Volume 16, Number 12—December 2010
Dispatch
Brucella ceti Infection in Harbor Porpoise (Phocoena phocoena)
Cite This Article
Citation for Media
Abstract
We describe Brucella sp. infection and associated lesions in a harbor porpoise (Phocoena phocoena) found on the coast of Belgium. The infection was diagnosed by immunohistochemistry, transmission electron microscopy, and bacteriology, and the organism was identified as B. ceti. The infection’s location in the porpoise raises questions of abortion and zoonotic risks.
In cetaceans, Brucella spp. infections and related lesions have been found in bottlenose dolphins (Tursiops truncatus) (1), striped dolphins (Stenella coeruleoalba) (2–5), Atlantic white-sided dolphins (Lagenorhynchus acutus) (6,7), common dolphins (Delphinus delphi) (6,8), harbor porpoises (Phocoena phocoena) (6,9), and a minke whale (Balaenoptera acutorostrata) (6). Recently, B. ceti was described as being the cetacean Brucella sp. strain that infects dolphins (10). We report a case of B. ceti infection and associated lesions in a harbor porpoise found on the coast of Belgium in 2008.
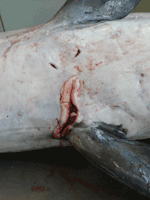
An adult female harbor porpoise died on the coast of Belgium in 2008, and a necropsy was immediately performed by the Marine Animals Research and Intervention Network (Belgium). The most relevant findings (Table) were emaciation and multiple large skin ulcers (acute to chronic) around the genital split and between flippers (Figure 1). Internally, mild to severe nematode infestation (of the right ventricle, pulmonary blood vessels, airways) was associated with acute pulmonary thrombi and severe acute necrotizing pneumonia. The liver was enlarged and yellowish with multiple 1–2-mm red to dark red spots. The uterus was dilated with a larger left uterine horn and prominent congested blood vessels; a corpus luteum cyst was present in the left ovary. Microscopic examination showed severe, acute, necrotizing pneumonia and interstitial subacute to chronic pneumonitis with arteritis (mostly associated with lungworms); multiple foci of acute coagulative necrosis in the liver; and mild, multifocal, nonsuppurative meningitis. The mammary gland contained numerous small acini with small amounts of milk in the acini and ducts. Infiltrate of mononuclear cells under the endometrium suggested endometritis.
Immunohistochemical investigation, using polyclonal antiserum obtained from a rabbit experimentally infected with B. melitensis, showed diffuse intracytoplasmic positive staining for Brucella spp., primarily in mononuclear and inflammatory cells on various tissues (spleen [Figure 2], lymph nodes, lung, uterus, liver, pancreas, and brain), in lesions, in lungworms, and in mammary gland acini and milk. By transmission electron microscopy, large numbers of relatively small (diameter 380–450 nm) intracellular coccoid bacteria that suggested Brucella spp. were observed in the genital ulcer. A Brucella sp. isolate was obtained from brain and lung tissue. The strain grew on Brucella agar supplemented with 5% horse serum in the presence of basic fuchsine, thionine, and growth on safranin O. CO2 was not required for growth, and H2S was not produced. The isolates showed catalase, oxidase, and urease activity. This biotype profile is in agreement with the strain type profile of B. ceti (10). Multilocus variable number tandem repeat analysis (MLVA) typing, which used MLVA panel 1 (8 minisatellite loci: bruce06, bruce08, bruce11, bruce12, bruce42, bruce43, bruce45, and bruce55, which are useful for species identification), showed that the strains belong to genotype 23 (11). MLVA panel 2 was split into 2 groups, panels 2A and 2B, comprising 3 (bruce18, bruce19, bruce21) and 5 (bruce04, bruce07, bruce09, bruce16, bruce30) markers, respectively (12). Using panel 2A, we obtained the same profile as the one described for all B. ceti strains isolated from porpoises (11), whereas panel 2B showed a new genotype (bruce04: 6 repeats, bruce07: 6 repeats, bruce09: 3 repeats, bruce16: 7 repeats, bruce30: 6 repeats), closely related to genotypes ascribed to B. ceti strains isolated from porpoises mainly stranded in Scotland (11). The new genotype identified by panel 2B is possibly associated with southern North Sea porpoises. However, panel 2B contains the more variable loci, and this panel has been given a lower weight in clustering analysis (12).
