Volume 16, Number 3—March 2010
Dispatch
Increasing Skin Infections and Staphylococcus aureus Complications in Children, England, 1997–2006
Cite This Article
Citation for Media
Abstract
During 1997–2006, general practitioner consultations for skin conditions for children <18 years of age in England increased 19%, from 128.5 to 152.9/1,000 child-years, and antistaphylococcal drug prescription rates increased 64%, from 17.8 to 29.1/1,000 child-years. During the same time period, hospital admissions for Staphylococcus aureus infections rose 49% from 53.4 to 79.3/100,000 child-years.
Staphylococcus aureus infection is a leading cause of staphylococcal bacteremia in adults (1) and children (2) in hospitals in the United Kingdom, and recent reports suggest invasive staphylococci are emerging from the community (3). Flucloxacillin is the antimicrobial drug recommended for treating S. aureus skin infection in UK primary care centers (4). Therefore, its use provides a proxy marker of S. aureus skin infection in children. Flucloxacillin prescribing in children has increased over the past 15 years (5), despite well-documented reductions in prescribing rates for other commonly prescribed antibacterial drugs during 1995–2000 (6), which suggests that S. aureus skin infections in the community may be increasing.
We examined the incidence of local complications of S. aureus disease in children over a 10-year period using nationally representative data from primary care clinicians in England. Ethics approval for this study was obtained from the Independent Scientific and Ethical Advisory Committee, application no. 2006/ISEAC/012.
The MediPlus UK database contains anonymized longitudinal data from >500 UK general practitioners who contribute clinical data on >1 million patients (7) that have been used widely for research (8). Consultations are coded by using the International Classification of Diseases, Tenth Revision (ICD-10), and antimicrobial drug prescriptions are coded by using the British National Formulary for children, Chapters 5.1.1–5.1.3 (9). Using Mediplus UK, we extracted data on all skin conditions (ICD-10 code) and atopic dermatitis (ICD-10 code L20) as an index condition in children <18 years of age who saw general practitioners in England from January 1, 1997, through December 31, 2006. We counted prescriptions for all oral and topical antibacterial drugs prescribed for skin infections, and used all oral preparations containing flucloxacillin prescribed for skin conditions as a proxy measure of unresolved S. aureus skin infection. We calculated age–sex adjusted annual consulting and prescribing rates by totaling the number of consultations or prescriptions and dividing by the number of person–years contributed by each child in the registered population for each calendar year. We then directly standardized these rates by using the age–sex distribution for the reference year 2000.
The Hospital Episode Statistics (HES) database has recorded all inpatient hospital activity in National Health Service hospitals across England since 1989 and is used widely to monitor disease trends in England (www.hesonline.nhs.uk) (10). The main reason for admission, i.e., primary diagnosis, is recorded by using ICD-10 codes. We used HES data to calculate age–sex adjusted admission rates per 100,000 resident population for children <18 years of age for each calendar year from January 1, 1997, through December 31, 2006, for conditions commonly caused by S. aureus, including septic arthritis (ICD-10 codes M00.0 for staphylococcal arthritis and M00.9 for pyogenic arthritis), osteomyelitis (M86), and locally invasive skin infections (L02, cutaneous abscesses and boils; L03, cellulitis). Rates were calculated as the total number of admissions per year divided by the mid-year estimate of the number of children residing in England (using the 2000 population in England as the reference population) (11). Confidence intervals (CIs) were generated with a Poisson approximation. We used linear regression to test for linear trends in age–sex adjusted admission rates across the period. We used Stata version 9 software (Stata Corp., College Park, TX, USA) for all statistical analysis.
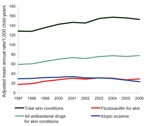
The Mediplus database contained 2,821,372 child-years of follow-up during 1997–2006. General practitioner consultation rates for all skin conditions in children rose from 128.5 (95% CI 127.2– 129.8) per 1,000 child-years to 152.9 (95% CI 151.4–154.5) per 1,000 child years (p = 0.011). Atopic eczema consultation rates decreased during this time (Figure 1).
