Volume 12, Number 8—August 2006
Dispatch
Changing Pattern of Visceral Leishmaniasis, United Kingdom, 1985-2004
Cite This Article
Citation for Media
Abstract
A 20-year (1985–2004) retrospective review of 39 patients with imported visceral leishmaniasis found that tourism to Mediterranean countries and HIV infection were associated with visceral leishmaniasis. Diagnosis was often delayed. Treatment with liposomal amphotericin B has improved prognosis. Visceral leishmaniasis should be made a reportable disease.
Each year, 500,000 new cases of visceral leishmaniasis (VL) are reported worldwide. The number of cases and endemic foci for VL have increased during the past 2 decades (1–4). These increases may be the result of improved detection and surveillance methods, or they may be actual increases in numbers, possibly driven by increasing rates of HIV infection (5,6). VL is a well-recognized but uncommon imported disease in the United Kingdom, but it is not reportable, so information on its importation is incomplete. Data for this report (e.g., date of confirmation of infection, country where acquired, HIV status) were collated and supplied by the Travel Health Surveillance Section of the Health Protection Agency, Communicable Disease and Surveillance Centre, United Kingdom.
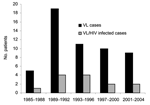
UK patients with VL are often referred to the Hospital for Tropical Diseases, London, for confirmation of diagnosis or treatment. We reviewed all cases of VL seen at this hospital from 1985 through 2004. Thirty-nine patients were identified from our hospital database and laboratory records, representing 83% of cases reported in the United Kingdom (Figure 1).
The mean age of these patients was 36 years (range 2–66 years); 4 patients were <15 years of age. The male:female ratio was 2:1. Patients acquired visceral leishmaniasis while in the following areas: 30 (76.9%) in Mediterranean countries (13 in Spain, 9 in Italy, 4 in Greece, 3 in Malta, 1 in Cyprus), 5 (12.8%) from Africa, 3 (7.7%) from Asia, and 1 (2.6%) from South America. During the entire 20-year period, 55.5% of patients had been tourists to these VL-endemic regions, and 44.5% were immigrants or refugees, but after 2000, all patients were tourists. One third of the patients were HIV-antibody positive. Two patients had significant immunosuppression from other causes: 1 from chronic lymphatic leukemia and 1 from immunosuppressive drugs received after kidney transplantation.
Time from onset of symptoms to diagnosis was 1–11 months (mean 3 months). Diagnoses of VL were confirmed by >1 method: microscopic identification of amastigotes in tissue aspirates; histologic examination of biopsy material from bone marrow, liver, or spleen; serologic analysis; and PCR for leishmania DNA (7). Leishmania amastigotes were found in 32 bone marrow aspirates, 3 liver biopsy specimens, 2 splenic aspirates, and 1 skin biopsy specimen. PCR to detect leishmania DNA has been performed in this study since 1995 and was positive in 7 of 11 cases. It was the method of confirmatory diagnosis for 1 patient (performed on a bone marrow aspirate that had no visible amastigotes). Serum analysis for leishmania antibodies was performed on samples from 33 patients; results were positive for all HIV-negative patients and for 2 of 13 HIV-positive patients (Table 1).
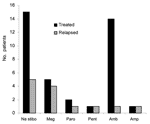
Before 1995, treatment of leishmaniasis involved several drugs, including sodium stibogluconate, paromomycin, meglumine antimoniate, and pentamidine. The latter 2 were used for patients who received initial treatment outside the United Kingdom; within the United Kingdom, most (59%) patients were treated with sodium stibogluconate. After 1995, liposomal amphotericin became the drug of choice and was used in 14 (83%) of 17 patients. HIV status did not affect drug choice.
Figure 2 shows the frequency of relapses with each drug treatment in our cohort. Of the 13 patients who had a relapse, 8 (53%) had HIV coinfection. Of the 13 who had HIV coinfection, 8 (62%) had a relapse, compared with 7 (27%) of the 26 HIV-negative patients who had a relapse. Two patients had other risk factors for relapse: 1 had had a kidney transplant, and the other had chronic active hepatitis B. Three patients who had relapses had no apparent risk factors. Relapse occurred in 5 (33%) of 15 patients who received sodium stibogluconate, compared with 1 (7%) of 14 who received liposomal amphotericin as their initial drug. At first, relapsed patients were treated with a combination of sodium stibogluconate and allopurinol, which was unsuccessful in contrast with its reported success in Kenya, or with amphotericin deoxycholate (8). HIV-infected patients who had relapses received further treatment with liposomal amphotericin, usually successful, but 2 became unresponsive to treatment. Miltefosine was used in 2 patients with HIV coinfection who had relapses, but relapses recurred, 1 while the patient was still receiving the drug (9). Since 2000, pentamidine prophylaxis has been used in 3 immunocompromised patients.
Table 2 summarizes outcomes. The number of patients in the final cure category excludes patients who had had a relapse. The 2 patients who refused treatment had HIV coinfection; 1 died. After 1995, the final cure rate improved and fewer patients required retreatment. The 3 patients who died had advanced HIV disease; 1 had an additional complication of liver failure secondary to hepatitis C cirrhosis.
The data from this cohort show that VL is imported into the United Kingdom at a constant rate, particularly in adult male tourists to the Mediterranean, and that during the past 20 years, HIV and VL had interacted strikingly. The Mediterranean VL-endemic zone accounts for only a small proportion of VL cases globally, but it is a popular holiday destination for the British, and tourism is driving the epidemiology of the imported infection. In the past 5 years, we have seen no immigrants or refugees with VL. The data show that high rates of tourism to an area of low endemicity for a parasitic disease can result in a substantial number of imported cases.
