Volume 12, Number 6—June 2006
Dispatch
Guillain-Barré Syndrome, Greater Paris Area
Cite This Article
Citation for Media
Abstract
We studied 263 cases of Guillain-Barré syndrome from 1996 to 2001, 40% of which were associated with a known causative agent, mainly Campylobacter jejuni (22%) or cytomegalovirus (15%). The cases with no known agent (60%) peaked in winter, and half were preceded by respiratory infection, influenzalike syndrome, or gastrointestinal illness.
Guillain-Barré syndrome (GBS) is a state of acute flaccid paralysis thought, in most cases, to result from an aberrant immune response triggered by microbial infections (1). Studies in Western countries have reported evidence of recent infection with Campylobacter jejuni in 15% to 40% of GBS cases and with cytomegalovirus (CMV) in 5% to 20% of cases (1–5). Recent infection with Epstein-Barr virus (EBV) or Mycoplasma pneumoniae was less frequent (1%–2% each) (1–5). No agent was identified in 60% to 70% of cases, although the patients often had a history of respiratory or gastrointestinal infection (1,2).
Previous studies have failed to identify any clear seasonal distribution of GBS cases in Europe and North America. It has been suggested that this failure to demonstrate seasonality in GBS is because most prevalent antecedent infections have inverse seasonal distributions (6). We tested this hypothesis to provide new insight into infectious agents associated with GBS in Western countries.
All GBS patients admitted to the medical intensive care unit of Raymond Poincaré Hospital in Garches, France, from January 1996 to December 2001 were included in the study. GBS was diagnosed according to the criteria of Asbury and Cornblath (7). The following data were collected at the time of hospital admission: time from onset of neurologic signs of GBS to admission; history of infections in the 2 months preceding the onset of neurologic signs; and time from the infectious event to onset of neurologic signs.
Serum samples were collected at hospital admission. Serum antibodies against C. jejuni and M. pneumoniae were assayed with complement fixation tests (Institut Virion, Würzburg, Germany); cutoff titers (C. jejuni 20; M. pneumoniae 80) were selected to give >95% specificity on the basis of data provided by the manufacturer. Serum samples were tested for immunoglobulin M (IgM) and IgG antibodies to CMV with the miniVIDAS system (bioMérieux, Marcy l'Etoile, France). IgG avidity was measured in samples positive for IgM by using the Enzygnost anti-CMV/IgG test (Dade Behring S.A., Paris la Défense, France) and 8 mol/L urea. Recent CMV infection was identified by detection of IgM with IgG avidity <35% (8). Serum antibodies against EBV were detected with commercial dot blot tests (ImmunoDOT EBV MONO M and G kits, GenBio, San Diego, CA, USA). Recent EBV infection was identified by detecting IgM antibodies to viral capsid antigen. IgM and IgG antibodies against gangliosides GM1 and GM2 were identified by an enzyme immunoassay (GanglioCombi, Bühlmann Laboratories AG, Schönenbuch, Switzerland) and an immunodot blot assay (9).
Statistical analyses were performed with the R 2.0.1 statistical package (R Development Core Team, Vienna, Austria). Groups were compared in pairs, and the Hochberg method for multiple testing was used to correct p values (10). Categorical variables were compared by Fisher exact test, and continuous variables were compared by Student t test or Wilcoxon rank sum test. Seasonal trends for GBS cases were analyzed by using the method of Jones et al. (11). The number of harmonics (seasonality periods) was determined by using the Akaike information criterion. All tests were 2-tailed, and a p value <0.05 was considered significant.
During the study period, 279 consecutive patients with GBS were admitted to our center. Sixteen patients were excluded because of missing clinical data or serum samples; 263 were included in the study. On the basis of an annual incidence of 1.2 to 1.9 GBS cases per 100,000 persons (12) in a population of 10.952 million people (13), we estimated that 130–210 GBS cases occurred annually in the greater Paris area during the study period. Thus, this study included 20%–30% of all estimated GBS cases in this area.
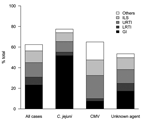
We observed serologic evidence of recent infection with C. jejuni in 58 patients (21.9%), CMV in 40 (15.1%) patients, M. pneumoniae in 6 (2.3%) patients, and EBV in 3 (1.15%) patients. Recent infection with C. jejuni and CMV was observed in 1 patient. Thus, 106 cases (40%) had >1 known agent of GBS (known agent group), and 157 cases (60%) had no known agent (unknown agent group) (Table). Most patients in the C. jejuni group were male, were >50 years of age, had a history of gastrointestinal illness (Figure 1), and exhibited a severe motor form of GBS with serum IgG antibodies against ganglioside GM1. Patients in the CMV group were significantly younger (p<0.0001), more likely to have respiratory or influenzalike symptoms than gastrointestinal symptoms (p<0.0001) before the onset of GBS symptoms (Figure 1), and showed a longer time from first neurologic signs to hospital admission (p = 0.048). These patients rarely showed a pure motor form of GBS (p = 0.037) and frequently had IgM antibodies against GM2 but did not have IgG antibodies against GM1 (p<0.0001).
Patients in the unknown agent group were older than those in the CMV group (p<0.0001), less likely to have had a history of infectious events than patients in the C. jejuni group (p = 0.0048), and had a significantly different antiganglioside response than those in C. jejuni and CMV groups (p<0.0001 in each case) (Table). The unknown agent group had a higher proportion of patients with gastrointestinal illness than did the CMV group (p = 0.045) and a higher proportion of patients with respiratory tract or influenzalike symptoms than the C. jejuni group (p = 0.0024) (Figure 1).
