Volume 21, Number 10—October 2015
Research
Epidemiology of Lyme Disease, Nova Scotia, Canada, 2002–2013
Cite This Article
Citation for Media
Abstract
Ixodes scapularis ticks, which transmit Borrelia burgdorferi, the causative agent of Lyme disease (LD), are endemic to at least 6 regions of Nova Scotia, Canada. To assess the epidemiology and prevalence of LD in Nova Scotia, we analyzed data from 329 persons with LD reported in Nova Scotia during 2002–2013. Most patients reported symptoms of early localized infection with rash (89.7%), influenza-like illness (69.6%), or both; clinician-diagnosed erythema migrans was documented for 53.2%. In a separate serosurvey, of 1,855 serum samples screened for antibodies to B. burgdorferi, 2 were borderline positive (both with an indeterminate IgG on Western blot), resulting in an estimated seroprevalence of 0.14% (95% CI 0.02%–0.51%). Although LD incidence in Nova Scotia has risen sharply since 2002 and is the highest in Canada (16/100,000 population in 2013), the estimated number of residents with evidence of infection is low, and risk is localized to currently identified LD-endemic regions.
Lyme disease (LD) is an emerging vector-borne infection caused by Borrelia burgdorferi, which is transmitted to humans by infected ticks. In Nova Scotia, Canada, the vector is the blacklegged tick, Ixodes scapularis. Approximately 300,000 cases of LD occur in the United States each year (1). In Canada, infected Ixodes ticks are now endemic to parts of British Columbia (I. pacificus), Manitoba, Ontario, Quebec, New Brunswick, and Nova Scotia (I. scapularis) (2). The number of Canadians with LD has risen since LD became nationally reportable in 2009; a total of 682 cases from across the country were reported to the Public Health Agency of Canada (PHAC) in 2013, which most likely underrepresents the true number (2–4). As the geographic range of I. scapularis ticks expands, more Canadians will be at risk for LD.
Since the first case of locally acquired LD was reported in 2002, the number of human infections in Nova Scotia has risen sharply; 154 cases were reported in 2013 alone. LD can manifest with localized disease, most commonly erythema migrans (EM), or disseminated illness with neurologic, cardiac, and/or joint involvement. In the United States, up to 7% of cases are asymptomatic (5). In Canada, tick surveillance, coordinated and conducted by the Nova Scotia Departments of Health and Wellness and Natural Resources and the National Microbiology Laboratory (NML) of the PHAC, has identified the establishment of infected blacklegged tick populations in 6 regions in Nova Scotia, and these ticks have been found sporadically in many other locations, suggesting potential LD risk across the province. However, contemporary LD risk has been difficult to quantify in Nova Scotia because of the dynamic and expanding nature of vector tick populations, occurrence of missed and/or asymptomatic infections, and changes in surveillance methods and case definitions. Here we describe the epidemiology of LD and the results of a 2012 provincial serosurvey for antibodies to B. burgdorferi to characterize and estimate the risk for features of LD in Nova Scotia.
LD Case Data
LD is reportable in Nova Scotia. Case data were provided by Population Health Assessment and Surveillance, Nova Scotia Department of Health and Wellness (Table 1). Public health nurses investigate all LD cases by follow-up with the health care provider and patient. Data presented were extracted from case report forms. Numbers of tests conducted by the Capital District Health Authority Division of Microbiology, the Nova Scotia testing site for LD, were extracted from the laboratory information system. Regions to which LD is endemic (endemic regions) are defined by using the Canadian national definition. A confirmed endemic region is one in which active field surveillance has identified a reproducing population of ticks confirmed by the presence of all 3 stages on resident animals or in the environment for at least 2 consecutive years, and B. burgdorferi is detected in these ticks and/or wild animal hosts in the locality (2,6). For the last 2 regions identified, all 3 stages were identified in 1 year only.
