Volume 22, Number 4—April 2016
Dispatch
Definitive Hosts of Versteria Tapeworms (Cestoda: Taeniidae) Causing Fatal Infection in North America
Figure 1
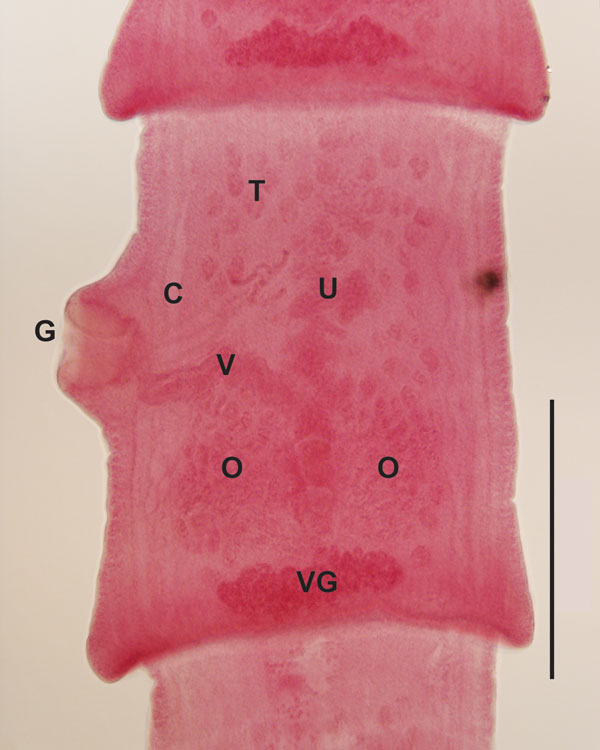
Figure 1. Microscope image of a mature segment of an adult Versteria sp. tapeworm recovered from an ermine in Wisconsin, USA (original magnification ×10). Characteristic reproductive structures are visible, including genital pore (G), cirrus sac (C), vagina (V), ovary (O), testes (T), uterine stem (U), and vitelline gland (VG). Tapeworm specimens were preserved in 70% ethanol for concurrent morphologic and molecular analyses. A series of proglottids was subsampled from each worm as a basis for sequencing; remaining strobila was stained, cleared and mounted in Canada balsam as permanent vouchers based on standard methods (5). Specimens are deposited in the Museum of Southwestern Biology, Parasitology Division, University of New Mexico, Albuquerque, New Mexico, USA (accession no. MSB 23169), and in the collections of the Denver Museum of Nature and Science, Denver, Colorado, USA (accession no. DZTM.3170). Scale bar indicates 500 µm.
References
- Nakao M, Lavikainen A, Iwaki T, Haukisalmi V, Konyaev S, Oku Y, Molecular phylogeny of the genus Taenia (Cestoda: Taeniidae): proposals for the resurrection of Hydatigera Lamarck, 1816 and the creation of a new genus Versteria. Int J Parasitol. 2013;43:427–37. DOIPubMedGoogle Scholar
- Goldberg TL, Gendron-Fitzpatrick A, Deering KM, Wallace RS, Clyde VL, Lauck M, Fatal metacestode infection in Bornean orangutan caused by unknown Versteria species. Emerg Infect Dis. 2014;20:109–13. DOIPubMedGoogle Scholar
- Loos-Frank B. An up-date of Verster’s (1969) ‘Taxonomic revision of the genus Taenia Linnaeus’ (Cestoda) in table format. Syst Parasitol. 2000;45:155–84. DOIPubMedGoogle Scholar
- Baer JG, Fain A. Newly discovered Cestoda in Belgian Congo [in French]. Acta Trop. 1951;8:59–63 .PubMedGoogle Scholar
- Pritchard MH, Kruse GOW. The collection and preservation of animal parasites. Lincoln (Nebraska): University of Nebraska Press; 1982.
- Tamura K, Stecher G, Peterson D, Filipski A, Kumar S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol. 2013;30:2725–9. DOIPubMedGoogle Scholar
- Smith MF, Patton JL. The diversification of South American murid rodents: evidence from mitochondrial DNA sequence data for the akodontine tribe. Biol J Linn Soc Lond. 1993;50:149–77. DOIGoogle Scholar
- Fleming MA, Cook JA. Phylogeography of endemic ermine (Mustela erminea) in southeast Alaska. Mol Ecol. 2002;11:795–807. DOIPubMedGoogle Scholar
- Dawson NG, Hope AG, Talbot SL, Cook JA. A multilocus evaluation of ermine (Mustela erminea) across the Holarctic, testing hypotheses of Pleistocene diversification in response to climate change. J Biogeogr. 2014;41:464–75. DOIGoogle Scholar
- Zhao F, Ma JY, Cai HX, Su JP, Hou ZB, Zhang TZ, Molecular identification of Taenia mustelae cysts in subterranean rodent plateau zokors (Eospalax baileyi). Dongwuxue Yanjiu. 2014;35:313–8.
- Hoberg EP, Galbreath KE, Cook JA, Kutz SJ, Polley L. Northern host–parasite assemblages: history and biogeography on the borderlands of episodic climate and environmental transition. Adv Parasitol. 2012;79:1–97. DOIPubMedGoogle Scholar
- Whitfield PJ, Evans NA. Parthenogenesis and asexual multiplication among parasitic platyhelminths. Parasitology. 1983;86:121–60. DOIPubMedGoogle Scholar
- Hoberg EP, Ebinger W, Render JA. Fatal cysticercosis by Taenia crassiceps (Cyclophyllidea: Taeniidae) in a presumed immunocompromised canine host. J Parasitol. 1999;85:1174–8. DOIPubMedGoogle Scholar
Medline reports the last page should be "183" not "84" in reference 3 "Loos-Frank, 2000".
Medline indexes "Biol J Linn Soc Lond" but cannot find a listing for reference 7 "Smith, Patton, 1993". Please check the reference for accuracy.
Medline indexes "J Biogeogr" but cannot find a listing for reference 9 "Dawson, Hope, Talbot, Cook, 2014". Please check the reference for accuracy.