Volume 15, Number 12—December 2009
Dispatch
Spatiotemporal Dynamics of Hemorrhagic Fever with Renal Syndrome, Beijing, People’s Republic of China
Cite This Article
Citation for Media
Abstract
We used geographic information systems to characterize the dynamic change in spatial distribution of hemorrhagic fever with renal syndrome (HFRS) in Beijing, People’s Republic of China. The seasonal variation in its incidence was observed by creating an epidemic curve. HFRS was associated with developed land, orchards, and rice paddies.
Hemorrhagic fever with renal syndrome (HFRS), a rodent-borne disease caused by hantaviruses (family Bunyaviridae), is characterized by fever, acute renal dysfunction, and hemorrhage manifestations. Various rodent species are natural hosts and serve as sources of infection (1). Humans usually acquire hantavirus infection by contact with or inhalation of aerosols or excreta from infected rodents (2,3). In the People’s Republic of China, HFRS is caused mainly by 2 types of hantaviruses, Hantaan virus (HTNV) and Seoul virus (SEOV), each of which has co-evolved with a distinct rodent host. HTNV is associated with Apodemus agrarius, whereas SEOV, which causes a less severe form of HFRS, is associated with Rattus norvegicus. Although HFRS infection has long been recognized in many places throughout mainland China, HFRS was first reported in metropolitan Beijing in 1997. Since then, the natural foci have been established, and human cases were continuously diagnosed in the new disease-endemic region (4).
The presence and transmission of hantavirus depend on the distribution and infection of its animal hosts, which largely determine the incidence and extent of HFRS; such distribution and infection are usually determined by environmental elements (5,6). Ecologic studies in China demonstrated that elevation, precipitation, temperature, vegetation type, and soil type influenced transmission of HTNV (7,8). However, these studies were conducted on a relatively large scale, usually at the county or even province level. Environmental factors driving variability in HFRS incidence at a finer scale (e.g., township) remain poorly understood. The availability of detailed records of HFRS cases and environmental information in the newly established disease-endemic region provides an opportunity to explore possible factors underlying the emergence of the rodent-borne disease. In this study, we aimed to learn the current situation of endemic HFRS in Beijing, characterize its spatiotemporal distribution, and identify environmental factors possibly contributing to the incidence of the disease.
The study area covered metropolitan Beijing (between 115°20′ and 117°30′E and 39°28′ and 41°05′N), including 220 townships of 18 districts with an area of 16,800 km2. The data on reported HFRS cases were obtained from the National Notifiable Disease Surveillance System, which included information about sex, age, residential address, and onset date of symptoms for each case.
A total of 852 HFRS cases were reported in Beijing metropolis during 1997–2006. The annual incidence of each district was calculated by using the fifth national census data in 2000 and mapped by using a geographic information system (GIS) technique by digitalizing village, street, and boundaries on the 1:100,000 topographic map of Beijing in ArcGIS 9.0 software (ESRI Inc., Redlands, CA, USA). Each HFRS case was geocoded according to residential address, and a layer including information about HFRS cases was created and overlayed on the digital map. By 2000, the disease had affected all the area of the city. However, the incidence in each district varied during the 10-year study period (Figure 1).
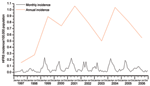
An epidemic curve was created to show the temporal distribution of HFRS in Beijing. The annual incidence had sharply increased from 1997 to 1999; thereafter it fluctuated around 0.8 ± 0.2/100,000 persons (Figure 2). Within each year, the incidence varied markedly; most cases occurred in winter and spring, usually peaking in April, although the disease was reported in almost every month (Figure 2).
To study factors related to HFRS spread, we compared the incidence with various environmental indicators. Data on elevation were collected from a digital elevation model. Land cover data were derived from 2005 Landsat 7 enhanced thematic mapper image by using ENVI 4.0 software (Research Systems Inc., Boulder, CO, USA). Land use types were classified as follows: built-up land, water body, dry land, scrub, orchard, irrigable land, rice paddies, and forest. Average elevation and area of different land use type of each township were calculated by ArcTools and Spatial Analyst module in ArcGIS 9.0 software (ESRI Inc.).
To determine the associations between the number of HFRS cases per township during the study period and elevation, as well as land use type, we performed standard Poisson regression analysis by using STATA 9.1 software (StataCorp LP, College Station TX, USA) (9). The percentage change in incidence in response to the value change of the variable by a given amount and its 95% confidence interval and corresponding p value were estimated after correction for overdispersion. The Poisson regression analysis indicated that built-up land, orchard, and rice paddies were significantly associated with HFRS at p<0.05 (Table). The incidence rose with increasing coverage of orchard and rice paddies, but dropped with decreasing coverage of built-up land.