Serosurvey
We used samples of residual serum from specimens submitted for diagnostic testing that would otherwise have been discarded. Aliquots of residual serum submitted for prenatal screening, electrolyte testing, cholesterol testing, or HIV screening were collected from regional laboratories during May 1–August 30, 2012. Each serum sample was deidentified so that it could not be linked to a specific patient. These specimens were chosen with the intent of obtaining samples from healthy persons undergoing blood tests as part of a regular health check-up. Serum samples were stratified by patient age, sex, and District Health Authority (DHA), and sampling was proportionate to the Nova Scotia population in five 10-year age groups for ages 10–59 years and 1 age group for ages 60–64 years. The Research Ethics Board of each DHA approved the serosurvey. One Research Ethics Board required an opt-out option for patients undergoing blood collection during the study period, achieved by publicizing the study using posters and asking patients who did not want their serum used to identify themselves at the time of collection. No patients opted out.
Serologic Testing
We detected antibodies to B. burgdorferi using a commercially available enzyme immunoassay (EIA) that used a whole-cell sonicate of B. burgdorferi (B. burgdorferi ELISA II, Wampole Laboratories, Princeton, NJ, USA). Samples were tested at the Capital DHA microbiology laboratory. In accordance with Canadian and US guidelines (7,8), specimens that screened positive or equivocal on the EIA were sent to the NML for confirmatory testing. The NML uses a 2-tiered approach whereby specimens that test positive or equivocal by EIA are retested by using C6 ELISA (Immunetics, Boston, MA, USA) and Western blot (Euroimmun, Morris Plains, NJ, USA). Consistent with criteria from the US Centers for Disease Control and Prevention, a positive IgG Western blot (WB) (5 of 10 significant bands reactive) was considered conclusive evidence of previous infection (9). In addition, the NML has created a borderline category for serum for which 4 of 10 reactive significant bands are documented but includes an additional fifth band that is visible on the blot but is insufficiently intense to be considered reactive.
Statistics
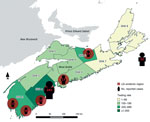
We calculated sample size for the serosurvey on the basis of an estimated seroprevalence of 1.0% ± 0.5% precision within a 95% CI, with oversampling in DHA 1, DHA 2, and DHA 3, where greater LD activity has been noted (Figure 1). Descriptive statistics were used to characterize reported LD cases and testing results from the serosurvey. Seroprevalence estimates were produced and 95% CIs calculated by using the Clopper-Pearson Exact method. Design weights accounted for regional oversampling in the provincial estimates. Statistical analyses were conducted by using SAS version 9.4 (SAS Institute, Inc., Cary, NC, USA). Maps were created by using QGIS 2.0 (http://qgis.org/en/site/). Population estimates were based on 2011 census data from Statistics Canada (10).
Endemic Regions
The first established B. burgdorferi–infected blacklegged tick population in Nova Scotia was identified in a rural region within DHA 1 in 2003. The second endemic region was identified in a park within the largest urban center in DHA 9 in 2006. In 2008, a rural region within DHA 2 was declared to be endemic, and in 2010, a rural region in DHA 6 was declared endemic. In 2011 and 2012 the fifth and sixth endemic regions were identified in other rural regions within DHAs 1 and 2, >20 km away from regions previously identified.
LD Cases
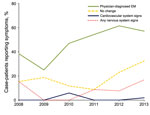
A total of 329 LD cases were reported in Nova Scotia during 2002–2013. Case-patients were a median of 56 (range 0–85) years of age, and most (76.9%) were male (Table 2). Only 26.4% of LD patients reported a definite history of a tick bite. Most reported symptoms of early localized LD, including influenza-like illness and EM or other non-EM rash. Physician-diagnosed EM was reported for 53.2% of cases. Central clearing (i.e., reduction or of the erythema near the center of the rash) was reported for 49.7%; a total of 33.7% reported no central clearing, and for 16.6%, central clearing was unknown. A total of 125 LD patients had early disseminated or late infection, including recurrent joint swelling and Bell palsy. The percentage of cases with clinician-observed EM increased over time, and brief recurrent joint swelling was reported in a greater percentage of patients in 2013 than previously (Figure 2). Thirteen LD patients were hospitalized (Table 3), but the reasons for admission were unknown.
