Volume 16, Number 9—September 2010
Dispatch
Novel Hepatitis E Virus Genotype in Norway Rats, Germany
Cite This Article
Citation for Media
Abstract
Human hepatitis E virus infections may be caused by zoonotic transmission of virus genotypes 3 and 4. To determine whether rodents are a reservoir, we analyzed the complete nucleotide sequence of a hepatitis E–like virus from 2 Norway rats in Germany. The sequence suggests a separate genotype for this hepatotropic virus.
Hepatitis E virus (HEV) is a nonenveloped virus, diameter 30–34 nm, that belongs to the genus Hepevirus. Its single-stranded, positive-polarity RNA genome of 6.6–7.3 kb harbors 3 major open reading frames (ORFs) flanked by a capped 5′ end and a poly A at the 3′ end. ORF1 at the 5′ end of the genome codes for several nonstructural proteins, ORF2 encodes the immunodominant capsid protein, and the partially overlapping ORF3 codes for a cytoskeleton-associated phosphoprotein with multiple functions (1).
Hepatitis E, an acute self-limiting disease, occurs worldwide; large outbreaks have occurred in developing countries, as was recently reported from Uganda (2). Initially, hepatitis E was believed to be endemic only to developing countries in Asia, Africa, and Central America, but recent studies have demonstrated autochthonous infections in industrialized countries (Europe, Japan) (3). In contrast to the fecal–oral transmission of HEV that occurs in developing countries, it is suspected that these human infections result from zoonotic transmission of HEV genotypes 3 and 4; domestic pigs, wild boars, and deer represent major reservoir hosts (1,4). However, rodents, especially commensal rodents, may represent an additional HEV reservoir and may play a role in the epidemiology of hepatitis E. HEV-reactive antibodies have been detected in several rat species (Rattus norvegicus, R. rattus, R. exulans) but also in some noncommensal wild rodent species (5–8). By using broad-spectrum, nested, reverse transcription–PCR (RT-PCR), we recently detected HEV-like sequences in fecal samples of Norway rats (R. norvegicus) trapped as part of the Rodent-borne Pathogens network (which coordinates activities with regard to rodent trapping during outbreaks) (9,10). These sequence fragments had high nucleotide sequence divergence to genotypes 1–4 and to avian HEV strains.
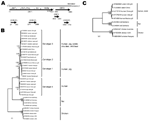
During July 8–16, 2009, a total of 6 Norway rats, 3 male and 3 female, 65–432 g, were trapped in manholes of the sewer system of Hamburg, northern Germany, at the same locations where ≈12 months before HEV RNA had been detected in rat feces (10). Standardized necropsy (9) found no morphologic abnormalities. Initial serologic screening with a commercial genotype 1–based ELISA (Axiom, Bürstadt, Germany) detected no reactive antibodies in transudates of any of the 6 rats. Liver RNA from 1 female (no. 68, 311 g) and 1 male (no. 63, 313 g) rat yielded an amplification product of the expected size (331–334 nt) and a sequence identity of 83.8%–94.6 % with the HEV sequences recently obtained from rat feces (data not shown). Using a strategy according to Schielke et al. (4), we determined the entire rat HEV genome sequences from each sample to be 6,945 nt and 6,948 nt; the sequences differed by an insertion–deletion polymorphism in the 3′ noncoding region. The sequence identity between each complete sequence was 95.3% and reached 55.1%–55.9% to HEV genotypes 1–4 and 49.3%–50.2% to avian HEV strains (Table). Using prediction software, we identified the major ORFs 1, 2, and 3 in the new genomes in an organization typical for HEV (Figure 1, panel A). In contrast to HEV genotypes 1–3, rat HEV ORFs 1 and 3 do not overlap. Three additional putative ORFs of 280–600 nt that overlap with ORFs 1 or 2 were predicted for each rat HEV genome (Figure 1, panel A). However, before the meaning of these findings can be verified, sequence information from additional rat HEV strains and experimental proof are needed. Phylogenetic analyses of a 1,576-nt segment available for all published rat HEV sequences demonstrated clear separation from mammalian genotypes 1–4 and avian strains (Figure 1, panel B). The same 3 phylogenetic clusters were obtained when the complete genomes were analyzed (Figure 1, panel C) and when the nucleotide and deduced amino acid sequences of ORF1, ORF2, and ORF3 were investigated separately (data not shown).
