Volume 20, Number 4—April 2014
Letter
Detection of Rickettsia sibirica mongolitimonae by Using Cutaneous Swab Samples and Quantitative PCR
Cite This Article
Citation for Media
To the Editor: Tick-borne rickettsioses are caused by the obligate intracellular bacteria spotted fever group (SFG) Rickettsia spp. These zoonoses are now recognized as emerging or reemerging human infections worldwide, with ≈15 new tick-borne rickettsial species or subspecies recognized as human pathogens during the 30 past years (1). New approaches have emerged in recent years to definitively identify the causative agents, including emerging pathogens. Using cutaneous swab specimens from patients for quantitative PCR (qPCR) testing rather than cutaneous biopsy specimens is a major innovation in the diagnosis of SFG rickettsioses (2–4). Using this approach, we report 1 of the few documented infections caused by Rickettsia sibirica mongolitimonae.
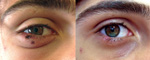
A 16-year-old boy with no medical history was admitted to the Department of Infectious diseases at University Hospital in Marseille on May 25, 2012, with a fever (40°C) and skin lesions on his lower right eyelid. He had been fishing 7 days earlier at a pond situated in southern France near Marseille (43°26′N, 5°6′E). He had been given amoxicillin/clavulanic acid by his family doctor and showed no improvement after 2 days. The only sign on physical examination was the presence of 2 eschars on his lower right eyelid, associated with right periorbital edema (Figure) and painful right-sided cervical lymphadenopathies. Results of standard laboratory tests were normal except for the C-reactive protein level (21 mg/L; reference value <10 mg/L). He reported that the black spots on his lower eyelid were most likely related to bites from ticks that he got while fishing. He removed the ticks the next day. Because a tick-borne rickettsiosis was suspected, oral empirical treatment with doxycycline (200 mg/daily) was started. The patient improved in 48 hours and remained well (Figure).
The first serologic test result for Rickettsia spp. was negative. Because of the location of the eschars, it was not possible to obtain biopsy specimens from them. Nevertheless, real-time qPCR that was performed on 2 eschar swab specimens showed positive results for Rickettsia spp in 24 hours. The specific qPCR test results were positive for Rickettsia sibirica mongolitimonae in both samples (1).
Amplification and sequencing of a fragment of ompA gene on these samples showed 100% (533/533) identity with R. sibirica mongolitimonae HA-91 (RHU43796). Four days later, after doxycycline treatment, 1 additional swab specimen was positive by specific qPCR for R. sibirica mongolitimonae. The convalescent-phase serum specimen (obtained 14 days after admission) was positive by indirect immunofluorescence assay for rickettsial antigens against SFG, suggesting seroconversion.
R. sibirica mongolitimonae is an intracellular bacterium that was recognized as a human pathogen in 1996 (1). The inoculation eschar at the tick bite site is a hallmark of many tick-borne SPG rickettsioses. However, because lymphangitis was also observed in a few of the patients reported subsequently, R. sibirica mongolitimonae infection was named lymphangitis-associated rickettsiosis (5). To date, 24 cases have been reported in Europe (France, Spain, Portugal, Greece) and 3 in Africa (Egypt, Algeria, South Africa) (6,7). Vectors include ticks in the genus Hyalomma and also Rhipicephalus pusillus, a species of tick found on the European wild rabbit (also can be found on wild carnivorous animals, dogs, and domestic cats), which may bite humans (7). The life-threatening Mediterranean spotted fever caused by R. conorii peaks in the warmer months of July and August because of a heat-mediated increase in the aggressiveness and, therefore propensity to bite humans, of the brown dog tick vector, R. sanguineus (8). In contrast, R. sibirica mongolitimonae infection is more frequently reported in the spring (7).
