Volume 17, Number 11—November 2011
Dispatch
Clonal Origins of Vibrio cholerae O1 El Tor Strains, Papua New Guinea, 2009–2011
Cite This Article
Citation for Media
Abstract
We used multilocus sequence typing and variable number tandem repeat analysis to determine the clonal origins of Vibrio cholerae O1 El Tor strains from an outbreak of cholera that began in 2009 in Papua New Guinea. The epidemic is ongoing, and transmission risk is elevated within the Pacific region.
In July 2009, an outbreak of cholera began in the Morobe Province of Papua New Guinea (PNG) (1), and in the following months the disease spread throughout the coastal regions of the country. Although environmental and social conditions are conducive to the transmission and sustained presence of cholera, to our knowledge, this was the first outbreak of cholera in PNG. Sporadic outbreaks have occurred in the nearby Indonesian province of West Papua (formerly Irian Jaya) in the 1960s, 1990s, and, most recently, in 2008 (2,3). As such, conjecture has existed about whether this outbreak was the result of a new incursion of Vibrio cholerae or a reemergence of previously undetected strains endemic to PNG. We used multilocus sequence typing (MLST) and variable number tandem repeat (VNTR) analysis to investigate the diversity of the PNG V. cholerae strains and to elucidate the origin of this outbreak.
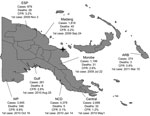
The PNG cholera outbreak was first reported in Lambutina and Nambariwa villages in Morobe Province on the northeast coast of mainland PNG in July 2009 (1). The outbreak spread within the province and then to Madang and East Sepik Provinces along the northwest coast. In January 2010, the epidemic reached the national capital, Port Moresby, resulting in a large sustained outbreak in National Capital District and surrounding Central Province. In the following months, the outbreak spread along the south coast to Gulf and Western Provinces. Recently, the cholera outbreak has spread to the Autonomous Region of Bougainville. Since July 2009, >15,500 cases of cholera have been reported in PNG, with 493 recorded deaths (Figure).
Clinical isolates from Morobe Province (n = 2), Madang Province (n = 2), Eastern Highlands Province (n = 2), East Sepik Province (n = 2), and National Capital District (n = 4) were isolated and identified as V. cholerae O1 El Tor Ogawa by using standard bacterial culture methods. The isolates were confirmed as V. cholerae serogroup O1 and carriers of the ctxA gene by multiplex PCR (4). All isolates were identified as altered El Tor through PCR detection of the classical rstR gene (5).
Nine loci were targeted for MLST analysis: dnaE, lap, recA, pgm, gyrB, cat, chi, rstA, and gmd, as described (6). The PCR products were visualized on an agarose gel, and direct sequencing was performed in both directions by using the MLST primers (Macrogen, Seoul, Korea). Contiguous nucleotide sequences were assembled by using Sequencher software (http://www.genecodes.com), and all nucleotide positions were confirmed by >2 independent sequencing reactions in each direction. The PNG MLST sequences were compared with previously reported sequences by using the same 9 loci outlined in previous studies (5–7). All PNG isolates displayed 100% nt identity across the 9 MLST loci and were identical to the Bangladesh strain MJ-1236 (8).
Five loci were analyzed for tandem repeats by using VNTR-specific primers, as described (9). The targeted regions were VC0147, VC0436–7, VC1650, VCA0171, and VCA0283. Contiguous nucleotide sequences were prepared as described above. Sequence types were designated by the actual number of repeats at the target loci as described in recent studies (10,11) and compared with sequence-derived VNTR data from the international literature and databases. Three sequence types were detected that were all within clonal complex 10,6,8,X,X (Table). The isolates from PNG were most closely related to strains from Vietnam (1995–2004) in the MLVA group III reported by Choi et al. (12). As reported, loci on the small chromosome (VCA0171 and VCA0283) were more variable than the loci on the large chromosome (9,11,12).
