Volume 19, Number 7—July 2013
Letter
Human Infection with Marten Tapeworm
Cite This Article
Citation for Media
To the Editor: Cysticercosis-like human infections with the tapeworm Taenia crassiceps, which infects foxes as terminal hosts, have been reported (1,2). We report a case of a cysticercosis-like eye infection caused by the tapeworm T. martis (marten tapeworm) in a woman.
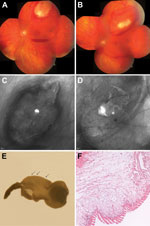
The patient was a 43-year-old German woman who sought care during July 2010, after 4 days of perceiving flashing lights in her visual field and a paracentral scotoma in her left eye. Visual acuity in both eyes was 20/20. Examination of the left fundus revealed a mobile subretinal tumor at the temporal upper retinal branch vessel with adjacent intraretinal and subhyaloid bleeding (Figure, panels A–C; Video). The subretinal tumor resembled a cestode larva.
Direct Video Link:
The patient reported no other symptoms at that time. Laboratory evaluation found no eosinophilia or elevation of total IgE. Serologic testing results were negative for antibodies against the following parasites: Taenia solium, Echinococcus multilocularis, E. granulosus, Dirofilaria immitis, Strongyloides spp., and Toxocara canis. Fecal testing results were negative for worm eggs. Images from ultrasonography of the liver and magnetic resonance imaging of the head were unremarkable. The patient’s travel history included—in addition to southern European countries—trips to Nepal and Thailand 15 years previously.
At the time of examination, the patient lived in a small village near Freiburg (im Breisgau) in southwestern Germany. She grew vegetables in the family garden, which was next to a forest. Her 3 children and husband did not report any health problems. For the past 6 years, the family had owned a dog, which received antiparasitic medications on a regular basis; recent checks for intestinal parasitic infection found no ova.
The suspected cause of the woman’s illness was cysticercosis caused by the larva of T. solium; systemic antiparasitic therapy was started (albendazole 400 mg 2×/d, dexamethasone 20 mg/d). The size of the larva diminished (Figure, panel D; Video), but the patient remained symptomatic. Therefore, after 8 days of therapy, the cyst was removed by retinotomy. A few days later, peripheral retinal detachment occurred and was treated by a second vitrectomy and intravitreal gas injection. Because of the repeated gas tamponade, a gas cataract developed, which necessitated cataract surgery. At the end of March 2011, the patient’s visual acuity had returned to 20/20 in both eyes.
The removed cyst showed the characteristic macroscopic and histologic features of a cysticercus bladder wall (Figure, panels E, F). To determine the exact species by using molecular methods, we isolated DNA from the cyst, conducted different PCRs selective for mitochondrial genes, determined the corresponding sequences, and used a BLAST search (3) to compare these sequences with publically available sequences. Sequences of the following mitochondrial genes were determined by using the given primers and later submitted to GenBank: small ribosomal subunit (primers 12S Taenia FF 5′-CACAGTGCCAGCATCYGCGGT-3′ and 12S Taenia RR 5′-GAGGGTGACGGGCGGTGTGTAC-3′, PCR product of 426 bp, GenBank accession no. JX415820); NADH dehydrogenase subunit 1 (primers: NAD1-FF 5′-ATTGGKTTATTTCAGAGTTTTTCTGATTTA-3′ and NAD1-RR 5′-CTCMCCATAATCAAATGGACTACG-3′, 394 bp, GenBank accession no. JX415819); and the cytochrome-c oxidase subunit 1 (determined by using previously published primers [4,5]; 376 bp, GenBank accession no. JX415821). All sequences showed highest identity with T. martis (99%–100%) but substantially lower identity with T. crassiceps (91%–97%) and T. solium (87%–89%) tapeworms.
Thus, molecular methods unequivocally identified the larva as that of a T. martis tapeworm. T. martis tapeworms (cestodes) live and produce eggs in the intestines of definitive hosts, weasels (family Mustelidae), which also includes pine martens, stone martens, polecats, badgers, wolverines, and stoats (6). The intermediate hosts are prey animals of the definitive hosts, such as arvicoline (voles and muskrats) or murid rodents. When intermediate hosts ingest eggs, cysticerci develop in the pleural and peritoneal cavities. T. martis tapeworms probably occur worldwide wherever suitable definitive and intermediate hosts are present (6,7). A study in southwest Germany reported that 36% of stone martens were infected with T. martis tapeworms (6).