One third of our patients were immunocompromised. The highest rates of reported HIV coinfection worldwide are from Europe, where 85% of the cases are from the southwest; 71% of these are in intravenous drug users (2). HIV/VL coinfection is a serious disease that has high death rates, reflected by the 3 deaths in our cohort. These patients had late-stage HIV disease and additional coexisting conditions. HIV/VL-coinfected patients have higher relapse rates and decreased life expectancy. Highly active antiretroviral therapy for HIV is decreasing the numbers of these coinfections and improving survival rates (10,11).
The mean time from symptom onset to diagnosis was 3 months. This delay resulted mainly from physicians’ failure to consider the diagnosis, although a few cases were difficult to diagnose. For 2 patients, initial microscopic examination of bone marrow biopsy results did not show amastigotes; reinspection showed scarce amastigotes. For another patient, serologic results by direct agglutination were negative when performed early in the course of disease but positive when repeated. The low sensitivity of serologic tests for HIV-infected patients in this cohort confirms that serologic tests cannot be relied on as a means of excluding VL in HIV-infected patients.
Our cohort shows a rise and fall in the proportion of cases of HIV/VL coinfection, starting in the early 1990s, rising to a peak of 50% during 1993–1996, and then decreasing, possibly because of greater use of highly active antiretroviral therapy. Official UK data on VL are incomplete and do not accurately reflect trends. VL remains an imported disease in the United Kingdom and is often associated with HIV infection. We suggest that a formal notification system for visceral leishmaniasis in the United Kingdom, as elsewhere in Europe, would be beneficial for monitoring trends and health planning (12). Our data also show the importance of looking at rare imported diseases over a long period so that emerging risk factors can be identified. These data also highlight the usefulness of having 1 center that deals with unusual infections, where expertise in diagnosis and management can be built up and maintained.
Dr Malik was senior house officer at the Hospital for Tropical Diseases when she became interested in VL. She is now pursuing a career in ophthalmology.
Acknowledgment
We thank Maggie Armstrong for help with the hospital database and our colleagues in the Department of Parasitology, particularly Kalim Lalloo, for their sustained interest and the provision of parasitologic data. Rob Miller gave valuable help in collecting information from HIV-coinfected patients.
References
- Desjeux P. The increase in risk factors for leishmaniasis worldwide. Trans R Soc Trop Med Hyg. 2001;95:239–43. DOIPubMedGoogle Scholar
- Gabutti G, Balestra G, Fkego G, Crovari P. Visceral leishmaniasis in Liguria, Italy. Lancet. 1998;351:1136. DOIPubMedGoogle Scholar
- Punda-Polic V, Sardelic S, Bradaric N. Visceral leishmaniasis in southern Croatia. Lancet. 1998;351:188. DOIPubMedGoogle Scholar
- World Health Organization. Leishmaniasis and leishmania/HIV co-infection. WHO report on global surveillance of epidemic-prone infectious diseases 2000. Report no. WHO/CDS/CSR/ISR/2000.1. Geneva: The Organization; 2000.
- Guerin PJ, Olliaro P, Sundar S, Boelaert M, Croft SL, Desjeux P, Visceral leishmaniasis: current status of control, diagnosis, and treatment, and a proposed research and development agenda. Lancet Infect Dis. 2002;2:494–501. DOIPubMedGoogle Scholar
- Alvar J, Canavate C, Guiterrez-Solar B, Jimenez M, Laguna F, Lopez-Velez R, Leishmania and human immunodeficiency virus co-infection: the first 10 years. Clin Microbiol Rev. 1997;10:298–319.PubMedGoogle Scholar
- Noyes HA, Reyburn H, Bailey W, Smith D. A nested PCR based schizodeme method for identifying Leishmania kinetoplast minicircle classes directly from clinical samples and its application to the study of the epidemiology of Leishmania tropica in Pakistan. J Clin Microbiol. 1998;36:2877–81.PubMedGoogle Scholar
- Chunge CN, Gachini G, Muigai R, Wasunna K, Rashid JR, Chulay JD, Visceral leishmaniasis in response to antimonial drugs. III. Successful treatment using a combination of sodium stibogluconate plus allopurinol. Trans R Soc Trop Med Hyg. 1985;79:715–8. DOIPubMedGoogle Scholar
- Sindermann H, Engel KR, Fischer C, Bommer W. Oral miltefosine for leishmaniasis in immunocompromised patients: compassionate use in 39 patients with HIV co-infection. Clin Infect Dis. 2004;39:1520–3. DOIPubMedGoogle Scholar
- Pagliano P, Rossi M, Rescigno C, Altieri S, Coppola MG, Gramiccia M, Mediterranean visceral leishmaniasis in HIV negative adults: a retrospective analysis of 64 consecutive cases (1995–2001). J Antimicrob Chemother. 2003;52:264–8. DOIPubMedGoogle Scholar
- Russo R, Laguna F, Lopez-Velez R, Medrano FJ, Rosenthal E, Cacopardo B, Visceral leishmaniasis in those infected with HIV: clinical aspects and other opportunistic infections. Ann Trop Med Parasitol. 2003;97:99–105. DOIPubMedGoogle Scholar
- Harms G, Schonian G, Feldmeier H. Leishmaniasis in Germany. Emerg Infect Dis. 2003;9:872–5.PubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 12, Number 8—August 2006
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Diana NJ Lockwood, Hospital for Tropical Diseases, London, UK WC1E 6AU
Top