The results suggest a bacteremia associated with B. ceti. The infection was suspected after examination by electron microscopy and confirmed by bacteriologic and immunohistochemical investigations; finally, the bacterium was identified as B. ceti. In Europe, most reported cases of cetacean brucellosis have been reported from the coasts of Scotland and England and found in striped dolphins, Atlantic white-sided dolphins, common dolphins, harbor porpoises, and a minke whale (2,3,6,7,9). Meningoencephalitis associated with Brucella spp. infection has been reported for striped dolphins (2,3) and 1 Atlantic white-sided dolphin (7). Necrosis of spleen, liver, and lymph nodes associated with Brucella spp. infection has also been reported for Atlantic white-sided dolphins (6). In porpoises, Brucella spp. have been isolated from different organs without associated pathologic changes other than coagulative necrosis of the spleen (6) and a testicular abscess (9). Finally, in the minke whale, foci of liver necrosis and inflammation were consistent with lesions caused by Brucella spp (6). In our study, the enlarged uterine horn, the corpus luteum cyst, and the presence of milk in mammary acini suggested recent pregnancy, and the positive immunolabeling of the endometrium raised the question of a possible abortion. Indeed, Brucella spp. are known to be responsible for abortions in terrestrial mammals, Brucella spp.–induced abortions have been described in 2 bottlenose dolphins with associated placentitis (1), and Brucella spp. have been isolated from an aborted bottlenose dolphin fetus (13). Brucella antigens were detected in the placenta of a stranded striped dolphin with a 7-month-old dead fetus (5). In addition, vaginal lithiasis suspected to be the result of ossification of aborted fetuses in 2 common dolphins positive for Brucella spp. in the uterus has been reported (8).
In the present case, a final conclusion cannot be drawn with respect to a possible abortion. Identification of B. ceti in milk (as in the present study) and in fetal tissues and secretions of a pregnant dolphin suggest that B. ceti has tropism for placental and fetal tissues and that it can be shed externally (4). This finding suggests potential vertical and horizontal transmission to newborns (4). Nevertheless, indirect transmission through parasites should not be excluded because Brucella spp. have been identified from lungworms (14). In addition, the observation of Brucella spp. antigens in milk and in skin ulcers may represent routes of bacterial transmission between individual animals and raises the question of the risk for transmission to a person handling the cetacean (e.g., on the beach or in a rehabilitation center). All persons handling wild or captive marine mammals (alive or dead) or samples collected from the mammals should be aware of such risks and take necessary precautions. To date, 4 cases of human infection with Brucella spp. from marine mammals are known. One was mild and uncomplicated in a laboratory worker; however, the 3 other cases were severe naturally acquired without direct contact with marine mammals but with a history of eating raw fish or shellfish (15).
We emphasize that further investigations are needed to improve knowledge of the prevalence, the impact on individual cetaceans and populations, and the zoonotic potential of marine mammal brucellosis. The zoonotic risk should be taken into account by all persons in contact (direct or indirect) with marine mammals. Finally, the present case confirms the need for careful monitoring and complete postmortem examinations of stranded marine mammals.
Dr Jauniaux is a veterinary pathologist working as senior assistant in the Department of Veterinary Pathology, University of Liege. His research interest is the causes of death of marine mammals stranded on the coastline of Belgium and northern France.
Acknowledgments
We thank J.J. Letesson for providing the polyclonal antibody against B. melitensis.
The Marine Animals Research and Intervention Network is operated under a contract of the Royal Belgian Institute of Natural Sciences.