Although nearly all patients who had cysticercosis-like infections caused by T. crassiceps tapeworms were immunosuppressed (1,2), we found no signs of immunosuppression in the patient reported here. The only identified risk factor for this patient was consumption of homegrown vegetables, which could have been contaminated by marten feces.
The clinical and histologic appearance of the organism in this patient suggested cysticercosis caused by a T. solium tapeworm. However, the specific diagnosis of T. martis tapeworm infection was possible only by use of molecular methods. Thus, human infections with T. martis and other animal tapeworms might occur at times but might be misdiagnosed as T. solium cysticercosis. For therapy, the rules and considerations are probably the same as those for T. solium cysticercosis, as described (8,9). Concerning antiparasitic therapy, one must be aware of possible complications caused by intraocular immunologic reactions. As demonstrated by the case reported here, surgical removal of a subretinal larva is connected with the risk for retinal detachment and cataract formation. The identification of the responsible tapeworm is useful for epidemiologic reasons, for determining the source of infection. We therefore suggest using molecular methods to determine the exact species of parasites removed from human tissue.
Acknowledgment
We gratefully acknowledge Heidrun von Thien for her technical assistance. We thank the patient for her collaboration and her approval to publish this case report, and we thank the board of directors of the Bernhard Nocht Institute for Tropical Medicine, Rolf Horstmann, Bernhard Fleischer, and Egbert Tannich for their support.
References
- Goesseringer N, Lindenblatt N, Mihic-Probst D, Grimm F, Giovanoli P. Taenia crassiceps upper limb fasciitis in a patient with untreated acquired immunodeficiency syndrome and chronic hepatitis C infection—the role of surgical debridement. J Plast Reconstr Aesthet Surg. 2011;64:e174–6 . DOIPubMedGoogle Scholar
- Heldwein K, Biedermann HG, Hamperl WD, Bretzel G, Loscher T, Laregina D, Subcutaneous Taenia crassiceps infection in a patient with non-Hodgkin's lymphoma. Am J Trop Med Hyg. 2006;75:108–11 .PubMedGoogle Scholar
- Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res. 1997;25:3389–402. DOIPubMedGoogle Scholar
- Bowles J, Blair D, McManus DP. Genetic variants within the genus Echinococcus identified by mitochondrial DNA sequencing. Mol Biochem Parasitol. 1992;54:165–73 . DOIPubMedGoogle Scholar
- Bowles J, McManus DP. NADH dehydrogenase 1 gene sequences compared for species and strains of the genus Echinococcus. Int J Parasitol. 1993;23:969–72 . DOIPubMedGoogle Scholar
- Loos-Frank B, Zeyhle E. The intestinal helminths of the red fox and some other carnivores in southwest Germany. Z Parasitenkd. 1982;67:99–113 . DOIPubMedGoogle Scholar
- Mathy A, Hanosset R, Adant S, Losson B. The carriage of larval Echinococcus multilocularis and other cestodes by the musk rat (Ondatra zibethicus) along the Ourthe River and its tributaries (Belgium). J Wildl Dis. 2009;45:279–87 .PubMedGoogle Scholar
- Palomares F, Palencia G, Ambrosio JR, Ortiz A, Jung-Cook H. Evaluation of the efficacy of albendazole sulphoxide and praziquantel in combination on Taenia crassiceps cysts: in vitro studies. J Antimicrob Chemother. 2006;57:482–8 . DOIPubMedGoogle Scholar
- Nash TE, Garcia HH. Diagnosis and treatment of neurocysticercosis. Nature reviews. Neurology. 2011;7:584–94 .PubMedGoogle Scholar
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 19, Number 7—July 2013
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Sven Poppert, Bernhard-Nocht-Institut für Tropenmedizin Bernhard-Nocht-Strasse 74, 20359 Hamburg, Germany
Top