A total of 307 (93%) case-patients reported living in or traveling to an endemic region in Nova Scotia (Figure 1). Of the remaining case-patients, 17 were infected outside the province including patients who traveled to Europe and the United States; for 5 case-patients, the location of exposure was either unknown or outside of regions where LD is known to be endemic. Most (73.3%) cases met the definition for a confirmed case. Most (71.4%) case-patients lived in DHA 1. The number of cases increased substantially during 2012–2013, attributable to increased reports of cases associated with known endemic regions (Figure 3).
A total of 263 (80%) cases had serologic evidence of infection. Of the 66 cases classified as “probable: EM + endemic exposure” that were reported to Nova Scotia public health professionals, 10 did not have any information about laboratory testing, 42 did not have serologic testing, and 14 had serologic test results that were negative by the 2-tier algorithm. Of these 14 patients, 3 were negative on the whole-cell EIA, and 11 were positive by the C6 ELISA but negative by WB.
The annual number of serologic tests for LD in Nova Scotia increased from 1,659 in 2010 to 2,421 in 2013. Testing rates varied by DHA (Figure 1), ranging from 69.1 tests per 100,000 population in DHA 8 to 1,581.9 per 100,000 population in DHA 1 in 2013. Serologic incidence of LD in 2013 varied by DHA, ranging from 0 cases per 100,000 population in DHA 8 to 206.2 per 100,000 population in DHA 1.
Serosurvey Results
Of 1,855 serum samples tested for antibodies to B. burgdorferi, 215 (11.6%) screened positive by the whole-cell EIA (Walpole Laboratories, Walpole, MA, USA) and were sent to the NML for confirmatory testing. Of these, 17 (0.9% of total serosurvey) were positive or equivocal by the C6 ELISA. None were positive by IgG WB using Centers for Disease Control and Prevention (CDC) criteria, but 2 samples were borderline positive (Table 3). If we used CDC criteria for WB interpretation, the estimated seroprevalence of LD in Nova Scotia was 0% (95% CI 0.00%–0.20%). If we considered the 2 borderline WB results or the positive C6 ELISA as evidence of exposure to B. burgdorferi, the estimated seroprevalence was 0.14% (95% CI 0.02%–0.51%) and 0.98% (95% CI 0.56%−1.60%), respectively (Table 4).
LD is emerging in Nova Scotia, and public health surveillance data have been useful for characterizing risk and describing the clinical presentation of LD. Most cases are characterized by early localized disease. Compared with data from the United States, Nova Scotia case-patients were less likely to have EM reported as the presenting manifestation (69% vs. 53%) (11). Although published studies report that up to 80% of LD case-patients have EM, these findings were in the context of a vaccine trial or active laboratory-based surveillance study in a hyperendemic region where patients were followed closely and physicians were very experienced in diagnosing EM (12,13). The public health surveillance system in Nova Scotia captures both clinician-diagnosed EM and other skin rashes. Although almost 90% of case-patients reported a rash, only 53% had clinician-diagnosed EM. However, some of the other skin rash cases are also likely to represent EM. Central clearing in only 50% of EM lesions was consistent with other reports that EM might have diffuse erythema or enhanced central erythema rather than central clearing (14). An increase in the proportion of cases with clinician-diagnosed EM over time suggests physicians have become more aware of and/or better able to diagnose EM.
The most common manifestation of late LD in Nova Scotia was arthritis. Although up to 60% of untreated LD results in arthritis, earlier recognition and treatment is expected to greatly reduce its frequency (7,15–17). The increase in the proportion of Nova Scotia case-patients reporting recurrent brief episodes of large joint swelling to 32.5% in 2013 is similar to the proportion of US cases of Lyme arthritis (30%) (11). The higher proportion of cases with arthritis in 2013 does not necessarily reflect a large proportion of missed cases during earlier years but might be related to misreporting of arthralgia and reporting bias toward serologically confirmed cases, as well as a general increase in the number of cases and duration of LD in Nova Scotia. Further investigation is needed to understand how many case-patients reporting joint symptoms fit the clinical diagnosis of Lyme arthritis.
