Volume 23, Number 3—March 2017
Dispatch
Mycobacterium chimaera in Heater–Cooler Units in Denmark Related to Isolates from the United States and United Kingdom
Cite This Article
Citation for Media
Abstract
Mycobacterium chimaera was present at high rates (>80%) in heater–cooler units (HCUs) from all 5 thoracic surgery departments in Denmark. Isolates were clonal to HCU-associated isolates from the United States (including some from patients) and United Kingdom. However, M. chimaera from 2 brands of HCU were genetically distinct.
Based on reports from 2015 (1,2), the European Centre for Disease Prevention and Control issued a Rapid Risk Assessment alert on April 30, 2015, associating invasive cardiovascular infections with Mycobacterium chimaera in water tanks of heater–cooler units (HCUs) used during open-chest heart and vascular surgery (3). Subsequently, additional cases from Europe (4) and the United States potentially associated with HCUs have been described (5–7). Preliminary data indicate that the isolates from the patients, the HCUs in hospitals, and the HCUs at the manufacturer are similar (8). The aim of this study was to determine M. chimaera prevalence in Denmark HCUs and, if present, phylogenetically characterize and quantify the strains.
Statens Serum Institut, the Danish Patient Safety Authority, and the Danish Medicines Agency decided to investigate all the HCUs in Denmark. Approval from a human or animal research ethics board was not required to conduct this study. The infection prevention control units and thoracic surgery department staff from 5 local hospitals were instructed to collect water and biofilm samples from the HCUs and send them to Statens Serum Institut for testing. In brief, a water culture method adapted for low concentrations of mycobacteria with high concentrations of contaminants was used, and mycobacterial isolates were identified by internal transcribed spacer sequencing (Technical Appendix). One M. chimaera isolate from each thoracic surgery department and one unrelated patient isolate were subjected to whole-genome sequencing (WGS). A total of 10 million paired-end Illumina sequencing reads (Illumina Denmark ApS, Copenhagen, Denmark) were deposited in the European Nucleotide Archive under study number PRJEB18427.
M. chimaera was found in 18/21 (86%) HCUs, representing all 5 thoracic surgery departments in Denmark (Table). Four sites used the Sorin 3T HCU (Sorin Group, Arvada, CO, USA); 14/16 (88%) units contained M. chimaera. One site used Maquet brand HCUs (Maquet, Wayne, NJ, USA), 4/5 (80%) units contained M. chimaera. The strain M. gordonae was found irregularly throughout the HCUs (Table). Both water and biofilm samples could be cultured and were equally effective for the detection of mycobacteria. We used the filter culture method for quantitative analysis purposes and for a simpler workflow. However, the quantitative culture results were poor quality because the analytic sensitivity was low and many samples were heavily contaminated (Table; Technical Appendix Figure).
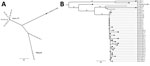
WGS analysis (Figure) showed that the 4 isolates from the Denmark Sorin 3T HCUs were nearly identical (<3 single nucleotide polymorphisms [SNPs]). Conversely, the isolate collected from the Maquet HCU was genetically distinct, showing 47–49 SNP differences compared with the isolates from the Sorin 3T HCUs. The unrelated patient isolate was not closely related to the HCU isolates (30–37 SNP differences).
We compared the sequences we collected with 31 other sequenced M. chimeaera isolates previously collected (2009–2016) and available in the European Nucleotide Archive (http://www.ebi.ac.uk/ena). This dataset comprised 3 non–HCU-associated patient isolates from Ireland; 9 Sorin 3T HCU isolates and 11 HCU-associated patient isolates from Pennsylvania and Iowa, USA (6); and 8 HCU water sample isolates from the United Kingdom (accession nos. PRJNA294775, PRJNA344472, PRJNA345021, and PRJNA324238 for the 4 groups, respectively). Unexpectedly, the M. chimaera sequences from the Denmark Sorin 3T HCUs were nearly identical to the isolates from the United States and United Kingdom (median difference 3 SNPs; interquartile range [IQR] 1–5 SNPs) and were similar to all Sorin 3T-associated patient isolates (median difference 6 SNPs; IQR 3–9 SNPs). We saw a distinctly closer relationship between the isolates from Denmark Sorin 3T HCUs and the isolates from UK and US HCUs than between Denmark Sorin 3T HCUs and the unrelated Denmark or Ireland patient isolates or the Denmark Maquet HCU isolate (Figure).
Overall, the 32 isolates associated with the Sorin 3T HCUs (online Technical Appendix Table) were found to have 15 common SNPs and 0–18 SNP differences between any 2 isolates (median difference 5 SNPs; IQR 3–8 SNPs). These findings support the conclusion by Haller et al. that M. chimaera from the Sorin 3T HCUs have a common source (8). The M. chimaera sequences from the UK HCU water samples were genetically nearly identical to the US and Denmark isolates; we therefore conclude that the UK isolates also originated from Sorin 3T HCUs.
