Volume 16, Number 9—September 2010
Dispatch
Novel Hepatitis E Virus Genotype in Norway Rats, Germany
Figure 1
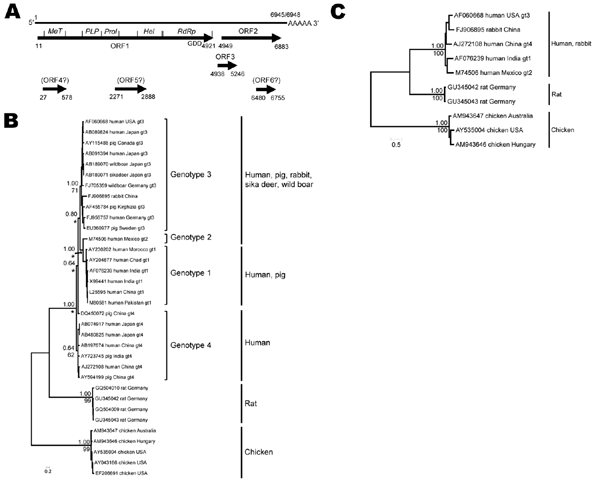
Figure 1. Genome structure and localization of putative open reading frames (ORFs) and functional domains in ORF1 of hepatitis E virus (HEV) sequences from Norway rats nos. 63 and 68, collected in Germany, July 2009 (A); phylogenetic trees based on a partial nucleotide sequence of 1,576 nt (B); and the complete genomes (C). RNA was isolated from liver samples by using the RNeasy Mini Kit and a QIAshredder (QIAGEN, Hilden, Germany). The entire rat HEV genome sequences of each rat were determined by a primer walking strategy and rapid amplification of cDNA ends protocols (GenBank accession nos. GU345042 and GU345043). ORFs were predicted by using the SeqBuilder Module of the DNASTAR software package (Lasergene, Madison, WI, USA). Putative functional domains in ORF1 were compared with those predicted in the corresponding regions of ORF1 from HEV genotypes 1–4 (11). The methyltransferase (MeT), helicase (Hel), and RNA-dependent RNA polymerase (RdRp, GDD motif indicated) domains are conserved and in the same order in the rat HEV genomes. In contrast, the papain-like protease domain (PLP) and the proline-rich domain (Prol) were more variable. Three additional ORFs (4, 5, 6) were predicted for both rat HEV genomes, for which no similar amino acid sequence could be found by BLASTp (http://blast.ncbi.nlm.nih.gov/Blast.cgi) search, sequence profile search in Pfam, and no functional pattern by Prosite (www.expasy.ch/prosite/) search; however, several similar sequences were retrieved from the Uniprot collection by comparison of translated nucleotide sequences with BLASTx (http://blast.ncbi.nlm.nih.gov/Blast.cgi). In addition, these ORFs showed less difference to the host codon usage than ORF3 as determined by Graphical Codon Usage Analyzer (http://gcua.schoedl.de/) and STRAP (http://3d-alignment.eu/). Phylogenetic relationships were reconstructed by using neighbor-joining and Bayesian algorithms after substitution model estimation (12). Robustness of nodes in phylogenetic trees is given above branches for Bayesian algorithms (sampling every 10 of 1 million generations; first 25,000 samples discarded as burn-in) and below branches for neighbor joining (10,000 bootstrap replicates). Only support values for main nodes that connect genotypes or major evolutionary lineages are displayed. *Indicates that neighbor-joining algorithms suggest instead a closer phylogenetic relationship between genotypes 3 and 4 with genotype 1 basal to these 2. Scale bar indicates phylogenetic distances in nucleotide substitutions per site.
References
- Meng XJ. Hepatitis E virus: animal reservoirs and zoonotic risk. Vet Microbiol. 2009;140:256–65. DOIPubMedGoogle Scholar
- Teshale EH, Howard CM, Grytdal SP, Handzel TR, Barry V, Kamili S, Hepatitis E epidemic, Uganda. Emerg Infect Dis. 2010;16:126–9. DOIPubMedGoogle Scholar
- Lewis HC, Wichmann O, Duizer E. Transmission routes and risk factors for autochthonous hepatitis E virus infection in Europe: a systematic review. Epidemiol Infect. 2010;138:145–66. DOIPubMedGoogle Scholar
- Schielke A, Sachs K, Lierz M, Appel B, Jansen A, Johne R. Detection of hepatitis E virus in wild boars of rural and urban regions in Germany and whole genome characterization of an endemic strain. Virol J. 2009;6:58. DOIPubMedGoogle Scholar
- Kabrane-Lazizi Y, Fine JB, Elm J, Glass GE, Higa H, Diwan A, Evidence for widespread infection of wild rats with hepatitis E virus in the United States. Am J Trop Med Hyg. 1999;61:331–5. PubMedGoogle Scholar
- Favorov MO, Kosoy MY, Tsarev SA, Childs JE, Margolis HS. Prevalence of antibody to hepatitis E virus among rodents in the United States. J Infect Dis. 2000;181:449–55. DOIPubMedGoogle Scholar
- Arankalle VA, Joshi MV, Kulkarni AM, Gandhe SS, Chobe LP, Rautmare SS, Prevalence of anti–hepatitis E virus antibodies in different Indian animal species. J Viral Hepat. 2001;8:223–7. DOIPubMedGoogle Scholar
- Easterbrook JD, Kaplan JB, Vanasco NB, Reeves WK, Purcell RH, Kosoy MY, A survey of zoonotic pathogens carried by Norway rats in Baltimore, Maryland, USA. Epidemiol Infect. 2007;15:1–8.PubMedGoogle Scholar
- Ulrich RG, Schmidt-Chanasit J, Schlegel M, Jacob J, Pelz H-J, Mertens M, Network “Rodent-borne pathogens“ in Germany: longitudinal studies on the geographical distribution and prevalence of hantavirus infections. Parasitol Res. 2008;103(Suppl. 1):S121–9. DOIPubMedGoogle Scholar
- Johne R, Plenge-Bönig A, Hess M, Ulrich RG, Reetz J, Schielke A. Detection of a novel hepatitis E–like virus in feces of wild rats using a nested broad-spectrum RT-PCR. J Gen Virol. 2010;91:750–8. DOIPubMedGoogle Scholar
- Koonin EV, Gorbalenya AE, Purdy MA, Rozanov MN, Reyes GR, Bradley DW. Computer-assisted assignment of functional domains in the nonstructural polyprotein of hepatitis E virus: delineation of an additional group of positive-strand RNA plant and animal viruses. Proc Natl Acad Sci U S A. 1992;89:8259–63. Medline DOIPubMedGoogle Scholar
- Braaker S, Heckel G. Transalpine colonisation and partial phylogeographic erosion by dispersal in the common vole Microtus arvalis. Mol Ecol. 2009;18:2518–31. DOIPubMedGoogle Scholar
- Zhao C, Ma Z, Harrison TJ, Feng R, Zhang C, Qiao Z, A novel genotype of hepatitis E virus prevalent among farmed rabbits in China. J Med Virol. 2009;81:1371–9. DOIPubMedGoogle Scholar
- Emerson SU, Anderson D, Arankalle A, Meng X-J, Purdy M, Schlauder GG, Hepevirus. In: CM Fauquet, MA Mayo, J Maniloff, U Desselberger, LA Ball, editors. Virus taxonomy. San Diego: Elsevier Academic Press; 2005. p. 853–7.
- Lee YH, Ha Y, Ahn KK, Chae C. Localisation of swine hepatitis E virus in experimentally infected pigs. Vet J. 2009;179:417–21. DOIPubMedGoogle Scholar