The homogeneity of outbreak strains in PNG and the relatedness to strains from Vietnam by VNTR analysis is indicative of a recent incursion from the Southeast Asia region. It is unlikely that this outbreak is because of a previously undetected autochthonous endemic strain, given the close relationship with the isolates from Vietnam.
MLST and VNTR have been used by numerous research groups to analyze the diversity of outbreak strains and to investigate the origin of epidemics (6,9–11). MLST analysis of the V. cholerae strains and comparison with previously published MLST data suggested that the most closely related isolate was from Bangladesh (8). All isolates from PNG were identical to the strain MJ-1236 across the 9 housekeeping genes examined by sequence analysis. In contrast, the VNTR sequence analysis suggested that the PNG outbreak strains were most closely related to strains isolated from Vietnam in 1995, 2002, 2003, and 2004 (12). MLST data were not available for the Vietnam strains in the international literature or databases; therefore, a direct comparison cannot be made between the PNG MLST and VNTR results.
This outbreak highlights the continued challenge that cholera presents to authorities worldwide: the disease can spread rapidly and the causative organism persists in the environment (13), which makes prevention and control of the disease complex. In PNG the large estuarine waterways (e.g., Sepik and Fly Rivers) and the settlement areas (which are often in estuarine areas with limited water and sanitary infrastructure and are more densely populated than rural and urban areas) present potential reservoirs for V. cholerae. The prevalence of enteric diseases remains high in PNG where access to safe drinking water is limited, particularly in rural areas where an estimated 87% of the population lives (14). These factors may aid the persistence of V. cholerae and result in a transition to endemicity of cholera in PNG.
During this outbreak, a relatively high national case-fatality ratio (CFR) of 3.2% was recorded. The provincial CFRs varied widely from 0.1% in the National Capital District, where oral rehydration salts points and treatment centers provided timely accessible treatment, to 8.8% in Western Province, where health system access and preparedness were weak. Strong leadership and coordination contributed to effective response but were limited where CFRs were high.
Despite road networks linking affected coastal areas to the mountainous interior, where most of the country’s population resides, imported cases have not resulted in ongoing transmission. This lack of transmission may be related to a less favorable habitat for environmental persistence of the organism and ongoing transmission. A similar situation was reported from the current outbreak of cholera in Haiti, where location on a coastal plain was a notable risk factor for cholera cases (15). Nonetheless, cholera remains a high risk for both affected and unaffected provinces in PNG. Moreover, the frequent international migration between PNG and neighboring communities with no prior cholera exposure and with vulnerable sanitary conditions heightens the risk for international spread.
Although the MLST and VNTR results concur that the PNG strains are closely related, our data suggest that VNTR has greater discriminatory power when used for investigations into the clonality and relatedness of V. cholerae strains. Other studies have highlighted the value of VNTR for strain typing of V. cholerae (9–12), but reports that directly compare VNTR and MLST are lacking. However, analysis by either VNTR or MLST is hampered by the paucity of data available to compare outbreak strains from around the world. An online V. cholerae VNTR database would enable more accurate tracking of the evolution of outbreaks and provide evidence for the mode of spread of V. cholerae strains between countries and geographic areas. A more comprehensive analysis of V. cholerae strains from around the world is also required to gain a better understanding of the global and regional spread of these strains.
Dr Horwood is a senior research fellow at the Papua New Guinea Institute of Medical Research. His research interests include molecular characterization and epidemiology of emerging tropical infectious diseases.
Acknowledgment
This study was funded by the World Health Organization. The authors acknowledge the staff at Port Moresby General Hospital Pathology Laboratory for providing V. cholerae isolates.
References
- Rosewell A, Dagina R, Murhekar M, Ropa B, Posanai E, Dutta SR, Vibrio cholerae O1 in 2 coastal villages, Papua New Guinea. Emerg Infect Dis. 2011;17:154–6. DOIPubMedGoogle Scholar
- Cholera—Indonesia (West Papua). ProMed. 2008 Jul 29. http://www.promedmail.org, archive no. 20080730.2334.
