MERS-CoV Antibodies in Humans, Africa, 2013–2014
Anne M. Liljander
1, Benjamin Meyer
1, Joerg Jores, Marcel A. Müller, Erik Lattwein, Ian Njeru, Bernard Bett, Christian Drosten

, and Victor Max Corman
Author affiliations: International Livestock Research Institute, Nairobi, Kenya (A. Liljander, J. Jores, B. Bett); University of Bonn Medical Centre, Bonn, Germany (B. Meyer, M.A. Müller, C. Drosten, V.M. Corman); University of Bern, Vetsuisse Faculty, Bern, Switzerland (J. Jores); EUROIMMUN AG, Lübeck, Germany (E. Lattwein); Ministry of Health, Nairobi, Kenya (I. Njeru); German Centre for Infection Research, Bonn (C. Drosten, V.M. Corman)
Main Article
Figure 2
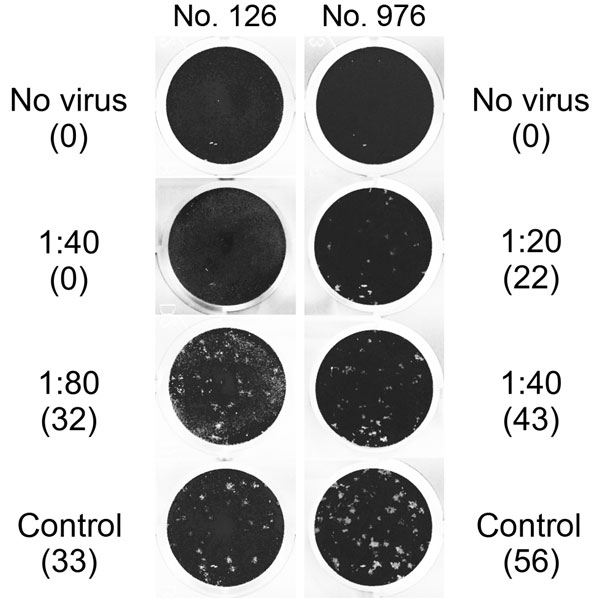
Figure 2. Middle East respiratory syndrome coronavirus (MERS-CoV) plaque-reduction neutralization test (PRNT) results for 2 serum samples positive by recombinant ELISA, showing virus neutralization activity against MERS-CoV strain EMC/2012 exceeding a titer of 1:10. Titers and number of plaques (in parenthesis) are shown next to the corresponding images. Sample no. 976 showed >50% plaque reduction up to a titer of 1:20, and sample no. 126 showed >90% plaque reduction up to a titer of 1:40. No serum was added to the control wells. Note that the image cannot represent the morphology and the contrast of plaques that was visible with direct inspection of cell culture plates with an appropriate light source, as was done for these experiments.
Main Article
Page created: May 16, 2016
Page updated: May 16, 2016
Page reviewed: May 16, 2016
The conclusions, findings, and opinions expressed by authors contributing to this journal do not necessarily reflect the official position of the U.S. Department of Health and Human Services, the Public Health Service, the Centers for Disease Control and Prevention, or the authors' affiliated institutions. Use of trade names is for identification only and does not imply endorsement by any of the groups named above.