As in the United States, other manifestations, including atrioventricular block and neuroborreliosis, were uncommon (11). Only 26% of LD patients in Nova Scotia recalled a tick bite, consistent with US data where 84% and 86% of patients with EM and Lyme arthritis, respectively, failed to recall exposure to blacklegged ticks (18–21).
The incidence of LD in Canada has increased during the last decade (2,4), and Nova Scotia has the highest reported incidence (16 cases/100,000 population in 2013). Incidence varies greatly geographically; 1 health region (DHA 1) reported case rates of ≈200/100,000 population in 2013, which is higher than those in northeastern US states (11). The increased incidence most likely resulted from the increased number of established blacklegged tick populations; increases in the size, geographic range, and pathogen prevalence within these populations; increasing rates of human infection; and better identification and reporting of LD. Infected blacklegged ticks can be sporadically identified in other regions of the province, but only 1.5% of LD cases were not linked to known endemic regions, suggesting the risk remains localized. Our serosurvey did not identify any persons with B. burgdorferi antibodies based on CDC criteria, and even if the 2 borderline results were included, the estimated provincial seroprevalence was much less than 1%. Even in DHAs known to contain endemic foci of LD, we failed to detect any positive serum and found an estimated seroprevalence of <2% in the DHA with the highest reported LD incidence. B. burgdorferi seroprevalence in the northeastern United States, with similar environmental conditions to Nova Scotia, ranges from 0% to 18.8% (22,23). The fact that tick populations endemic to the northeastern United States have been established for much longer than in Nova Scotia might explain this difference. Widespread clinical testing, coupled with the serosurvey results, suggests LD cases most likely are not being missed in Nova Scotia.
Although all of the serum used for the serosurvey was negative by 2-tier testing, 8% of serum samples positive by whole-cell EIA (17/215) were positive on the C6 ELISA. Although some data suggest that classifying LD on the basis of C6 alone is more sensitive than the 2-tier algorithms (24), the potential exists for false-positive and false-negative results. The literature suggests that the C6 ELISA specificity is 98.4%–98.6% (24,25). However, when used as the supplemental test to positive whole-cell lysate EIA, as in our study, its specificity is estimated at 99.1%–99.8% (26). The 17 positive C6 results in our study could be in keeping with false-positive results, with this reported specificity. Alternatively, these reactive C6 serum samples could represent patients infected with B. burgdorferi and treated early with appropriate antimicrobial drugs. However, because the serum used could not be linked to patients, we have no clinical information to support this possibility. Data suggest that the C6 ELISA has a higher sensitivity for early infection and can be positive before the IgG WB completely matures to include the required 5/10 bands (27). In addition, evidence suggests that patients treated early in infection abort the seroconversion response, and a positive IgG WB might not develop (27,28). Another possibility is that the positive C6 ELISA results from cross-reactivity with another Borrelia species, such as B. miyamotoi, which has been identified in blacklegged ticks in the United States and Canada, including Nova Scotia (29,30). Using EIA and WB assays specific for B. miyamotoi, Krause et al. found serologic evidence of acute infection in patients living in endemic regions who had a viral-like illness (31). Although a recent study found 2 of 34 ticks from Nova Scotia submitted as part of passive surveillance had positive PCR for B. miyamotoi (30), no data are available that examined the potential cross-reactivity of whole-cell sonicate or C6 B. burgdorferi EIAs with patient serum containing antibodies to B miyamotoi. If these hypotheses are correct, we should have seen a disproportionate number of positive serum samples from regions with the highest risk for exposure to infected blacklegged ticks, such as DHA 1, where an infected population of blacklegged ticks has been established since 2003 and where most case-patients reside and were exposed. However, seroprevalence of C6 positive serum did not differ significantly among any of the DHAs.