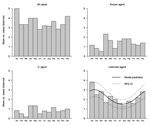
No seasonal variation was found for all patients combined (data not shown). However, this apparent absence of variation masked a substantial seasonal difference for the known agent and unknown agent groups. In the known agent group, 60% of cases occurred in spring and summer; only 16% occurred in winter. In the unknown agent group, only 17% of cases occurred in summer; 37% occurred in winter.
We used the method of Jones et al. (11) to test the seasonality of incidence. No seasonality was detected for the groups all cases, known agent, and C. jejuni (Figure 2). For the unknown agent group, a model with 1 harmonic (annual seasonality) gave a significantly better fit than a model without harmonics (p = 0.0089, by likelihood ratio test); additional harmonics did not improve the fit of the model. Since no significant linear trend was found (p = 0.49), this element was removed for model prediction. This best-fit, single-harmonic model indicated that incidence was highest at the beginning of February and lowest at the beginning of August (Figure 2).
This study provides new data about GBS patients not associated with known etiologic agents, which account for most patients in Western Europe (2,14). We have shown that GBS cases of unknown cause were more common in winter, with a peak incidence at the beginning of February. Moreover, in ≈50% of the patients, GBS symptoms were preceded by respiratory infection, influenzalike syndrome, or gastrointestinal illness. Together with the seasonality of cases, this finding suggests the involvement of winter infectious agents, probably respiratory or enteric viruses.
Dr Sivadon-Tardy is a microbiologist at Raymond Poincaré Hospital in Garches, France. Her primary research interests are molecular epidemiology and emerging and reemerging infectious diseases.
Acknowledgments
We thank Isabelle Sénégas for assistance and Marie-Hélène Canneson for technical assistance.
This work was supported by the Laboratoire Français du Fractionnement et des Biotechnologies.
References
- Hughes RA, Hadden RD, Gregson NA, Smith KJ. Pathogenesis of Guillain-Barré syndrome. J Neuroimmunol. 1999;100:74–97. DOIPubMedGoogle Scholar
- Hadden RD, Karch H, Hartung HP, Zielasek J, Weissbrich B, Schubert J, Preceding infections, immune factors, and outcome in Guillain-Barré syndrome. Neurology. 2001;56:758–65.PubMedGoogle Scholar
- Rees JH, Soudain SE, Gregson NA, Hughes RA. Campylobacter jejuni infection and Guillain-Barré syndrome. N Engl J Med. 1995;333:1374–9. DOIPubMedGoogle Scholar
- Visser LH, van der Meche FG, Meulstee J, Rothbarth PP, Jacobs BC, Schmitz PI, Cytomegalovirus infection and Guillain-Barré syndrome: the clinical, electrophysiologic, and prognostic features. Dutch Guillain-Barré Study Group. Neurology. 1996;47:668–73.PubMedGoogle Scholar
- Jacobs BC, Rothbarth PH, van der Meche FG, Herbrink P, Schmitz PI, de Klerk MA, The spectrum of antecedent infections in Guillain-Barré syndrome: a case-control study. Neurology. 1998;51:1110–5.PubMedGoogle Scholar
- Hughes RA, Rees JH. Clinical and epidemiologic features of Guillain-Barré syndrome. J Infect Dis. 1997;176(Suppl 2):S92–8. DOIPubMedGoogle Scholar
- Asbury AK, Cornblath DR. Assessment of current diagnostic criteria for Guillain-Barré syndrome. Ann Neurol. 1990;27:S21–4. DOIPubMedGoogle Scholar
- Grangeot-Keros L, Mayaux MJ, Lebon P, Freymuth F, Eugene G, Stricker R, Value of cytomegalovirus (CMV) IgG avidity index for the diagnosis of primary CMV infection in pregnant women. J Infect Dis. 1997;175:944–6. DOIPubMedGoogle Scholar
- Chabraoui F, Derrington EA, Mallie-Didier F, Confavreux C, Quincy C, Caudie C. Dot-blot immunodetection of antibodies against GM1 and other gangliosides on PVDF-P membranes. J Immunol Methods. 1993;165:225–30. DOIPubMedGoogle Scholar
- Hochberg Y. A sharper Bonferroni procedure for multiple tests of significance. Biometrika. 1988;75:800–3. DOIGoogle Scholar
- Jones RH, Ford PM, Hamman RF. Seasonality comparisons among groups using incidence data. Biometrics. 1988;44:1131–44. DOIPubMedGoogle Scholar
- French Population Census. March 1999. The results. Paris: National Institute for Statistics and Economic Studies. Version 2.30.07. 2005 Dec 12. [cited 2006 Feb 6]. Available from http://www.recensement.insee.fr/RP99
- Van Koningsveld R, van Doorn PA, Schmitz PI, Ang CW, van der Meche FG. Mild forms of Guillain-Barré syndrome in an epidemiologic survey in The Netherlands. Neurology. 2000;54:620–5.PubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 12, Number 6—June 2006
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Valérie Sivadon-Tardy, Laboratoire de Microbiologie, Hôpital Raymond Poincaré, 104 Blvd Raymond Poincaré, CEDEX 92380, Garches, France
Top