In parallel with the rising number of skin consultations was a 64% increase in prescribing rates for antistaphylococcal drugs (flucloxacillin), from 17.8 (95% CI 17.3–18.3) to 29.1 (95% CI 28.5–29.8) prescriptions per 1,000 child-years (p<0.001) (Figure 2). Prescribing of all other antibacterial drugs for children for any reason decreased from 541.4 (95% CI 538.8–544.1) per 1,000 child-years to 484.3 (95% CI 481.6–487.0) per 1,000 child-years (Table 1). Flucloxacillin was the most commonly prescribed antibacterial drug for all skin conditions (37%). Prescribing rates for other classes of antibacterial drugs used for skin infections, notably, combined preparations of amoxicillin and clavulanic acid 2% and fusidic acid (<2%), were stable over the time period (Figure 2).
During 1997–2006, unplanned hospital admission rates for skin, bone, and joint infections in all children increased by 49% from 53.4 (95% CI 52.1–54.7) to 79.3 (95% CI 77.7–80.9) per 100,000 child-years (p<0.001) including cellulitis (67.8% increase; p<0.001), skin abscesses (36.7% increase; p <0.001), and osteomyelitis (46.1% increase; p = 0.004) (Table 2). This trend was consistent across all age groups. Admission rates for septic arthritis increased but the result of the test for trend was not significant (p = 0.128).
The increasing incidence of childhood skin infections and prescribing of the major antistaphylococcal drug flucloxacillin seen in UK primary care, coupled with concurrent increases in childhood hospital admissions for skin bone and joint infections caused by S. aureus in hospitals in England, suggests an increase in community-onset S. aureus disease in England over the past 10 years. The time frame, a large nationally representative population, use of prospectively collected data, and consistency of patterns make it unlikely our findings arose by chance. Because HES data records primary diagnosis when patients are admitted, most infections will be community-, not hospital-, acquired. Flucloxacillin has remained the treatment of choice for S. aureus skin infections in UK primary care for decades (9), and its increased use is not explained by treatment drift from other groups of antibacterial drugs used to treat skin infections or by increasing antibacterial drug treatment of atopic eczema colonized with staphylococci. Total unplanned admission rates in children <19 years of age have increased by 13% during 1997–2006 (12), but these increases are modest compared with the 49% increase in S. aureus complications seen over the same time frame in our study.
Limitations of our study include the use of clinically coded proxy measures for S. aureus infections that are subject to recording bias. Because of the lack of microbiologic surveillance, we could not differentiate whether increases in S. aureus disease were caused by methicillin-sensitive S. aureus or methicillin-resistant S. aureus. Antimicrobial drugs for skin infections are available only by prescription in the UK but not exclusively from GPs. Thus, our prescribing data excluded prescriptions issued from other healthcare settings. Our findings that admission rates for osteomyelitis, boils, and cellulitis increased but septic arthritis rates were stable might be because septic arthritis is also caused by pneumococci, β-hemolytic streptococci, and gram negative organisms (13).
A growing body of evidence supports our findings of increases in community-onset S. aureus disease in children. Hospitalizations for S. aureus disease in all age groups are increasing; in several countries severe skin infections, particularly among children, are rising, caused by strains of S. aureus producing the Panton-Valentine leukocidin (14,15). Although currently no formal surveillance of this strain in the UK is available, referrals of isolates of S. aureus positive for Panton-Valentine leukocidin to the national Staphylococcal Reference Unit increased each year from 224 in 2005 to 1,361 2007. What is not known is whether S. aureus community-acquired infections in children have added to the recently reported increases of S. aureus infection and bacteremias acquired in hospital settings (4). Further work is required to monitor S. aureus disease and antimicrobial drug resistance and to identify community risk factors for S. aureus disease in children.
Dr Saxena is a National Health Service general practitioner in London and senior clinical lecturer in the Department of Primary Care and Social Medicine at Imperial College London. Her research interests are in the epidemiology and treatment of childhood illness in primary care.
Acknowledgments
We thank International Marketing Systems Health for providing access to the Mediplus UK data and all the members of the improving Children’s Antibiotic Prescribing group.