No patients with M. chimaera infections associated with open-chest surgery have been suspected or detected in Denmark. Searching the International Reference Laboratory of Mycobacteriology database, which includes all mycobacteria cultures in Denmark, from 1991 to 2016, we found no records of M. avium complex isolates from patients that had an open-chest operation.
Following our findings, 1 thoracic surgery department decided to keep the HCUs in the operating theater but encased them in housings with separate ventilation. Two departments were unable to take the HCUs out of the theaters but decided to move the HCUs as distant as possible from the patients and decontaminate more frequently. Two of the departments had their HCUs outside the operating room already and therefore kept their policies regarding HCUs.
We found that M. chimaera was present in most HCUs in Denmark. Isolates from Sorin 3T brand HCUs were identical to the HCU isolates from the United States and the United Kingdom, and thus they appear to have the same origin. Because all 5 of the thoracic surgery departments in Denmark had contaminated HCUs and because mycobacterial contamination has been reported in multiple published studies during 2015–2016 (4–6), we find it likely that most Sorin 3T HCUs made in the past 8–10 years potentially are contaminated by the same M. chimaera strain. In addition, because 80% of the Maquet HCUs also contained M. chimaera, although phylogenetically different from the Sorin 3T strains, we suggest mycobacterial contamination might be a general problem for HCUs.
Dr. Svensson specializes in clinical microbiology and works at Statens Serum Institut in Denmark as Technical Director for Diagnostics at the International Reference Laboratory of Mycobacteriology. His research interests are molecular epidemiology, mycobacteria, and pharmacodynamics of antibiotics.
References
- Sax H, Bloemberg G, Hasse B, Sommerstein R, Kohler P, Achermann Y, et al. Prolonged outbreak of Mycobacterium chimaera infection after open-chest heart surgery. Clin Infect Dis. 2015;61:67–75. DOIPubMedGoogle Scholar
- Kohler P, Kuster SP, Bloemberg G, Schulthess B, Frank M, Tanner FC, et al. Healthcare-associated prosthetic heart valve, aortic vascular graft, and disseminated Mycobacterium chimaera infections subsequent to open heart surgery. Eur Heart J. 2015;36:2745–53. DOIPubMedGoogle Scholar
- European Centre for Disease Prevention and Control. Invasive cardiovascular infection by Mycobacterium chimaera. 2015 Apr 30 [cited 2016 Jul 15]. http://ecdc.europa.eu/en/publications/Publications/mycobacterium-chimaera-infection-associated-with-heater-cooler-units-rapid-risk-assessment-30-April-2015.pdf
- European Centre for Disease Prevention and Control. Invasive cardiovascular infection by Mycobacterium chimaera associated with the 3T heater-cooler system used during open-heart surgery. 2016 Nov 18 [cited 2016 Nov 30]. http://ecdc.europa.eu/en/publications/Publications/RRA-mycobacterium-chimaera-November-2016.pdf
- Public Health England. Infections associated with heater cooler units used in cardiopulmonary bypass and ECMO: information for healthcare providers in England. 2015 Nov 5 [cited 2016 Oct 17]. https://www.gov.uk/government/publications/infections-associated-with-heater-cooler-units-used-in-cardiopulmonary-bypass-and-ecmo
- Perkins KM, Lawsin A, Hasan NA, Strong M, Halpin AL, Rodger RR, et al. Notes from the field: Mycobacterium chimaera contamination of heater-cooler devices used in cardiac surgery–United States. MMWR Morb Mortal Wkly Rep. 2016;65:1117–8. DOIPubMedGoogle Scholar
- Tan N, Sampath R, Abu Saleh OM, Tweet MS, Jevremovic D, Alniemi S, et al. Disseminated Mycobacterium chimaera infection after cardiothoracic surgery. Open Forum Infect Dis. 2016;3:ofw131. DOIPubMedGoogle Scholar
- Haller S, Höller C, Jacobshagen A, Hamouda O, Abu Sin M, Monnet DL, et al. Contamination during production of heater-cooler units by Mycobacterium chimaera potential cause for invasive cardiovascular infections: results of an outbreak investigation in Germany, April 2015 to February 2016. Euro Surveill. 2016;21:30215. DOIPubMedGoogle Scholar
Figure
Table
Cite This ArticleTable of Contents – Volume 23, Number 3—March 2017
| EID Search Options |
|---|
|
|
|
|
|
|
Please use the form below to submit correspondence to the authors or contact them at the following address:
Erik Svensson, International Reference Laboratory of Mycobacteriology, Statens Serum Institut, Artillerivej 5, DK-2300 Copenhagen S, Denmark
Top