Volume 18, Number 8—August 2012
Dispatch
Vertical Transmission of Babesia microti, United States
Cite This Article
Citation for Media
Abstract
Babesiosis is usually acquired from a tick bite or through a blood transfusion. We report a case of babesiosis in an infant for whom vertical transmission was suggested by evidence of Babesia spp. antibodies in the heel-stick blood sample and confirmed by detection of Babesia spp. DNA in placenta tissue.
Babesiosis is an emerging infection in the United States, principally caused by Babesia microti (1). The most common route of infection is the bite of an Ixodes scapularis tick; transmission can also occur by transfusion of infected blood products, and vertical transmission in animals has been documented (2,3) and is a potential route of transmission for humans. We report a case of babesiosis in an infant for whom vertical transmission was suggested by Babesia spp. antibodies in a heel spot blood sample and confirmed by detection of Babesia DNA in placenta tissue.
A 6-week-old girl from Yorktown Heights, New York, was admitted to the hospital on September 16, 2002, with a 2-day history of fever, irritability, and decreased oral intake. The mother was asymptomatic during and after her pregnancy. The infant was delivered vaginally and full term at 3,430 g without complications. The infant’s mother had visited parks in Westchester and Dutchess Counties in New York during the pregnancy but was unaware of any tick bites. The infant had no known tick exposure, and neither mother nor infant had a history of blood transfusion.
During examination, the infant was alert but irritable and pale. Axillary temperature was initially 36.8°C but increased to 38.1°C on the same day. Her conjunctivae were icteric, she had a palpable spleen tip, and her liver was palpable 3 cm below the costal margin. Initial laboratory findings included hemoglobin 7.1 g/dL, platelet count 100 × 103/μL, and leukocyte count 19.7 × 103 cells/μL with a differential of 4% segmented neutrophils, 80% lymphocytes, and 16% monocytes. Reticulocyte count was 5.5%. Total bilirubin concentration was 2 mg/dL with a direct fraction of 0.4 mg/dL; aspartate aminotransferase level was 66 U/L, alanine aminotransferase level was 50 U/L, and alkaline phosphatase level was 339 U/L. Cultures of blood, urine, and cerebrospinal fluid samples yielded negative results. Lyme disease serologic test result was negative.
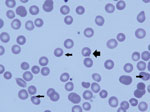
Routine examination of a peripheral blood smear showed B. microti in 4% of erythrocytes (Figure); a blood sample from the infant was positive by PCR for B. microti DNA. Total B. microti antibody titer was >256 by indirect immunofluorescence assay, with a polyvalent secondary antibody (anti-IgG+IgA+IgM) (4) that was presumed to be principally IgG because test results for IgM were negative (Technical Appendix). The heel-stick blood sample obtained on the infant’s third day of life as part of newborn screening was tested and found to be negative for B. microti by PCR (5) and for IgM but total antibody positive (>128) (Technical Appendix).
Examination of the placenta showed focal basal decidual inflammation, mild chorangiosis, and villus dysmaturity. Babesia spp. piroplasms were not detected in maternal or fetal blood by histologic examination of hematoxylin and eosin–stained sections of formalin-fixed, paraffin-embedded tissue of the placenta disk, amnion/chorion, and umbilical cord. Babesia DNA was detected by real-time PCR testing of paraffin-embedded placenta tissue (Technical Appendix) (6). Cycle threshold values were relatively high (37.1–38.2), indicating that the amount of parasite DNA in the sample was close to the limit of detection; results were reproducible on duplicate testing of DNA samples extracted from separate paraffin blocks. The real-time PCR product was of the correct size, and the melting curve demonstrated melting temperatures within 1°C from the placenta, the positive control, and a positive sample from an unrelated patient , confirming that the correct product was amplified. At time of the illness in the infant, the mother was negative for Babesia spp. according to PCR and smear but positive for total antibodies (>256).
The infant was treated with a 9-day course of azithromycin plus atovaquone. A blood transfusion was administered when her hemoglobin concentration fell to 5.2 g/dL. The infant became afebrile by 72 hours and was discharged after a 5-day hospitalization. Repeat blood smears revealed a parasite load of 0.3% at discharge. On final evaluation at 22 months of age, physical examination revealed no abnormalities; hemoglobin level was 11.7 g/dL, Babesia PCR was negative, and total Babesia antibody level was positive at 128.
Congenital babesiosis has been rarely reported (Table) (7–10). This case provided convincing evidence for congenital babesiosis because of prepartum infection involving the placenta in the mother. On the basis of experience with congenital malaria, we assume that Babesia spp. parasites cross the placenta during pregnancy or at the time of delivery (11,12). In congenital malaria, increasing evidence suggests that the malaria parasites are most often acquired antenatally by transplacental transmission of infected erythrocytes (12).
Reported cases of congenital babesiosis share many similarities, including asymptomatic maternal infection and development of fever, hemolytic anemia, and thrombocytopenia in the infant detected between 19 and 41 days after birth. All of the infants responded to antimicrobial drug therapy; 3 were treated with azithromycin plus atovaquone (9,10), the preferred treatment regimen for mild babesiosis (1). All infants required a blood transfusion because of severe anemia. The clinical signs and symptoms for these cases of congenital babesiosis are similar to those of congenital malaria in non–disease endemic areas (11,13).