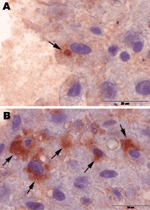
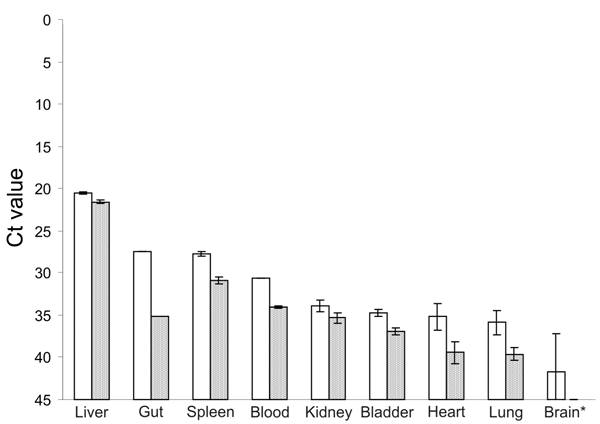
To compare viral load in different tissues of the 2 HEV–positive rats, we developed a real-time RT-PCR selective for a region in the ORF2 of rat HEV. Parallel analysis of RNA isolated from 10 mg of each tissue or 10 µL of blood reproducibly showed the highest viral load to be in the liver; cycle threshold values for liver were 20.5 and 21.6 for each animal and lower for all other tissues (Figure A1). Further, immunohistochemical analysis, using anti-HEV serum, detected viral antigen in the cytoplasm of a few hepatocytes from each HEV-positive rat. Antigen was also observed in some activated hepatic stellate cells (Figure 2). Hematoxylin-eosin staining showed a marginally increased number of monocytes and granulocytes in sinusoids as well as a moderately increased number of lymphocytes and plasma cells in some Glisson triads of the livers (data not shown).
Phylogenetic analyses and nucleotide and amino acid sequence comparisons demonstrated that the complete rat HEV genome sequences were consistently well separated from those of mammalian genotypes 1–4 and the tentative avian genotype. This finding suggests that these sequences represent an additional genotype (Figure 1, Table). In our analyses, the recently described HEV strain found in domestic rabbits, proposed to represent a separate genotype (13), clustered with human HEV genotypes irrespective of the genome part, nucleotide, or deduced amino acid sequences analyzed (Figure 1, panels B, C, and data not shown). Therefore, this strain may represent the consequence of recent spillover rather than the result of long-term virus–host coevolution. In contrast, the nonzoonotic avian HEV strains strongly differ from the mammalian HEV genotypes 1–4 (Figure 1, panels B, C). Although in the genus Hepevirus no species demarcation criteria have been defined, the marked sequence diversities suggest that the rat HEV represents an additional virus species other than HEV-1, HEV-2, HEV-3, HEV-4, and the tentative species avian HEV, which are currently classified in this genus (14).
Detection of rat HEV RNA and antigen in the liver cells of the infected Norway rats may indicate hepatotropism of this virus. Therefore, regarding its organ and cell-type tropism, this virus seems to be similar to the human and pig HEV genotypes (15). Because the virus was also detected in the intestine and, in the previous study, in feces (10), fecal–oral transmission as for genotypes 1–4 is plausible. The common properties of this virus and the human HEV genotypes suggest the usefulness of developing an HEV model in laboratory rats. In addition, the detection of rat HEV in animals from an urban region in Germany raises questions about the putative epidemiologic role of rat HEV for hepatitis E in humans.
Dr Johne is a senior scientist at the Federal Institute for Risk Assessment, Berlin. His research interests are the epidemiology, molecular evolution, and pathogenesis of food-borne viruses.
Acknowledgments
We thank Paul Dremsek, Dörte Kaufmann, Konrad Wanka, and Fabian Deutskens for necropsy of the rats; Ina Römer and Maria-Margarida Vargas for excellent technical assistance; Christoph Gille for ORF analysis and helpful discussions; and the governmental pest control staff of the Institute of Hygiene and Environment, Hamburg, for trapping the rats.
This work was funded in part by the German Federal Ministry of Food, Agriculture and Consumer Protection, through the Federal Institute for Agriculture and Nutrition, grant nos. 07HS026 and 07HS027.
