens and 28.4% of the 88 specimens with any pathogen identified. Detection of any pathogen in feces was most likely in children <5 years of age (32.1%) and decreased significantly with age to 3.6% for persons >65 years of age (p<0.001).
Most (62.4%) specimens were received for laboratory testing within 3 days of the patient’s medical visit. However, 90 (15.7%) were received >1 week later. Time lag did not differ by age group. Pathogens were detected most frequently among specimens received by the laboratory within 3 days of outpatient visit (18.2%) and were significantly less likely to be detected with increasing delay, decreasing to 6.7% among specimens received after >1 week (p<0.001). This time lag effect was more pronounced for viruses than bacteria and was likely caused by the decrease in viral shedding and test sensitivity after the acute phase of illness. For example, norovirus was identified in 5.0% of specimens received <7 days of outpatient visit, but in only 1.1% of those received >1 week after outpatient visit; overall detection of bacteria decreased from 5.6% to 3.3% during the same time frame. Most (81.4%) specimens were unformed (i.e., took on the shape of the collection cup), indicating that the patients were still symptomatic at the time of specimen collection. No differences in rates of pathogen detection were found between formed and unformed specimens.
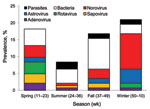
Overall rate of pathogen detection and the relative distribution of pathogens showed apparent seasonal variation, although insufficient sample size precluded identification of significant temporal trends. Overall, viruses predominated during winter and spring, and bacteria predominated during summer and fall (Figure 2). Relatively low prevalence of parasites was observed year round with no discernible seasonal pattern. Pronounced winter peaks in prevalence were observed for norovirus (10.5%) and astrovirus (4.2%). All rotavirus-positive specimens were received during winter and spring, during which prevalence was 1.4% and 3.5%, respectively. Similarly, peak prevalence of sapovirus (2.8%) and adenovirus (2.1%) was during spring.
Overall estimated community incidence of AGE of any etiology was 41,000 cases/100,000 person-years (90% CI 38,000–44,000 cases), of which 13,000 (32%, 90% CI 10,000–20,000 cases) were caused by identified agents (Table 3). Estimated incidence of AGE in outpatients was 5,400 cases/100,000 person-years (90% CI 4,400–6,700 cases), of which 1,600 (30%, 90% CI 1,300–2,400 cases) could be attributed to a specific pathogen. Norovirus was the leading identifiable cause of illness in community and outpatient settings and was associated with 16% of all community AGE (6,500 cases/100,000 person-years, 90% CI 3,700–12,000 cases) and 12% of all outpatient AGE (640/100,000 person-years, 90% CI 360–1,200 cases).
In comparison with estimated community incidence of AGE caused by bacteria (1,700 cases/100,000 person-years, 90% CI 1,100–2,300) and parasites (420 cases/100,000 person-years, 90% CI 200–790), the community incidence of norovirus was ≈4-fold and >15-fold greater, respectively. Likewise, the incidence of norovirus infection prompting medical care was ≈3-fold greater than that of bacterial pathogens (240 cases/100,000 person-years, 90% CI 160–320 cases) and >10-fold greater than that of parasites (60 cases/100,000 person-years, 90% CI 29–110 cases). Astrovirus was the second leading identifiable pathogen associated with AGE in community and outpatient settings; estimated incidences were 1,800 cases (90% CI 880–3,400 cases) and 270 (90% CI 130–590 cases) per 100,000 person-years, respectively.
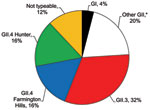
Sequence analysis of the 25 norovirus-positive samples identified 24 (96%) as GII strains and 1 (4%) as a GI strain (Figure 3). Of all strains, approximately one third could be typed as GII.4, specifically the Farmington Hills and Hunter subclusters. GII.3 viruses were equally prevalent in this population. Other norovirus genotypes identified included GI3b, GII.2, GII.14, and GII.17.