References
- Miller WG, Adams LG, Ficht TA, Cheville NF, Payeur JP, Harley DR, Brucella-induced abortions and infection in bottlenose dolphins (Tursiops truncatus). J Zoo Wildl Med. 1999;30:100–10.PubMedGoogle Scholar
- González L, Patterson IA, Reid RJ, Foster G, Barberan M, Blasco JM, Chronic meningoencephalitis associated with Brucella sp. infection in live-stranded striped dolphins (Stenella coeruleoalba). J Comp Pathol. 2002;126:147–52. DOIPubMedGoogle Scholar
- Davison NJ, Cranwell MP, Perrett LL, Dawson CE, Deaville R, Stubberfield EJ, Meningoencephalitis associated with Brucella species in a live-stranded striped dolphin (Stenella coeruleoalba) in south-west England. Vet Rec. 2009;165:86–9. DOIPubMedGoogle Scholar
- Hernández-Mora G, Gonzalez-Barrientos R, Morales JA, Chaves-Olarte E, Guzman-Verri C, Barquero-Calvo E, Neurobrucellosis in stranded dolphins, Costa Rica. Emerg Infect Dis. 2008;14:1430–3. DOIPubMedGoogle Scholar
- González-Barrientos R, Morales JA, Hernandez-Mora G, Barquero-Calvo E, Guzman-Verri C, Chaves-Olarte E, Pathology of striped dolphins (Stenella coeruleoalba) infected with Brucella ceti. J Comp Pathol. 2010;142:347–52. DOIPubMedGoogle Scholar
- Foster G, MacMillan AP, Godfroid J, Howie F, Ross HM, Cloeckaert A, A review of Brucella sp. infection of sea mammals with particular emphasis on isolates from Scotland. Vet Microbiol. 2002;90:563–80. DOIPubMedGoogle Scholar
- Dagleish MP, Barley J, Howie FE, Reid RJ, Herman J, Foster G. Isolation of Brucella species from a diseased atlanto-occipital joint of an Atlantic white-sided dolphin (Lagenorhynchus acutus). Vet Rec. 2007;160:876–8. DOIPubMedGoogle Scholar
- Bogomolni AL, Gast RJ, Ellis JC, Dennett M, Pugliares KR, Lentell BJ, Victims or vectors: a survey of marine vertebrate zoonoses from coastal waters of the northwest Atlantic. Dis Aquat Organ. 2008;81:13–38. DOIPubMedGoogle Scholar
- Dagleish MP, Barley J, Finlayson J, Reid RJ, Foster G. Brucella ceti associated pathology in the testicle of a harbour porpoise (Phocoena phocoena). J Comp Pathol. 2008;139:54–9. DOIPubMedGoogle Scholar
- Foster G, Osterman BS, Godfroid J, Jacques I, Cloeckaert A. Brucella ceti sp. nov. and Brucella pinnipedialis sp. nov. for Brucella strains with cetaceans and seals as their preferred hosts. Int J Syst Evol Microbiol. 2007;57:2688–93. DOIPubMedGoogle Scholar
- Maquart M, Le Fleche P, Foster G, Tryland M, Ramisse F, Djonne B, MLVA-16 typing of 295 marine mammal Brucella isolates from different animal and geographic origins identifies 7 major groups within Brucella ceti and Brucella pinnipedialis. BMC Microbiol. 2009;9:145. DOIPubMedGoogle Scholar
- Al Dahouk S, Le Fleche P, Nockler K, Jacques I, Grayon M, Scholz HC, Evaluation of Brucella MLVA typing for human brucellosis. J Microbiol Methods. 2007;69:137–45. DOIPubMedGoogle Scholar
- Ewalt DR, Payeur JB, Martin BM, Cummins DR, Miller WG. Characteristics of a Brucella species from a bottlenose dolphin (Tursiops truncatus). J Vet Diagn Invest. 1994;6:448–52. DOIPubMedGoogle Scholar
- Perrett LL, Dawson CE, Davison N, Quinney S. Brucella infection of lungworms from a harbour porpoise. Vet Rec. 2004;154:800.PubMedGoogle Scholar
- Whatmore AM, Dawson CE, Groussaud P, Koylass MS, King AC, Shankster SJ, Marine mammal Brucella genotype associated with zoonotic infection. Emerg Infect Dis. 2008;14:517–8. DOIPubMedGoogle Scholar
Figures
Table
Cite This ArticleTable of Contents – Volume 16, Number 12—December 2010
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Thierry P. Jauniaux, University of Liege–Veterinary Pathology, Sart Tilman B4, Liege 4000, Belgium
Top