Our study has several limitations. Our clinical data are limited to the case report forms used by Nova Scotia public health professionals; thus, the precision regarding the clinical presentation is limited. For example, the inclusion of categories of influenza-like illness is not specific and could represent a respiratory illness. Furthermore, the case reports do not always differentiate between single EM and multiple EM, so classification of early localized and early disseminated infection was not possible. Early LD, the most common presentation, is predominantly a clinical diagnosis for which sensitivity of serologic testing is poor (<50%) and not recommended. Thus, surveillance data might underestimate incidence if not all clinical cases are captured. We recognize that estimating the extent of underreporting is difficult and that underreporting could vary geographically. However, in the cohort reported here, 42 of 66 cases classified as “probable: EM + endemic exposure” followed the Infectious Diseases Society of America guidelines (7) and were not tested serologically. These reports have come from 5 different districts across the province suggesting that physicians are reporting at least some B. burgdorferi infections on the basis of clinical presentation alone.
Another limitation is that the serosurvey samples might not be representative of the population at risk. We used samples of residual serum from diagnostic testing that might be biased toward a population with more medical co-morbidities or different risk and health-seeking behaviors (32). Our study attempted to reduce this bias by selecting samples that were originally collected for routine diagnostic testing, with the aim of capturing persons undergoing routine well-person screening. However, this sample will exclude persons who generally do not access regular medical care. Despite this limitation, the sampling method has been used for other infections and is thought to provide an acceptable balance between representativeness and feasibility (both practical and financial). In fact, the only published study that has compared the residual serum approach with population-based sampling yielded comparable estimates of immunity against 5 vaccine-preventable diseases, with an ≈7-fold increased cost for the population-based approach (33). Still, risk for exposure to blacklegged ticks through outdoor activity in endemic regions was unavailable for serosurvey specimens and probably has greater heterogeneity than probability of vaccination. Also, our sample did not include the 0–9- or >65-year age groups.
Although LD incidence is increasing in Nova Scotia, infections appear to be restricted to regions within the province where populations of infected blacklegged ticks are known to be endemic. These findings support a targeted approach to public health risk messaging. Our seroprevalence study suggests that <1% of Nova Scotia residents have been exposed to B. burgdorferi. However, as tick populations continue to expand, we expect LD rates to continue to increase. Residents of, and travelers, to Nova Scotia need to be vigilant and take precautions to reduce their risk for LD when they venture into regions where ticks are present. Because only a minority of patients will report a tick bite, physicians should be aware of the manifestations of LD and consider it when patients have compatible symptoms and exposure to an endemic region, through residence or travel, in Nova Scotia.
Dr. Hatchette is the head and service chief for the Division of Microbiology in the Department of Pathology and Laboratory Medicine, Central Zone, Nova Scotia Health Authority, and professor of pathology at Dalhousie University. His research interests include diagnostic virology and emerging pathogens.
Acknowledgments
We thank Colleen Jackson, Charles Heinstein, Carol Pelton, Catherine Roberts, Katrina Bernat, and Antonia Dibernardo for their help in processing and testing the specimens. We are indebted to the public health nurses and the laboratory staff in the regional laboratories across the Province of Nova Scotia, without whom this project and manuscript would not have been possible.
The Capital Health Research Fund and the Nova Scotia Department of Health and Wellness provided funding for this study.
References
- Kuehn BM. CDC estimates 300,000 US cases of Lyme disease annually. JAMA. 2013;310:1110 . DOIPubMedGoogle Scholar
- Ogden NH, Koffi JK, Pelcat Y, Lindsay LR. Environmental risk from Lyme disease in central and eastern Canada: a summary of recent surveillance information. Can Commun Dis Rep. 2014;40:74–83.