This study provides US laboratory–based incidence rates for norovirus disease determined by using direct outpatient surveillance of routinely collected fecal specimens. Despite examination of only fecal specimens submitted for bacterial culture, our data demonstrate that viruses, and noroviruses specifically, were the leading cause of AGE among persons of all ages seeking medical care. We estimate that 24% of persons seeking medical care for AGE do so because of viral infection, including 12% because of norovirus infection. Overall, viruses accounted for 27% of AGE episodes in the community and noroviruses accounted for 16%.
In contrast to bacterial and parasitic pathogens, viral AGE pathogens were primarily detected during winter and spring months, which is consistent with previous descriptions of norovirus and rotavirus seasonality in temperate climates (16). Norovirus strains identified in this study were similar to those identified in studies of sporadic AGE (17) and those implicated in norovirus outbreaks confirmed by CDC during the same period (18). The predominance of GII.4 strains in epidemic and sporadic norovirus disease demonstrates the need for including this genotype in vaccine development efforts.
Our findings are consistent with those of studies conducted in other industrialized countries, which demonstrated the relative role of viral pathogens in causing AGE (19–23). A recent systematic literature review that included 13 etiologic studies of community and outpatient cases of sporadic diarrhea, most of which focused on children, determined that norovirus was responsible for 12% (range 5%–36%) of AGE cases (17). In a community-based study in Germany, the incidence of AGE requiring medical consultation attributable to norovirus was 626 cases/100,000 person-years (19). In England, the incidence of general practice consultations for norovirus-associated infectious intestinal disease was 540 cases/100,000 person-years (24). The estimated incidence of norovirus among outpatients in our study (640 cases/100,000 person-years) supports the findings of the studies in England and Germany. A similar study in the Netherlands reported a considerably lower incidence of gastroenteritis in general practices (797 cases/100,000 person-years), in which 5% (40 cases/100,000 person-years) were caused by norovirus (20). However, standardized gastroenteritis incidence reported in the Dutch community of 28,300 cases/100,000 person-years is more similar to that estimated in our study (41,000 cases/100,000 person-years) (25). Dutch guidelines for general practitioners, which recommend that cases of uncomplicated AGE be handled by telephone consultation (http://nhg.artsennet.nl), may explain the apparent difference in health care utilization practices.
Prior US estimates of the incidence of norovirus have been based on a bottom-up approach of using overall incidence of AGE from population surveys and estimating the proportion attributable to norovirus on the basis of data from other countries, given the lack of laboratory-based data available at the time (3). This approach yielded an estimate of ≈21 million cases of norovirus illness annually, corresponding to a community incidence similar to that estimated in our study (6,974 cases/100,000 person-years vs. 6,500 cases/100,000 person-years). Similarly generated bottom-up estimates for astrovirus, rotavirus, and sapovirus (each 1,024 cases/100,000 person-years) were likewise within 90% CIs of our community incidence estimates (3).
Although markedly different approaches were used, each subject to considerable uncertainty, concordance of the respective incidence estimates generated is reassuring. However, given the inclusion of only specimens submitted for routine bacterial diagnostics, our study likely underestimated the true incidence of norovirus disease in the community. Furthermore, use of health care utilization multipliers based primarily on diarrhea may underestimate the incidence of pathogens that can cause other AGE signs (e.g., vomiting) in the absence of diarrhea, as has been reported for norovirus (26). Previous studies have demonstrated that seeking medical care is influenced by disease severity and social factors (27,28), which leads to a smaller proportion of viral AGE patients in the community who seek medical care, compared with bacterial and parasitic AGE (22). Identifying laboratory-confirmed cases of norovirus infection and extrapolating community incidence by using pathogen-specific health care utilization multipliers in a top-down approach may be preferable when such data are available and would likely yield more reliable estimates.
