Volume 17, Number 7—July 2011
Letter
Endemic Angiostrongyliasis, Rio de Janeiro, Brazil
Cite This Article
Citation for Media
To the Editor: The nematode Angiostrongylus cantonensis (rat lung worm), a zoonotic parasite that can accidentally infect humans and cause eosinophilic meningoencephalitis, has the Norway rat (Rattus norvegicus) as one of its most frequent definitive vertebrate hosts (1). Adult worms live in the pulmonary arteries of the definitive hosts, which excrete first-stage larvae in their feces. Intermediate hosts, such as snails and slugs, are infected by first-stage larvae, which reach the infective third stage after 2 molts. Third-stage larvae are then ingested by rats as they feed on the intermediate hosts, thus closing the life cycle. Humans become infected by eating raw or undercooked snails and slugs and through paratemic hosts and vegetables contaminated with infected snail mucus (2).
In Brazil, the first 3 documented cases of eosinophilic meningoencephalitis occurred in 2007 in 2 cities in the southeastern state of Espírito Santo (3). In 2009, a new case was reported in Pernambuco in the northeast region (4). Only intermediate hosts have been found naturally infected with rat lung worm in Brazil. Infected terrestrial and freshwater snails of the species Achatina fulica, Sarasinula marginata, Subulina octona, and Bradybaena similaris in Espírito Santo; A. fulica and Pomacea lineata in Pernambuco; and A. fulica in Rio de Janeiro and Santa Catarina have been reported (3,5,6). Thus, because of the recent cases of eosinophilic meningoencephalitis in Brazil and the occurrence of naturally infected A. fulica snails in Rio de Janeiro, we investigated the existence of potential natural reservoirs for the parasite in São Gonçalo.
São Gonçalo (22°48′26.7′′S, 43°00′49.1′′W) is a densely populated city (≈1 million inhabitants) with a tropical Atlantic climate (14°C–35°C) that is part of the metropolitan region of Rio de Janeiro. Two collections were made in March and June 2010. Forty live traps (20 Tomahawk [Tomahawk Live Trap Company, Tomahawk, WI, USA] and 20 Sherman [H.B. Sherman Traps Inc., Tallahassee, FL, USA] traps) were placed along two 30-m transects for 4 consecutive nights (Brazilian Institute of Environment and Renewable Natural Resources license no. 2227–1/2010) in an urban area where A. fulica snails had been collected in high numbers. Twenty-seven Norway rats (16 males) were captured. We collected 265 adult lung worms from the pulmonary arteries of the captured animals, fixed the worms in 70% ethanol, and taxonomically identified them as A. cantonensis on the basis of the large size of the spicules and the patterns of the bursal rays (7). Voucher specimens have been deposited in the Helminthological Collection of the Oswaldo Cruz Institute (no. 35712). Nineteen (74%) rats were infected; mean intensity and mean abundance were 13.52 ± 2.36 and 9.81 ± 1.96, respectively.
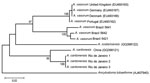
To confirm the morphologic identification of the Angiostrongylus specimens obtained, a DNA bar coding approach was used. DNA was extracted from 3 ethanol-preserved adult worms previously recovered from the pulmonary arteries of a naturally infected Norway rat, PCR-amplified, sequenced for a partial region of the COI gene (8), and subsequently compared with available GenBank Angiostrongylus spp. sequences. The three 360-bp COI sequences obtained (GenBank accession no. HQ440217) were Clustal-aligned (www.clustal.org) with homologous COI fragments of A. cantonensis (GenBank accession no. GQ398121), A. vasorum (GenBank accession nos. EU493162, EU493163, EU493166, EU493167), and A. costaricensis (GenBank accession no. GQ398122) and subjected to phylogenetic analysis. Ancylostoma tubaeforme (GenBank accession no. AJ407940) was used as the outgroup. Haplotypes for A. vasorum isolates from Brazil (A. vasorum 5421, 5641, and 5642) were reconstructed from published information (9) and included in the alignment. We used MEGA4 (www.megasoftware.net) to construct a neighbor-joining phylogenetic tree based on Kimura 2-parameter (K2-p) distances (Figure). The 3 A. cantonensis specimens from São Gonçalo, Rio de Janeiro, yielded a single haplotype, which formed a clade with the A. cantonensis haplotype from the People’s Republic of China with low genetic distance (K2-p 0.038) and high bootstrap support (98), thus confirming the morphologic identification. Comparisons with the other 2 Angiostrongylus species yielded higher genetic distance values (K2-p 0.120, with A. vasorum, and 0.149, with A. costaricensis).