References
- Meng XJ. Hepatitis E virus: animal reservoirs and zoonotic risk. Vet Microbiol. 2009;140:256–65. DOIPubMedGoogle Scholar
- Teshale EH, Howard CM, Grytdal SP, Handzel TR, Barry V, Kamili S, Hepatitis E epidemic, Uganda. Emerg Infect Dis. 2010;16:126–9. DOIPubMedGoogle Scholar
- Lewis HC, Wichmann O, Duizer E. Transmission routes and risk factors for autochthonous hepatitis E virus infection in Europe: a systematic review. Epidemiol Infect. 2010;138:145–66. DOIPubMedGoogle Scholar
- Schielke A, Sachs K, Lierz M, Appel B, Jansen A, Johne R. Detection of hepatitis E virus in wild boars of rural and urban regions in Germany and whole genome characterization of an endemic strain. Virol J. 2009;6:58. DOIPubMedGoogle Scholar
- Kabrane-Lazizi Y, Fine JB, Elm J, Glass GE, Higa H, Diwan A, Evidence for widespread infection of wild rats with hepatitis E virus in the United States. Am J Trop Med Hyg. 1999;61:331–5. PubMedGoogle Scholar
- Favorov MO, Kosoy MY, Tsarev SA, Childs JE, Margolis HS. Prevalence of antibody to hepatitis E virus among rodents in the United States. J Infect Dis. 2000;181:449–55. DOIPubMedGoogle Scholar
- Arankalle VA, Joshi MV, Kulkarni AM, Gandhe SS, Chobe LP, Rautmare SS, Prevalence of anti–hepatitis E virus antibodies in different Indian animal species. J Viral Hepat. 2001;8:223–7. DOIPubMedGoogle Scholar
- Easterbrook JD, Kaplan JB, Vanasco NB, Reeves WK, Purcell RH, Kosoy MY, A survey of zoonotic pathogens carried by Norway rats in Baltimore, Maryland, USA. Epidemiol Infect. 2007;15:1–8.PubMedGoogle Scholar
- Ulrich RG, Schmidt-Chanasit J, Schlegel M, Jacob J, Pelz H-J, Mertens M, Network “Rodent-borne pathogens“ in Germany: longitudinal studies on the geographical distribution and prevalence of hantavirus infections. Parasitol Res. 2008;103(Suppl. 1):S121–9. DOIPubMedGoogle Scholar
- Johne R, Plenge-Bönig A, Hess M, Ulrich RG, Reetz J, Schielke A. Detection of a novel hepatitis E–like virus in feces of wild rats using a nested broad-spectrum RT-PCR. J Gen Virol. 2010;91:750–8. DOIPubMedGoogle Scholar
- Koonin EV, Gorbalenya AE, Purdy MA, Rozanov MN, Reyes GR, Bradley DW. Computer-assisted assignment of functional domains in the nonstructural polyprotein of hepatitis E virus: delineation of an additional group of positive-strand RNA plant and animal viruses. Proc Natl Acad Sci U S A. 1992;89:8259–63. Medline DOIPubMedGoogle Scholar
- Braaker S, Heckel G. Transalpine colonisation and partial phylogeographic erosion by dispersal in the common vole Microtus arvalis. Mol Ecol. 2009;18:2518–31. DOIPubMedGoogle Scholar
- Zhao C, Ma Z, Harrison TJ, Feng R, Zhang C, Qiao Z, A novel genotype of hepatitis E virus prevalent among farmed rabbits in China. J Med Virol. 2009;81:1371–9. DOIPubMedGoogle Scholar
- Emerson SU, Anderson D, Arankalle A, Meng X-J, Purdy M, Schlauder GG, Hepevirus. In: CM Fauquet, MA Mayo, J Maniloff, U Desselberger, LA Ball, editors. Virus taxonomy. San Diego: Elsevier Academic Press; 2005. p. 853–7.
- Lee YH, Ha Y, Ahn KK, Chae C. Localisation of swine hepatitis E virus in experimentally infected pigs. Vet J. 2009;179:417–21. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 16, Number 9—September 2010
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Address for correspondence. Rainer G. Ulrich, Friedrich-Loeffler-Institut, Federal Research Institute for Animal Health, OIE Collaborating Centre for Zoonoses in Europe, Institute for Novel and Emerging Infectious Diseases, Südufer 10, D-17493 Greifswald–Insel Riems, Germany
Top