The primary limitation of this study results from use of routinely collected specimens, as opposed to systematic, active patient recruitment. The study specimens may have therefore been obtained from patients with noninfectious or chronic diseases, which may have contributed to the low detection rate for pathogens in this study. Delays in collection of specimens may have also contributed to the low rates of pathogen detection. Routinely collected samples also overrepresent some age groups, as demonstrated by differential fecal submission rates (Table 1), which may not reflect age groups at greatest risk for infection by specific pathogens or risk for the overall population.
We have adjusted for this sampling artifact by development of pathogen-specific, age-adjusted health care utilization multipliers. These multipliers resulted in the observed increase from pathogen prevalence in specimens (e.g., 4.4% for norovirus) to pathogen prevalence among estimated community AGE cases (e.g., 16% for norovirus). In addition to age, pathogen-specific health care utilization multipliers also accounted for duration of illness and yielded different pathogen distributions in community and outpatient settings. These results are consistent with those of previous studies (20,22,25). In contrast to a previous study that estimated US incidence of AGE (3), our study did not adjust for differential health care utilization rates based on illness severity (i.e., bloody vs. nonbloody diarrhea) to maintain adequate power to enable the adjustment for age group. Nonetheless, there was insufficient sample size within each age group to generate age group–specific disease incidence estimates.
Attributing causality of AGE to norovirus infection is complicated by the fact that asymptomatic infections may occur; healthy persons used in previous studies as controls have shown background rates of infection of 1%–16% (17). However, low prevalence (1.1%) of norovirus in specimens received >1 week after outpatient visit and low number of mixed infections identified in our study suggest that norovirus was likely the etiologic agent when detected. Incidence estimates were not adjusted for test sensitivity because the molecular methods used for viral diagnostics generally show extremely high sensitivities if appropriate and timely specimens are tested. Prevalence and incidence of parasites and C. difficile may be underestimated given that not all specimens were tested for these pathogens and indications for clinicians requesting specific tests are unknown and likely varied. Finally, given that the study population included only Georgia residents and health maintenance organization members, which tends toward younger age and higher socioeconomic status, the results may not be generalizable to the overall US population. Restriction of FoodNet data to only respondents in Georgia who had health insurance was evaluated for comparison but did not differ. Therefore, all respondents were ultimately included to enable adequate power for age stratification.
Development of sensitive clinical assays for identification of viral agents of AGE, such as norovirus, and more widespread use of such assays may help close the diagnostic gap on sporadic AGE cases and guide more appropriate case management. The demonstrated predominance of viruses among medically attended AGE cases should help prevent the unnecessary use of antimicrobial drugs and spur development of novel interventions specific for the unique transmission pathways of viruses. Compared with bacterial AGE etiologies, many of which result from foodborne transmission from infected animal sources, viral AGE pathogens originate in human reservoirs and usually involve direct or indirect person-to-person spread. Although occasionally implicated in outbreaks (29,30), the role of foods contaminated during processing in the overall norovirus disease incidence remains largely unknown. Further assessment of the incidence of enteric viruses, including hospital-based studies of risk factors for severe disease and attribution to specific transmission pathways, are needed to improve control measures and assess future potential of vaccines.
Dr Hall is an epidemiologist with the Viral Gastroenteritis Team, Division of Viral Diseases, National Center for Immunization and Respiratory Diseases, CDC. His research interests include all aspects of the epidemiology of noroviruses and other agents of viral gastroenteritis.
Acknowledgments
We thank Elaine Scallan and Mike Hoekstra for critically reviewing the manuscript.
This study was supported in part by the Atlanta Research and Education Foundation and by a Henry Aldrich Student Research Grant of the Southeastern Branch of the American Society for Microbiology.
