Volume 11, Number 7—July 2005
Letter
West Nile Virus Detection and Commercial Assays
Cite This Article
Citation for Media
To the Editor: Roehrig and colleagues described the long-term persistence of immunoglobulin (Ig) M antibody in patients with West Nile virus (WNV) infection, as tested using an in-house Centers for Disease Control and Prevention (CDC) enzyme immunoassay (EIA) (1). This result suggests that interpreting WNV IgM results in subsequent years would be difficult. With the commercial availability and widespread use of US Food and Drug Administration–approved WNV IgM tests, we were concerned that this phenomenon might also occur with new tests. Thus in 2004, we initiated a follow-up study of patients infected during the inaugural (2003) WNV season in Alberta, Canada.
Fifty patients who were WNV IgM positive by 2 commercial IgM kits (West Nile virus capture EIAs, Focus Technologies, Cypress, CA, USA, and Panbio, Windsor, Queensland, Australia) during the fall of 2003 were contacted. Sera were recollected and tested for IgM and IgG antibodies to WNV with current kits from these 2 companies. Sera were also tested for hemagglutination-inhibiting (HI) antibodies to WNV (2).
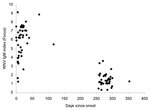
Of 39 serum samples from 38 patients, 28 were positive, 5 were indeterminate, and 6 were negative with the Focus IgM kit. Twenty-one were positive, 3 were indeterminate, and 14 were negative with the Panbio IgM kit. All had WNV IgG antibodies detected by Focus and Panbio IgG kits. We detected HI antibodies to WNV in all patients, and titers in 12 were ≥320. The time course for IgM index values for the Focus IgM kit used in 2003 and 2004 is shown in the Figure.
These data show that when tests are conducted with newly available kits, as with the CDC in-house test, IgM antibody to WNV persists for ≥8 months in most patients. A single high HI titer is not helpful in identifying recent infection. In addition, the IgM test cannot differentiate between recent and past infections. Interpreting a positive IgM result in WNV-endemic areas will be complex because a positive WNV IgM result could indicate a current acute infection or a previous WNV infection even in a person with a different acute illness.
Acknowledgments
We thank M. O'Connor and the staff of the Medicine Hat Regional Hospital laboratory for assistance with specimen collection and transport.
This work was supported by Alberta Health and Wellness, Calgary, Alberta, Canada.
References
- Roehrig JT, Nash D, Maldin B, Labowitz A, Martin DA, Lanciotti RS, Persistence of virus-reactive serum immunoglobulin M antibody in confirmed West Nile virus encephalitis cases. Emerg Infect Dis. 2003;9:376–9.PubMedGoogle Scholar
- Beaty BJ, Calisher CH, Shope RE. Arboviruses. In: Schmidt NJ, Emmons RW, editors. Diagnostic procedures for viral, rickettsial, and chlamydial infections. 6th ed. Washington: American Public Health Association, 1989; p.797–856.
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 11, Number 7—July 2005
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Peter A.G. Tilley, Provincial Laboratory for Public Health, 3030 Hospital Drive, Calgary, Alberta, Canada T2N 4W4; fax: 403-283-0142
Top