S.S. has a postdoctoral award from the National Institute for Health Research (NIHR). I.W.’s post was funded by a Department of Health Public Health Career Scientist Award. N.S.’s post is funded by the European Society for Pediatric Infectious Diseases. We are grateful for support from the NIHR Biomedical Research Centre funding scheme.
References
- Hayward AB, Knott F, Petersen I, Livermore DM, Duckworth G, Islam A, Increasing hospitalizations and general practice prescriptions for community-onset staphylococcal disease, England. Emerg Infect Dis. 2008;14:720–6. DOIPubMedGoogle Scholar
- Sharland M, Paediatric Subgroup SACAR. The use of antibacterials in children: a report of the Specialist Advisory Committee on Antimicrobial Resistance (SACAR) Paediatric Subgroup. J Antimicrob Chemother. 2007;60:i15–26. DOIPubMedGoogle Scholar
- Robinson DA, Kearns AM, Holmes A, Morrison D, Grundmann H, Edwards G, Re-emergence of early pandemic Staphylococcus aureus as a community-acquired methicillin-resistant clone. Lancet. 2005;365:1256–8. DOIPubMedGoogle Scholar
- Rayner C, Munckhof WJ. Antibiotics currently used in the treatment of infections caused by Staphylococcus aureus. Intern Med J. 2005;35(Suppl 2):S3–16. DOIPubMedGoogle Scholar
- Thompson PL, Spyridis N, Sharland M, Changes in clinical indications for community antibiotic prescribing for children in the UK from 1996–2006: will the new NICE prescribing guidance on upper respiratory tract infections be ignored? Arch Dis Child. Epub 2008 Dec 9.
- Ashworth M, Cox K, Latinovic R, Charlton J, Gulliford M, Rowlands G. Why has antibiotic prescribing for respiratory illness declined in primary care? A longitudinal study using the General Practice Research Database. J Public Health (Oxf). 2004;26:268–74. DOIPubMedGoogle Scholar
- World Health Organization. International Classification of Diseases and Related Health Problems; Tenth revision. 2[2]. 2007. Geneva: The Organization. 2007.
- Wong IC, Murray ML. The potential of UK clinical databases in enhancing paediatric medication research. Br J Clin Pharmacol. 2005;59:750–5. DOIPubMedGoogle Scholar
- Royal College of Paediatrics and Child Health (RCPCH), Neonatal and Paediatric Pharmacists Group (NPPG), British Medical Association (BMA), Royal Pharmaceutical Society of Great Britain (RPSGB). BNF for Children, 3rd ed. London: Pharmaceutical Press; 2009.
- Department of Health. Hospital Episode Statistics: The Book. London: Office for National Statistics; 1998.
- Census Office for National Statistics. 2001. London: Her Majesty’s Stationery Office; 2001.
- Chief Nursing Officer's Directorate. Children Families and Maternity Analysis. Trends in children and young people’s care: Emergency admission statistics, 1996/97–2006/07, England. London: The Stationery Office; 2008.
- Gutierrez AM. Infectious and inflammatory arthritis. In: Principles and practice of paediatric infectious diseases. Philadelphia: Churchill Livingstone; 2003. p. 480–5.
- McCaskill ML, Mason EO Jr, Kaplan SL, Hammerman W, Lamberth LB, Hulten KG. Increase of the USA300 clone among community-acquired methicillin-susceptible Staphylococcus aureus causing invasive infections. Pediatr Infect Dis J. 2007;26:1122–7. DOIPubMedGoogle Scholar
- Chung HJ, Jeon HS, Sung H, Kim MN, Hong SJ. Epidemiological characteristics of methicillin-resistant Staphylococcus aureus isolates from children with eczematous atopic dermatitis lesions. J Clin Microbiol. 2008;46:991–5. DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 16, Number 3—March 2010
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Sonia Saxena, Rm 332 Reynolds Bldg, Imperial College London, Charing Cross Campus, St Dunstan’s Rd, London W6 8RF, UK
Top