References
- Scallan E, Griffin PM, Angulo FJ, Tauxe RV, Hoekstra RM. Foodborne illness acquired in the United States—unspecified agents. Emerg Infect Dis. 2011;17:16–22.PubMedGoogle Scholar
- Jones TF, McMillian MB, Scallan E, Frenzen PD, Cronquist AB, Thomas S, A population-based estimate of the substantial burden of diarrhoeal disease in the United States; FoodNet, 1996–2003. Epidemiol Infect. 2007;135:293–301. DOIPubMedGoogle Scholar
- Scallan E, Hoekstra RM, Angulo FJ, Tauxe RV, Widdowson MA, Roy SL, Foodborne illness acquired in the United States—major pathogens. Emerg Infect Dis. 2011;17:7–15.PubMedGoogle Scholar
- Widdowson MA, Sulka A, Bulens SN, Beard RS, Chaves SS, Hammond R, Norovirus and foodborne disease, United States, 1991–2000. Emerg Infect Dis. 2005;11:95–102.PubMedGoogle Scholar
- Harris JP, Edmunds WJ, Pebody R, Brown DW, Lopman BA. Deaths from norovirus among the elderly, England and Wales. Emerg Infect Dis. 2008;14:1546–52. DOIPubMedGoogle Scholar
- Rondy M, Koopmans M, Rotsaert C, Van Loon T, Beljaars B, Van Dijk G, Norovirus disease associated with excess mortality and use of statins: a retrospective cohort study of an outbreak following a pilgrimage to Lourdes. Epidemiol Infect. 2011;139:453–63. DOIPubMedGoogle Scholar
- Centers for Disease Control and Prevention. Norovirus activity—United States, 2006–2007. MMWR Morb Mortal Wkly Rep. 2007;56:842–6.PubMedGoogle Scholar
- Trujillo AA, McCaustland KA, Zheng DP, Hadley LA, Vaughn G, Adams SM, Use of TaqMan real-time reverse transcription-PCR for rapid detection, quantification, and typing of norovirus. J Clin Microbiol. 2006;44:1405–12. DOIPubMedGoogle Scholar
- Okada M, Yamashita Y, Oseto M, Shinozaki K. The detection of human sapoviruses with universal and genogroup-specific primers. Arch Virol. 2006;151:2503–9. DOIPubMedGoogle Scholar
- Jothikumar N, Cromeans TL, Hill VR, Lu X, Sobsey MD, Erdman DD. Quantitative real-time PCR assays for detection of human adenoviruses and identification of serotypes 40 and 41. Appl Environ Microbiol. 2005;71:3131–6. DOIPubMedGoogle Scholar
- Belliot G, Laveran H, Monroe SS. Detection and genetic differentiation of human astroviruses: phylogenetic grouping varies by coding region. Arch Virol. 1997;142:1323–34. DOIPubMedGoogle Scholar
- Grinde B, Jonassen TO, Ushijima H. Sensitive detection of group A rotaviruses by immunomagnetic separation and reverse transcription–polymerase chain reaction. J Virol Methods. 1995;55:327–38. DOIPubMedGoogle Scholar
- Kojima S, Kageyama T, Fukushi S, Hoshino FB, Shinohara M, Uchida K, Genogroup-specific PCR primers for detection of Norwalk-like viruses. J Virol Methods. 2002;100:107–14. DOIPubMedGoogle Scholar
- Xu W, McDonough MC, Erdman DD. Species-specific identification of human adenoviruses by a multiplex PCR assay. J Clin Microbiol. 2000;38:4114–20.PubMedGoogle Scholar
- Yan H, Yagyu F, Okitsu S, Nishio O, Ushijima H. Detection of norovirus (GI, GII), sapovirus and astrovirus in fecal samples using reverse transcription single-round multiplex PCR. J Virol Methods. 2003;114:37–44. DOIPubMedGoogle Scholar
- Mounts AW, Ando T, Koopmans M, Bresee JS, Noel J, Glass RI. Cold weather seasonality of gastroenteritis associated with Norwalk-like viruses. J Infect Dis. 2000;181(Suppl 2):S284–7. DOIPubMedGoogle Scholar