The diagnosis of rickettsioses is most commonly based on serologic testing (1). However, serologic evidence of infection generally appears in the second and third weeks of illness, as in the case-patient described here. The use of molecular tools or cell culture on a skin biopsy specimen from an eschar is the best method of identifying Rickettsia spp. However, this invasive and painful procedure needs to be performed in sterile conditions with local anesthesia. Swabbing an eschar is easy and painless; the physician only needs a dry sterile swab that must be directed, while being rotated vigorously, to the base of the eschar, after the crust is removed (4). The sensitivity of this technique is comparable with that of rickettsial detection on skin biopsy samples by molecular tools. If the eschar lesion is dry, a wet compress, previously humidified with sterile water, should be placed on the inoculation eschar for 1 minute before swabbing, to increase the quantity of material swabbed. In addition, the crust eschar also can be used for rickettsial diagnosis. Because sufficient material can be obtained during swabbing, this test can be used by any practitioner at the patient’s bedside. As soon as the samples are sent to a laboratory with qPCR capability, results can be obtained quickly. In any case, when a physician is confronted with a patient with a fever and an eschar, doxycycline treatment should be initiated immediately because β-lactam antimicrobial drugs are inefficient for the treatment of rickettsioses (9).
Acknowledgments
This research was funded by the Unité de Recherche en Maladies Infectieuses et Tropicales Emergentes (URMITE), Marseille, France.
D.R. is a cofounder of the biotechnology company Inodiag (www.inodiag.com) and is the inventor of a serologic diagnostic method for which the patent is held by Aix-Marseille University; this method was not used in this study. All other authors have no conflicts of interest.
References
- Parola P, Paddock CD, Socolovschi C, Labruna MB, Mediannikov O, Kernif T, Update on tick-borne rickettsioses around the world: a geographic approach. Clin Microbiol Rev. 2013;26:657–702. DOIPubMedGoogle Scholar
- Wang JM, Hudson BJ, Watts MR, Karagiannis T, Fisher NJ, Anderson C, Diagnosis of Queensland tick typhus and African tick bite fever by PCR of lesion swabs. Emerg Infect Dis. 2009;15:963–5. DOIPubMedGoogle Scholar
- Mouffok N, Socolovschi C, Benabdellah A, Renvoise A, Parola P, Raoult D. PCR diagnosis of rickettsiosis on eschar swabs, Algeria. Emerg Infect Dis. 2011;17:1968–9. DOIPubMedGoogle Scholar
- Socolovschi C, Renzulli A, Brouqui B, Parola P, Raoult D. The use of eschar swabs for the diagnosis of African tick-bite fever. Ticks Tick-borne Dis. 2012;3:361–3.
- Fournier PE, Gouriet F, Brouqui P, Lucht F, Raoult D. Lymphangitis-associated rickettsiosis, a new rickettsiosis caused by Rickettsia sibirica mongolotimonae: seven new cases and review of the literature. Clin Infect Dis. 2005;40:1435–44. DOIPubMedGoogle Scholar
- Ramos JM, Jado I, Padilla S, Masia M, Anda P, Gutierrez F. Human infection with Rickettsia sibirica mongolitimonae, Spain, 2007–2011. Emerg Infect Dis. 2013;19:267–9. DOIPubMedGoogle Scholar
- Edouard S, Parola P, Socolovschi C, Davoust B, La Scola B, Raoult D. Clustered cases of Rickettsia sibirica mongolitimonae infection, France. Emerg Infect Dis. 2013;19:337–8. DOIPubMedGoogle Scholar
- Parola P, Socolovschi C, Jeanjean L, Bitam I, Fournier PE, Sotto A, Warmer weather linked to tick attack and emergence of severe rickettsioses. PLoS Negl Trop Dis. 2008;2:e338. DOIPubMedGoogle Scholar
- Botelho-Nevers E, Socolovschi C, Raoult D, Parola P. Treatment of Rickettsia spp. infections: a review. Expert Rev Anti Infect Ther. 2012;10:1425–37 . DOIPubMedGoogle Scholar
Figure
Cite This ArticleRelated Links
Table of Contents – Volume 20, Number 4—April 2014
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Philippe Parola, Unité de Recherche en Maladies Infectieuses et Tropicales Emergentes (URMITE), WHO Collaborative Center for Rickettsioses and Other Arthropod-borne Bacterial Diseases, Faculté de Médecine, 27 Bd Jean Moulin, 13385 Marseille Cedex 05, FrancePhilippe Parola, Unité de Recherche en Maladies Infectieuses et Tropicales Emergentes (URMITE), WHO Collaborative Center for Rickettsioses and Other Arthropod-borne Bacterial Diseases, Faculté de Médecine, 27 Bd Jean Moulin, 13385 Marseille Cedex 05, France
Top