Volume 15, Number 12—December 2009
Dispatch
Cross-sectional Survey of Hantavirus Infection, Brazil
Cite This Article
Citation for Media
Abstract
A cross-sectional serosurvey was conducted to assess the proportion of persons exposed to hantaviruses in a virus-endemic area of the state of Minas Gerais, Brazil. Findings of this study suggested the presence of >1 hantaviruses circulating in this region causing hantavirus pulmonary syndrome, mild disease, or asymptomatic infection.
In Brazil, >1,080 cases of hantavirus pulmonary syndrome (HPS) have been confirmed since 1993 (case-fatality rate 40%). More HPS cases (209) are reported in the state of Minas Gerais than in any other state in Brazil (M.L. Nunes, pers. comm.). In Minas Gerais, molecular studies identified a hantavirus called Araraquara virus associated with HPS cases. The wild rodent Necromys lasiurus (the hairy-tailed bolo mouse, also named Bolomys lasiurus) was implicated as a reservoir of this virus (1). Because asymptomatic infection with hantaviruses also has been detected in Minas Gerais, we conducted a cross-sectional survey to assess the proportion of persons exposed to hantaviruses and to identify associated risk factors.
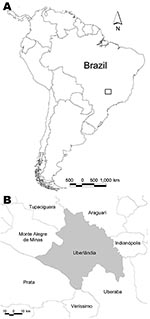
The hantavirus cross-sectional survey was carried out April through May 2006 in the municipality of Uberlândia, Minas Gerais, at an average altitude of 863 m (18º55′S,48º16′W) (Figure). A randomized and stratified (sex and age) sample was collected from the entire rural area and from the south sector of the municipality’s periurban area. The term periurban refers to a residential area on the outskirts of the city that is in close contact with the rural area. The participants answered a questionnaire that included demographic information (sex, age, place of birth, and address) and questions relating to HPS risk factors (type of dwelling, exposure to rodents at home or work, labor activity, risk activities, history of severe pneumonia, and direct contact with HPS patients). Blood samples were collected by venipuncture, centrifuged, and sent to the Laboratory of Hantaviruses and Rickettsioses at the Oswaldo Cruz Foundation, Rio de Janeiro, Brazil, for analysis. The ethics review board of the Federal University of Uberlândia approved the study.
We screened serum samples by ELISA for hantavirus-specific immunoglobulin G using a recombinant antigen of the nucleocapsid protein of Araraquara virus, produced in Escherichia coli and supplied by the Virus Research Unit of the University of São Paulo, Brazil, according to the procedure previously described (2). All positive serum samples were retested; only those that had 3 ELISA-positive results at >1:400 dilutions were considered positive.
The Mann-Whitney U and Fisher exact/binomial tests for 2 proportions were applied for comparison among medians and proportions, respectively, using EPI INFO 3.3.2 (www.cdc.gov/epiinfo) and BIOSTAT 5.0 (www.biostat.org) software. Fisher exact test was used to estimate the odds ratio and 95% confidence intervals.
The 400 study participants comprised 200 rural and 200 periurban residents ranging in age from 12 to 76 years (mean = 41 years). Twelve (3%) samples were hantavirus antibody-positive by ELISA. The 8 rural area antibody-positive samples were from male farmers (Table 1). Presence of antibody was significantly associated with male sex, older age class, and potential risk activities (Table 1). Although all case-patients reported exposure to rodents or their excreta, this exposure was not statistically significant (Table 1). In the periurban area, the presence of antibody was associated with age but not with sex, risk activity, or exposure to rodents (Table 1). The mean age of seropositive persons from periurban and rural areas was similar (p = 1.0). The relationship between antibody and sex depended on urban vs. rural residence (p = 0.02). Three antibody-positive persons in the rural zone and 2 in the urban zone reported a history of pneumonia, albeit without complications.
We examined data from an HPS outbreak in Uberlândia during 1998–2005. The largest number of cases occurred among periurban residents, but the highest cumulative incidence was among rural residents (Table 2). Nevertheless, rural and periurban areas did not differ significantly in either prevalence or incidence. We found higher prevalence among rural residents (Table 2).
Overall hantavirus antibody prevalence among periurban residents was 2.0%, with a higher prevalence among women (2.6%). In previous studies, the prevalence of hantavirus antibodies was higher in men (4–6). All the positive samples in the rural area came from male farm workers. This finding is similar to a situation reported in Colombia, where all positive samples came from men engaged in rural activities (6). These activities involve a high risk for infection by hantaviruses (7).