- Patel MM, Widdowson MA, Glass RI, Akazawa K, Vinje J, Parashar UD. Systematic literature review of role of noroviruses in sporadic gastroenteritis. Emerg Infect Dis. 2008;14:1224–31. DOIPubMedGoogle Scholar
- Zheng DP, Widdowson MA, Glass RI, Vinjé J. Molecular epidemiology of genogroup II–genotype 4 noroviruses in the United States between 1994 and 2006. J Clin Microbiol. 2010;48:168–77. DOIPubMedGoogle Scholar
- Karsten C, Baumgarte S, Friedrich AW, von Eiff C, Becker K, Wosniok W, Incidence and risk factors for community-acquired acute gastroenteritis in north-west Germany in 2004. Eur J Clin Microbiol Infect Dis. 2009;28:935–43. DOIPubMedGoogle Scholar
- de Wit MA, Koopmans MP, Kortbeek LM, van Leeuwen NJ, Bartelds AI, van Duynhoven YT. Gastroenteritis in sentinel general practices, the Netherlands. Emerg Infect Dis. 2001;7:82–91. DOIPubMedGoogle Scholar
- Jansen A, Stark K, Kunkel J, Schreier E, Ignatius R, Liesenfeld O, Aetiology of community-acquired, acute gastroenteritis in hospitalised adults: a prospective cohort study. BMC Infect Dis. 2008;8:143. DOIPubMedGoogle Scholar
- Wheeler JG, Sethi D, Cowden JM, Wall PG, Rodrigues LC, Tompkins DS, Study of infectious intestinal disease in England: rates in the community, presenting to general practice, and reported to national surveillance. The Infectious Intestinal Disease Study Executive. BMJ. 1999;318:1046–50.PubMedGoogle Scholar
- Huhulescu S, Kiss R, Brettlecker M, Cerney RJ, Hess C, Wewalka G, Etiology of acute gastroenteritis in three sentinel general practices, Austria 2007. Infection. 2009;37:103–8. DOIPubMedGoogle Scholar
- Phillips G, Tam CC, Conti S, Rodrigues LD, Brown D, Iturriza-Gomara M, Community incidence of norovirus-associated infectious intestinal disease in England: improved estimates using viral load for norovirus diagnosis. Am J Epidemiol. 2010;171:1014–22. DOIPubMedGoogle Scholar
- de Wit MA, Koopmans MP, Kortbeek LM, Wannet WJ, Vinjé J, van Leusden F, Sensor, a population-based cohort study on gastroenteritis in the Netherlands: incidence and etiology. Am J Epidemiol. 2001;154:666–74. DOIPubMedGoogle Scholar
- Glass RI, Parashar UD, Estes MK. Norovirus gastroenteritis. N Engl J Med. 2009;361:1776–85. DOIPubMedGoogle Scholar
- Scallan E, Jones TF, Cronquist A, Thomas S, Frenzen P, Hoefer D, Factors associated with seeking medical care and submitting a stool sample in estimating the burden of foodborne illness. Foodborne Pathog Dis. 2006;3:432–8. DOIPubMedGoogle Scholar
- Tam CC, Rodrigues LC, O’Brien SJ. The study of infectious intestinal disease in England: what risk factors for presentation to general practice tell us about potential for selection bias in case–control studies of reported cases of diarrhoea. Int J Epidemiol. 2003;32:99–105. DOIPubMedGoogle Scholar
- Malek M, Barzilay E, Kramer A, Camp B, Jaykus LA, Escudero-Abarca B, Outbreak of norovirus infection among river rafters associated with packaged delicatessen meat, Grand Canyon, 2005. Clin Infect Dis. 2009;48:31–7. DOIPubMedGoogle Scholar
- Maunula L, Roivainen M, Keränen M, Mäkela S, Söderberg K, Summa M, Detection of human norovirus from frozen raspberries in a cluster of gastroenteritis outbreaks. Euro Surveill. 2009;14: pii: 19435.
Figures
Tables
Cite This ArticleTable of Contents – Volume 17, Number 8—August 2011
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Aron J. Hall, Centers for Disease Control and Prevention, 1600 Clifton Rd NE, Mailstop A34, Atlanta, GA 30333, USA
Top