- Korthuis PT, Jones TR, Lesmana M, Clark SM, Okoseray M, Ingkokusumo G, An outbreak of El Tor cholera associated with a tribal funeral in Irian Jaya, Indonesia. Southeast Asian J Trop Med Public Health. 1998;29:550–4.PubMedGoogle Scholar
- Hoshino K, Yamasaki S, Mukhopadhyay AK, Chakraborty S, Basu A, Battacharya SK, Development and evaluation of a multiplex PCR assay for rapid detection of toxigenic Vibrio cholerae O1 and O139. FEMS Immunol Med Microbiol. 1998;20:201–7. DOIPubMedGoogle Scholar
- Lee JH, Han KH, Choi SY, Lucas MES, Mondlane C, Ansaruzzaman M, Multilocus sequence typing (MLST) analysis of Vibrio cholerae O1 El Tor isolates from Mozambique that harbour the classical CTX prophage. J Med Microbiol. 2006;55:165–70. DOIPubMedGoogle Scholar
- Garg P, Aydanian A, Smith D, Morris JG, Nair GB, Stine OC. Molecular epidemiology of O139 Vibrio cholerae: mutation, lateral gene transfer, and founder flush. Emerg Infect Dis. 2003;9:810–4.PubMedGoogle Scholar
- González-Fraga S, Pichel M, Binsztein N, Johnson JA, Morris JG, Stine OC. Lateral gene transfer of O1 serogroup encoding genes of Vibrio cholerae. FEMS Microbiol Lett. 2008;286:32–8. DOIPubMedGoogle Scholar
- Grim CJ, Hasan NA, Taviani E, Haley B, Chun J, Brettin TS, Genome sequence of hybrid Vibrio cholerae O1 MJ-1236, B-33, and CIRS101 and comparative genomics with V. cholerae. J Bacteriol. 2010;192:3524–33. DOIPubMedGoogle Scholar
- Ghosh R, Nair GB, Tang L, Morris JG, Sharma NC, Ballal M, Epidemiological study of Vibrio cholerae using variable number of tandem repeats. FEMS Microbiol Lett. 2008;288:196–201. DOIPubMedGoogle Scholar
- Kendall EA, Chowdhury F, Begum Y, Khan AI, Li S, Theirer JH, Relatedness of Vibrio cholerae O1/O139 isolates from patients and their household contacts, determined by multilocus variable-number tandem-repeat analysis. J Bacteriol. 2010;192:4367–76. DOIPubMedGoogle Scholar
- Ali A, Chen Y, Johnson JA, Redden E, Mayette Y, Rashid MH, Recent clonal origin of cholera in Haiti. Emerg Infect Dis. 2011;17:699–701.PubMedGoogle Scholar
- Choi SY, Lee JH, Jeon YS, Lee HR, Kim EJ, Ansaruzzaman M, Multilocus variable-number tandem repeat analysis of Vibrio cholerae O1 El Tor strains harbouring classical toxin B. J Med Microbiol. 2010;59:763–9. DOIPubMedGoogle Scholar
- Nelson EJ, Harris JB, Morris JG, Calderwood SB, Camilli A. Cholera transmission: the host, pathogen and bacteriophage dynamic. Nat Rev Microbiol. 2009;7:693–702. DOIPubMedGoogle Scholar
- Papua New Guinea Ministry of Health. National Health Plan 2001–2010. Health Vision 2010. Policy Directions and Priorities. Vol. 1. Port Moresby (Papua New Guinea): The Ministry; 2000.
- Piarroux R, Barrais R, Faucher B, Haus R, Piarroux M, Gaudart J, Understanding the cholera epidemic, Haiti. Emerg Infect Dis. 2011;17:1161–8. DOIPubMedGoogle Scholar
Figure
Table
Cite This ArticleTable of Contents – Volume 17, Number 11—November 2011
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Paul F. Horwood, Papua New Guinea Institute of Medical Research, PO Box 60, Goroka, EHP441 Papua New Guinea
Top