These results indicate that A. cantonensis lung worm infection is enzootic among the exotic Norway rat population in the region studied. The natural infection rate of 74% is the second highest reported among 14 severely A. cantonensis infection–endemic regions (2). These findings, together with the observation of dense populations of A. fulica snails in urban areas of the country (10), call attention to the risk for disease transmission to humans, given that Norway rats also are likely to be present in these areas.
Local residents should be informed about disease transmission and prevention, and physicians should consider A. cantonensis lung worm infection in the differential diagnosis when appropriate. Although public health authorities should consider implementation of surveillance and control strategies to reduce the populations of snail and rat hosts, a better understanding is needed of the epidemiologic significance of these findings, which can be attained through studies to identify human cases of eosinophilic meningitis in the region.
Acknowledgments
We thank the anonymous reviewers whose comments and suggestions helped improve an earlier version of the manuscript.
This study was supported by the Brazilian Research Council.
References
- Lindo JF, Waugh C, Hall J, Canningham-Myrie C, Ashley D, Eberhard ML, Enzootic Angiostrongylus cantonensis in rats and snails after an outbreak of an human eosinophlic meningitis, Jamaica. Emerg Infect Dis. 2002;8:324–6. DOIPubMedGoogle Scholar
- Wang QP, Lai DH, Zhu XQ, Chen XG, Lun ZR. Human angiostrongyliasis. Lancet Infect Dis. 2008;8:621–30. DOIPubMedGoogle Scholar
- Caldeira RL, Mendonça CLF, Gouveia CO, Lenzi HL, Graeff-Teixeira C, Lima WS, First record of molluscs naturally infected with Angiostrongylus cantonensis (Chen, 1935) (Nematoda: Metastrongylidae) in Brazil. Mem Inst Oswaldo Cruz. 2007;102:887–9. DOIPubMedGoogle Scholar
- Lima AR, Mesquita SD, Santos SS, Aquino ER, Rosa LRS, Duarte FS, Alicata disease: neuroinfestation by Angiostrongylus cantonensis in Recife, Pernambuco, Brazil. Arq Neuropsiquiatr. 2009;67:1093–6. DOIPubMedGoogle Scholar
- Thiengo SC, Maldonado A, Mota EM, Torres EJL, Caldeira R, Oliveira APM, The giant African snail Achatina fulica as natural intermediate host of Angiostrongylus cantonensis in Pernambuco, northeast Brazil. Acta Trop. 2010;115:194–9. DOIPubMedGoogle Scholar
- Maldonado A Jr, Simões RO, Oliveira APM, Motta EM, Fernandez MA, Pereira ZM, First report of Angiostrongylus cantonensis (Nematoda: Metastrongylidae) in Achatina fulica (Mollusca: Gastropoda) from southeast and south regions of Brazil. Mem Inst Oswaldo Cruz. 2010;105:938–41.PubMedGoogle Scholar
- Chen HT. Un nouveau nématode pulmonaire, Pulmonema cantonensis n. g., n. sp., des rats de Canton. Ann Parasitol Hum Comp. 1935;13:312–7.
- Bowles J, Blair D, McManus DP. Genetic variants within the genus Echinococcus identified by mitochondrial DNA sequencing. Mol Biochem Parasitol. 1992;54:165–73. DOIPubMedGoogle Scholar
- Jefferies R, Shaw SE, Viney ME, Morgan ER. Angiostrongylus vasorum from South America and Europe represent distinct lineages. Parasitology. 2009;136:107–15. DOIPubMedGoogle Scholar
- Thiengo SC, Faraco FA, Salgado NC, Cowie R, Fernandez MA. Rapid spread of an invasive snail in South America: the giant African snail, Achatina fulica in Brazil. Biol Invasions. 2007;9:693–702. DOIGoogle Scholar
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 17, Number 7—July 2011
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Arnaldo Maldonado Jr, Laboratório de Biologia e Parasitologia de Mamíferos Silvestres Reservatórios, Instituto Oswaldo Cruz/Fiocruz, Av. Brasil 4365 Manguinhos 21.040-900, Rio de Janeiro, RJ, Brazil
Top