Since the first local infection was reported in 1997, HFRS cases have occurred in all 18 districts of Beijing, with a fairly stable annual incidence since 1999. The dynamic change in spatial distribution confirmed the focal nature of the rodent-borne disease. The seasonality is one of the epidemiologic characteristics of HFRS, and further indicates that HFRS in the region is caused by SEOV with domestic rats, with mainly R. norvegicus as the source of infection (10,11) The rat population apparently peaks in winter, resulting in a lagged impact on transmission and seasonal variation of HFRS. In addition, rats are more active in homes in the winter because of cold temperature outside; this increases the chance for humans to acquire the infection by contact or inhalation of aerosols and secretions from infected rodents.
Our findings suggest that residents of townships with not too much built-up land but with orchard or rice paddies are at highest risk for infection. The land-use variables (acting as economic development indicators) are likely to be suitable for predicting the presence and distribution of HFRS in Beijing, where the disease recently emerged and economic development has greatly increased. Suitable control measures, such as removing rodents and preventing them from entering houses and human food storage buildings, should be taken to reduce incidence in this new disease-endemic region. However, the emergence and endemicity of HFRS are not determined only by the economic development activities, although they are important environmental contributors to the transmission of the disease. Biologic, ecologic, and social factors such as population immunity level, abundance and infection rate of host rodents, and human behavior, also may affect transmission of HFRS. Further epidemiologic and ecologic studies are required to understand the exact variables contributing to the emergence and extension of the disease during urbanization.
Dr Fang is an associate professor at the State Key Laboratory of Pathogen and Biosecurity, Beijing Institute of Microbiology and Epidemiology. His research interests focus on emerging infectious diseases, principally rodent-borne diseases and avian influenza.
Acknowledgment
This study was supported by the National Science Fund for Distinguished Young Scholars (no. 30725032), Special Program for Prevention and Control of Infectious Diseases (no. 2008ZX10004-012), Natural Science Foundation of China (no. 30590374), and Beijing Natural Science Foundation (no. 7061005).
References
- Li L-M, Ye D-Q, Shi L-Y. Epidemiology [in Chinese]. Beijing: China People’s Health Publishing House. 2003.
- Glass GE, Childs JE, Korch GW, LeDuc JW. Association of intraspecific wounding with hantaviral infection in wild rats (Rattus norvegicus). Epidemiol Infect. 1988;101:459–72. DOIPubMedGoogle Scholar
- Dournon E, Moriniere B, Matheron S, Girard PM, Gonzalez JP, Hirsch F, HFRS after a wild rodent bite in the Haute-Savoie and risk of exposure to Hantaan-like virus in a Paris laboratory. Lancet. 1984;323:676–7. DOIGoogle Scholar
- Zhang XC, Hu JY, Zhou SL, Guan ZZ. Study on epidemic state in Beijing. Chinese Journal of Vector Biology and Control. 2001;12:218–21.
- Engelthaler DM, Mosley DG, Cheek JE, Levy CE, Komatsu KK, Ettestad P, Climate and environmental patterns associcated with hantavirus pulmonary syndrome, Four Corners region, United States. Emerg Infect Dis. 1999;5:87–94. DOIPubMedGoogle Scholar
- Langlois JP, Fahrig L, Merriam G, Artsob H. Landscape structure influences continental distribution of hantavirus in deer mice. Landscape Ecol. 2001;16:255–66. DOIGoogle Scholar
- Bi P, Tong S, Donald K, Parton K, Ni J. Climatic, reservoir and occupational variables and the transmission of haemorrhagic fever with renal syndrome in China. Int J Epidemiol. 2002;31:189–93. DOIPubMedGoogle Scholar
- Yan L, Fang LQ, Huang HG, Zhang LQ, Feng D, Zhao WJ, Landscape elements and Hantaan virus–related hemorrhagic fever with renal syndrome, People’s Republic of China. Emerg Infect Dis. 2007;13:1301–6.PubMedGoogle Scholar
- Rabe-Hesketh S, Everitt BS. Handbook of statistical analyses using Stata. 4th ed. Chapman & Hall/CRC: Boca Raton (FL): 2006.
- Jiang JF, Zuo S, Zhang WY, Wu XM, Tang F, De Vlas SJ, Prevalence and gene diversities of hantaviruses in rodents in Beijing, China. Am J Trop Med Hyg. 2008;78:98–105.PubMedGoogle Scholar
- Zuo SQ, Zhang PH, Jiang JF, Zhan L, Wu XM, Zhao WJ, Seoul virus in patients and rodents from Beijing, China. Am J Trop Med Hyg. 2008;78:833–7.PubMedGoogle Scholar
Figures
Table
Cite This Article1These authors contributed equally to this article.
Table of Contents – Volume 15, Number 12—December 2009
| EID Search Options |
|---|
|
|
|
|
|
|