In this study, hantavirus positivity was found only in persons >39 years of age, and the difference in the mean age of the participants in relation to positivity was significant. This fact might suggest a historic high-risk event to which the older age class, but not the younger age class, was exposed.
High hantavirus antibody prevalence has been found in studies of some human populations in Latin America (5,8,9). The prevalence of Araraquara virus–reactive antibodies among the volunteers in this study demonstrates that transmission is not rare, reinforcing the hypothesis of the existence of mild disease or asymptomatic infections (10). Two hypotheses have been proposed: clinically mild disease or inapparent infections might result from differences in the nature of exposure (e.g., low inoculum or inefficient mechanism of transmission) or genetic differences in immune response to infection, or they might indicate the circulation of >1 hantavirus genotypes of greatly reduced virulence (10,11).
Mr Limongi is chief biologist of the Regional Center of Zoonotic Diseases of Uberlândia. His research interests focus on ecoepidemiology and of zoonotic disease control.
Acknowledgment
We thank Iram Martins Costa for excellent technical support.
References
- Suzuki A, Bisordi I, Levis S, Garcia J, Pereira LE, Souza RP, Identifying rodent hantavirus reservoirs, Brazil. Emerg Infect Dis. 2004;10:2127–34.PubMedGoogle Scholar
- Figueiredo LT, Moreli ML, Borges AA, Figueiredo GG, Souza RL, Aquino VH. Expression of a hantavirus N protein and its efficacy as antigen in immune assays. Braz J Med Biol Res. 2008;41:596–9. DOIPubMedGoogle Scholar
- Limongi JE, Costa FC, Paula MB, Pinto RM, Oliveira ML, Neto AA, Hantavirus cardiopulmonary syndrome in the Triângulo Mineiro and Alto Paranaíba regions, State of Minas Gerais, 1998–2005: clinical-epidemiological aspects of 23 cases [in Portuguese]. Rev Soc Bras Med Trop. 2007;40:295–9. DOIPubMedGoogle Scholar
- Mascarenhas-Batista AV, Rosa ES, Ksiazek TG, Rosa AP, Leduc JW, Pinheiro F, Antibodies anti-hantavirus in schoolchildren in Salvador, Bahia State, Brazil [in Portuguese]. Rev Soc Bras Med Trop. 1998;31:433–40. DOIPubMedGoogle Scholar
- Armien B, Pascale JM, Bayard V, Munoz C, Mosca I, Guerrero G, High seroprevalence of hantavirus infection on the Azuero Peninsula of Panama. Am J Trop Med Hyg. 2004;70:682–7.PubMedGoogle Scholar
- Máttar S, Parra M. Serologic evidence of hantavirus infection in humans, Colombia. Emerg Infect Dis. 2004;10:2263–4.PubMedGoogle Scholar
- Santos ED, Garrett DO. Evaluation of the hantavirus surveillance in Brazil [in Portuguese]. Epidemiologia e Serviços de Saúde. 2005;14:15–31.
- Ferrer JF, Jonsson CB, Esteban E, Galligan D, Basombrio MA, Peralta-Ramos M, High prevalence of hantavirus infection in Indian communities of the Paraguayan and Argentinean Gran Chaco. Am J Trop Med Hyg. 1998;59:438–44.PubMedGoogle Scholar
- Campos GM, Sousa RL, Badra SJ, Pane C, Gomes UA, Figueiredo LT. Serological survey of hantavirus infection in Jardinopolis County, Brazil. J Med Virol. 2003;71:417–22. DOIPubMedGoogle Scholar
- Pini N. Hantavirus pulmonary syndrome in Latin America. Curr Opin Infect Dis. 2004;17:427–31.Medline DOIPubMedGoogle Scholar
- Täger Frey MT, Vial PC, Castillo CH, Godoy PM, Hjelle B, Ferrés MG. Hantavirus prevalence in the IX Region of Chile. Emerg Infect Dis. 2003;9:827–32.PubMedGoogle Scholar
Figure
Tables
Cite This ArticleTable of Contents – Volume 15, Number 12—December 2009
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Jean E. Limongi, Regional Center of Zoonotic Diseases of Uberlândia, Alexandrino Alves Vieira Av, 1423 Liberdade District, 38401-240, Uberlândia, Minas Gerais, Brazil
Top