- Henry B, Roth D, Reilly R, MacDougall L, Mak S, Li M, How big is the Lyme problem? Using novel methods to estimate the true number of Lyme disease cases in British Columbia residents from 1997 to 2008. Vector Borne Zoonotic Dis. 2011;11:863–8 . DOIPubMedGoogle Scholar
- Public Health Agency of Canada. Lyme disease and other tick-borne diseases: information for healthcare professionals [cited 2015 Mar 26]. http://www.phac-aspc.gc.ca/id-mi/tickinfo-eng.php#sec-2
- Steere AC, Sikand VK, Schoen RT, Nowakowski J. Asymptomatic infection with Borrelia burgdorferi. Clin Infect Dis. 2003;37:528–32. DOIPubMedGoogle Scholar
- Wormser GP, Dattwyler RJ, Shapiro ED, Halperin JJ, Steere AC, Klempner MS, The clinical assessment, treatment, and prevention of Lyme disease, human granulocytic anaplasmosis, and babesiosis: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2006;43:1089–134. DOIPubMedGoogle Scholar
- Canadian Public Health Laboratory Network. The laboratory diagnosis of Lyme borreliosis: guidelines from the Canadian Public Health Laboratory Network. Can J Infect Dis Med Microbiol. 2007;18:145–8 .PubMedGoogle Scholar
- Centers for Disease Control and Prevention. Recommendations for test performance and interpretation from the Second National Conference on Serologic Diagnosis of Lyme Disease. MMWR Morb Mortal Wkly Rep. 1995;44:590–1 .PubMedGoogle Scholar
- Statistics Canada. Table 051–0001—Estimates of population, by age group and sex for July 1, Canada, provinces and territories, annual (persons unless otherwise noted) [cited 2013 Jul 31]. http://www5.statcan.gc.ca/cansim/
- Bacon RM, Kugeler KJ, Mead PS; Centers for Disease Control and Prevention. Surveillance for Lyme disease—United States, 1992–2006. MMWR Surveill Summ. 2008;57:1–9 .PubMedGoogle Scholar
- Steere AC, Sikand VK. The presenting manifestations of Lyme disease and the outcomes of treatment. N Engl J Med. 2003;348:2472–4. DOIPubMedGoogle Scholar
- Petersen LR, Sweeney AH, Checko PJ, Magnarelli LA, Mshar PA, Gunn RA, Epidemiological and clinical features of 1,149 persons with Lyme disease identified by laboratory-based surveillance in Connecticut. Yale J Biol Med. 1989;62:253–62 .PubMedGoogle Scholar
- Steere AC, Schoen RT, Taylor E. The clinical evolution of Lyme arthritis. Ann Intern Med. 1987;107:725–31. DOIPubMedGoogle Scholar
- Tory HO, Zurakowski D, Sundel RP. Outcomes of children treated for Lyme arthritis: results of a large pediatric cohort. J Rheumatol. 2010;37:1049–55. DOIPubMedGoogle Scholar
- Daikh BE, Emerson FE, Smith RP, Lucas FL, McCarthy CA. Lyme arthritis: a comparison of presentation, synovial fluid analysis, and treatment course in children and adults. Arthritis Care Res (Hoboken). 2013;65:1986–90 . DOIPubMedGoogle Scholar
- Nadelman RB, Wormser GP. Erythema migrans and early Lyme disease. Am J Med. Am J Med. 1995;98:15S–23S. DOIPubMedGoogle Scholar
- Müllegger RR, Glatz M. Skin manifestations of Lyme borreliosis: diagnosis and management. Am J Clin Dermatol. 2008;9:355–68. DOIPubMedGoogle Scholar
- Thompson A, Mannix R, Bachur R. Acute pediatric monoarticular arthritis: distinguishing Lyme arthritis from other etiologies. Pediatrics. 2009;123:959–65. DOIPubMedGoogle Scholar