We found Babesia spp. antibodies on day 3 of life by analyzing the patient’s heel-stick blood sample, which likely represented maternal transfer of IgG. Passive transfer of maternal antibodies is regarded as a protective factor against congenital malaria, and some newborns with malaria who are parasitemic at birth spontaneously clear the infection without ever becoming ill (11,14). The temporary presence of maternal IgG in infants has been suggested as an explanation for the typical 3–6 week incubation period of congenital malaria in non–disease endemic areas (14).
The real-time PCR used to find B. microti DNA in placenta tissue is ≈20× more sensitive than microscopic examination of Giemsa-stained blood smears (6). Assuming a blood sample with a parasitemia equivalent to that detected in the placental tissue, a blood smear would contain <10 infected cells per slide. Given the low level of Babesia DNA in the placenta tissue, it is not surprising that histologic examination did not reveal piroplasms. Nonetheless, limited evidence of placental abnormalities suggests a pathologic process.
In summary, babesiosis is an emerging infectious disease (15) that can rarely cause congenital infection. This diagnosis should be considered in the differential diagnosis of fever and hemolytic anemia in infants from disease-endemic areas.
Dr Joseph is an assistant professor of medicine in the Division of Infectious Diseases at New York Medical College. Her research interests are tick-borne illnesses, particularly babesiosis.
Acknowledgment
The authors thank Steven Smith, Jennifer Calder, Lisa Giarratano, Lenise Banwarie, Ewa Bajor-Dattilo, and Karen Kulas for their assistance.
References
- Vannier E, Gewurz BE, Krause PJ. Human babesiosis. Infect Dis Clin North Am. 2008;22:469–88. DOIPubMedGoogle Scholar
- de Vos AJ, Imes GD, Cullen JSC. Cerebral babesiosis in a new-born calf. Onderstepoort J Vet Res. 1976;43:75–8.PubMedGoogle Scholar
- Fukumoto S, Suzuki H, Igarashi I, Xuan X. Fatal experimental transplacental Babesia gibsoni infections in dogs. Int J Parasitol. 2005;35:1031–5. DOIPubMedGoogle Scholar
- Chisholm ES, Ruebush TK II, Sulzer AJ, Healy GR. Babesia microti infection in man: evaluation of an indirect immunofluorescent antibody test. Am J Trop Med Hyg. 1978;27:14–9.PubMedGoogle Scholar
- Persing DH, Mathiesen D, Marshall WF, Telford SR, Spielman A, Thomfod JW, Detection of Babesia microti by polymerase chain reaction. J Clin Microbiol. 1992;30:2097–103.PubMedGoogle Scholar
- Teal AE, Habura A, Ennis J, Keithly J, Madison-Antenucci S. A new real-time PCR assay for improved detection of the parasite Babesia microti. J Clin Microbiol. 2012;50:903–8. DOIPubMedGoogle Scholar
- Esernio-Jenssen D, Scimeca PG, Benach JL, Tenenbaum MJ. Transplacental/perinatal babesiosis. J Pediatr. 1987;110:570–2. DOIPubMedGoogle Scholar
- New DL, Quinn J, Qureshi MZ, Sigler S. Vertically transmitted babesiosis. J Pediatr. 1997;131:163–4. DOIPubMedGoogle Scholar
- Sethi S, Alcid D, Kesarwala H, Tolan RW Jr. Probable congenital babesiosis in infant, New Jersey, USA. Emerg Infect Dis. 2009;15:788–91. DOIPubMedGoogle Scholar
- Aderinboye O, Syed S. Congenital babesiosis in a four-week old female infant. Pediatr Infect Dis J. 2010;29:188. DOIPubMedGoogle Scholar
- Vottier G, Arsac M, Farnoux C, Mariani-Kurddjian P, Baud O, Aujard Y. Congenital malaria in neonates: two case reports and review of the literature. Acta Paediatr. 2008;97:505–8. DOIPubMedGoogle Scholar
- Malhotra I, Mungai P, Muchiri E, Kwiek JJ, Meshnick SR, King CL. Umbilical cord-blood infections with Plasmodium falciparum malaria are acquired antenatally in Kenya. J Infect Dis. 2006;194:176–83. DOIPubMedGoogle Scholar
- Lesko CR, Arguin PM, Newman RD. Congenital malaria in the United States. A review of cases from 1966 to 2005. Arch Pediatr Adolesc Med. 2007;161:1062–7. DOIPubMedGoogle Scholar
- Hagmann S, Khanna K, Niazi M, Purswani M, Robins EB. Congenital malaria, an important differential diagnosis to consider when evaluating febrile infants of immigrant mothers. Pediatr Emerg Care. 2007;23:326–9. DOIPubMedGoogle Scholar
- Joseph JT, Roy SS, Shams N, Visintainer P, Nadelman RB, Hosur S, Babesiosis in Lower Hudson Valley, New York, USA. Emerg Infect Dis. 2011;17:843–7.PubMedGoogle Scholar
Figure
Table
Cite This Article1Current affiliation: New York University School of Medicine, New York, New York, USA.
Table of Contents – Volume 18, Number 8—August 2012
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Julie T. Joseph, New York Medical College, Division of Infectious Diseases, Munger Pavilion Room 245, Valhalla, NY 10595, USA
Top