- Kowalski TJ, Tata S, Berth W, Mathiason MA, Agger WA. Antibiotic treatment duration and long-term outcomes of patients with early Lyme disease from a Lyme disease–hyperendemic area. Clin Infect Dis. 2010;50:512–20 . DOIPubMedGoogle Scholar
- Steere AC, Taylor E, Wilson ML, Levine JF, Spielman A. Longitudinal assessment of the clinical and epidemiological features of Lyme disease in a defined population. J Infect Dis. 1986;154:295–300. DOIPubMedGoogle Scholar
- Hanrahan JP, Benach JL, Coleman JL, Bosler EM, Morse DL, Cameron DJ, Incidence and cumulative frequency of endemic Lyme disease in a community. J Infect Dis. 1984;150:489–96. DOIPubMedGoogle Scholar
- Wormser GP, Schriefer M, Aguero-Rosenfeld ME, Levin A, Steere AC, Nadelman RB, Single-tier testing with the C6 peptide ELISA kit compared with two-tier testing for Lyme disease. Diagn Microbiol Infect Dis. 2013;75:9–15. DOIPubMedGoogle Scholar
- Branda JA, Linskey K, Kim YA, Steere AC, Ferraro MJ. Two-tiered antibody testing for Lyme disease with use of 2 enzyme immunoassays, a whole-cell sonicate enzyme immunoassay followed by a VlsE C6 peptide enzyme immunoassay. Clin Infect Dis. 2011;53:541–7. DOIPubMedGoogle Scholar
- Wormser GP, Levin A, Soman S, Adenikinju O, Longo MV, Branda JA. Comparative cost-effectiveness of two-tiered testing strategies for serodiagnosis of Lyme disease with noncutaneous manifestations. J Clin Microbiol. 2013;51:4045–9. DOIPubMedGoogle Scholar
- Steere AC, McHugh G, Damle N, Sikand VK. Prospective study of serologic tests for Lyme disease. Clin Infect Dis. 2008;47:188–95 and. DOIPubMedGoogle Scholar
- Aguero-Rosenfeld ME, Nowakowski J, Bittker S, Cooper D, Nadelman RB, Wormser GP. Evolution of the serologic response to Borrelia burgdorferi in treated patients with culture-confirmed erythema migrans. J Clin Microbiol. 1996;34:1–9 .PubMedGoogle Scholar
- Salkeld DJ, Cinkovich S, Nieto NC. Tick-borne pathogens in northwestern California, USA. Emerg Infect Dis. 2014;20:493–4. DOIPubMedGoogle Scholar
- Dibernardo A, Cote T, Ogden NH, Lindsay LR. The prevalence of Borrelia miyamotoi infection, and co-infections with other Borrelia spp. in Ixodes scapularis ticks collected in Canada. Parasit Vectors. 2014;7:183. PMID: 24731287
- Krause PJ, Narasimhan S, Wormser GP, Rollend L, Fikrig E, Lepore T, Human Borrelia miyamotoi infection in the United States. N Engl J Med. 2013;368:291–3. DOIPubMedGoogle Scholar
- Osborne K, Gay N, Hesketh L, Morgan-Capner P, Miller E. Ten years of serological surveillance in England and Wales: methods, results, implications and action. Int J Epidemiol. 2000;29:362–8. DOIPubMedGoogle Scholar
- Kelly H, Riddell MA, Gidding HF, Nolan T, Gilbert GL. A random cluster survey and a convenience sample give comparable estimates of immunity to vaccine preventable diseases in children of school age in Victoria, Australia. Vaccine. 2002;20:3130–6 . DOIPubMedGoogle Scholar
Figures
Tables
Cite This ArticleTable of Contents – Volume 21, Number 10—October 2015
| EID Search Options |
|---|
|
|
|
|
|
|

Please use the form below to submit correspondence to the authors or contact them at the following address:
Todd F. Hatchette, Division of Microbiology, Department of Pathology and Laboratory Medicine, QE II Health Science Center, Rm 315 Mackenzie Bldg, 5788 University Ave, Halifax, NS B3H 1V8, Canada
Top